Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Synthetic rubbers are usually obtained by copolymeisation of |
|
Answer» ALKANE and 1, 3 - butandience |
|
| 2. |
Synthetic rubber is a polymer which resrmble natural rubber is: |
|
Answer» Neoprene |
|
| 3. |
Synthetic rubber is a copolymer of which ? |
|
Answer» Buta-1,3-diene |
|
| 4. |
Synthetic rubber is: |
|
Answer» Polyester |
|
| 5. |
Synthetic rubber is : |
|
Answer» POLYAMIDE |
|
| 6. |
Synthetic rubber Buna-N is a polymer of |
|
Answer» `H_(2)C = CH - CH = CH_(2) " and " C_(6)H_(5)- CH =CH_(2)` |
|
| 7. |
Synthetic polymers like polystyrene, silicones and PVC, are ............ |
| Answer» SOLUTION :COLLOIDS | |
| 8. |
Synthetic polymers do not degrade in the environment for a long time. How can biodegradable synthetic polymers be made. Differentiate between biopolymers and biodegradable polymers and give examples of each type. |
|
Answer» Solution :SYNTHETIC polymers are quite resistant to ENVIRONMENTAL degradation process and are responsible for the accumulation of polymeric solid WASTE materials. New biodegradable synthetic polymers have been designed and developed. These polymers contain similar functional group as present in biopolymers. e.g., : aliphatic polyesters Biopolymers can or can.t be biodogradable e.g. : Protein, starch etc., biodegradable but Keratin are non-biodegradabele. Biodegradable POLYMER are polymers which can be degradable always. e.g. : PHBV |
|
| 9. |
Synthetic polymer prepared by using caprolactam is known as |
|
Answer» terylene 
|
|
| 10. |
Synthetic human hair wigs are made from a Co-polymer of vinyl chloride and acrylonitrile and is called: |
|
Answer» PVC |
|
| 11. |
Synthetic fibres like nylon -66 are very strong because |
|
Answer» They have high MOLECULAR weight and high melting points |
|
| 12. |
Synthetic human hair wigs are made from a copolymer of vinyl chloride and acrylonitrile and is called : |
|
Answer» PVC |
|
| 13. |
Synthetic detergents are more effective in hard water than soaps because |
|
Answer» they are non-ionic |
|
| 14. |
Synthetic compounds which imprint a sweet sensation and possess no or negligible nutritional value are called ............... |
| Answer» SOLUTION :ARTIFICIAL sweatness | |
| 15. |
Synthetic biopolymer , PHBV is made up to the followingmonomers , |
|
Answer» 3-hydroxybutanoic acid +3-hydroxypentanoic acid |
|
| 16. |
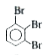
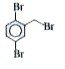
Synthesise the following compounds : a. b.Benzene rarr 2,4-D (2,4-Dichlorophenoxy acetic acid, broad leaf week killer) c.Nitrobenzene rarr PAS (p-Amini salicylic acid, Aniti-tuberculosis drug) d. e.Chlorobenzene rarr f.Phenol rarr overset (Methyl salicylate) underset (Oil of wintergreen, a flavouring agent) g. h.Benezene rarr |
Answer» SOLUTION :a.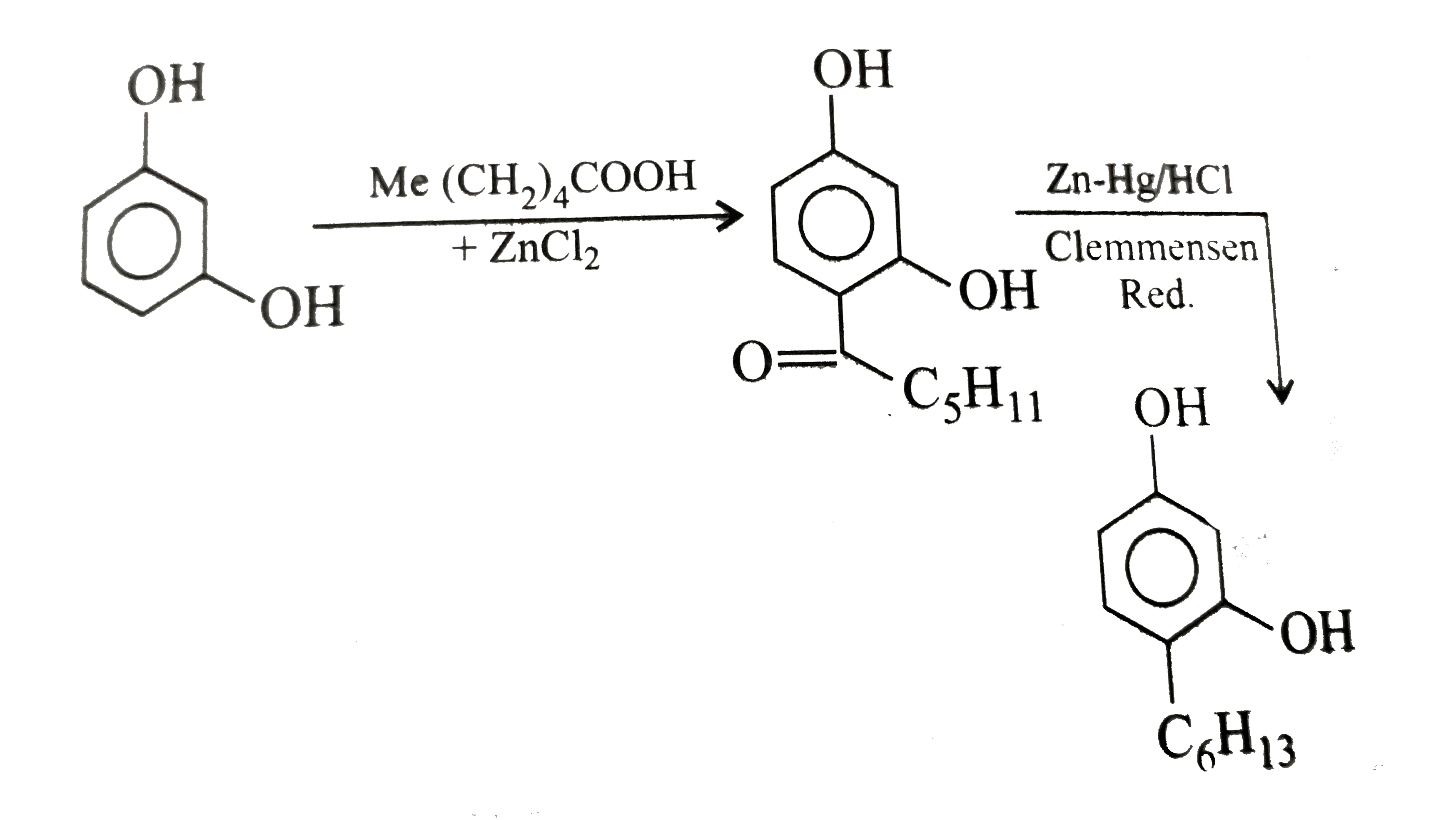 B. 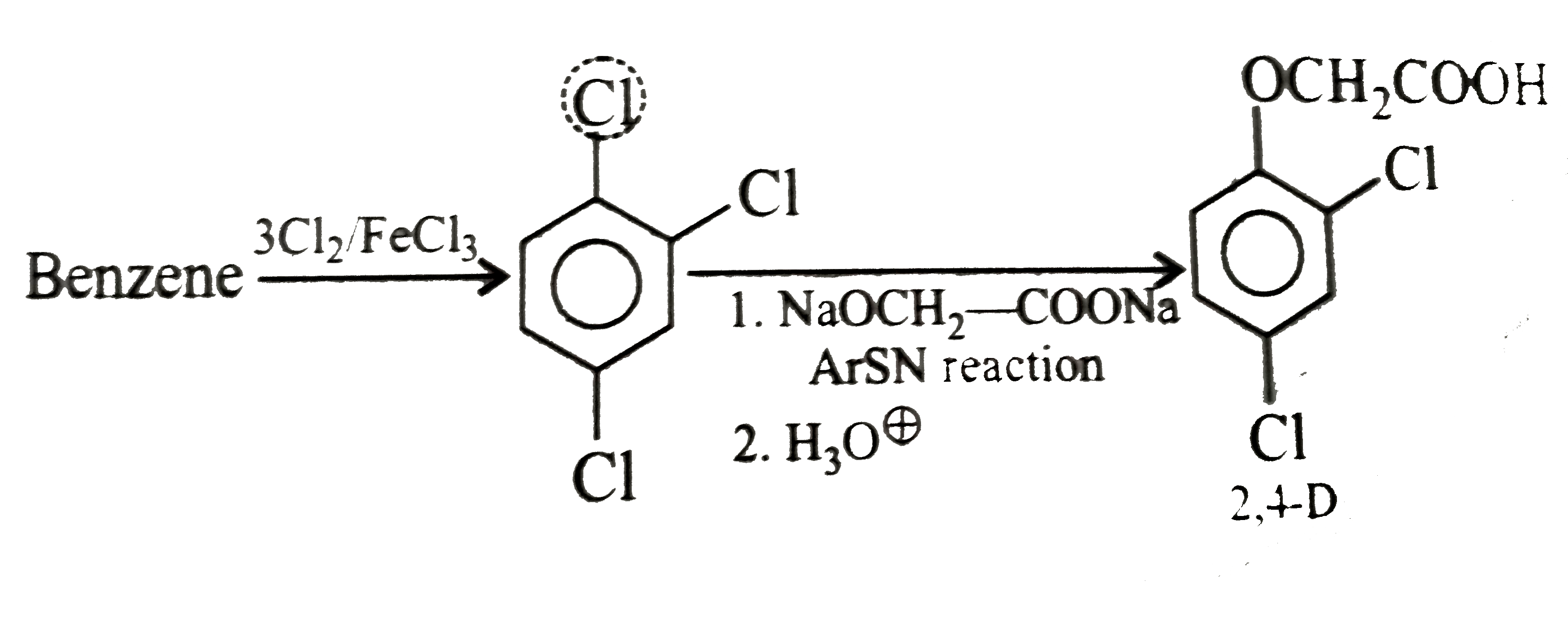 c. 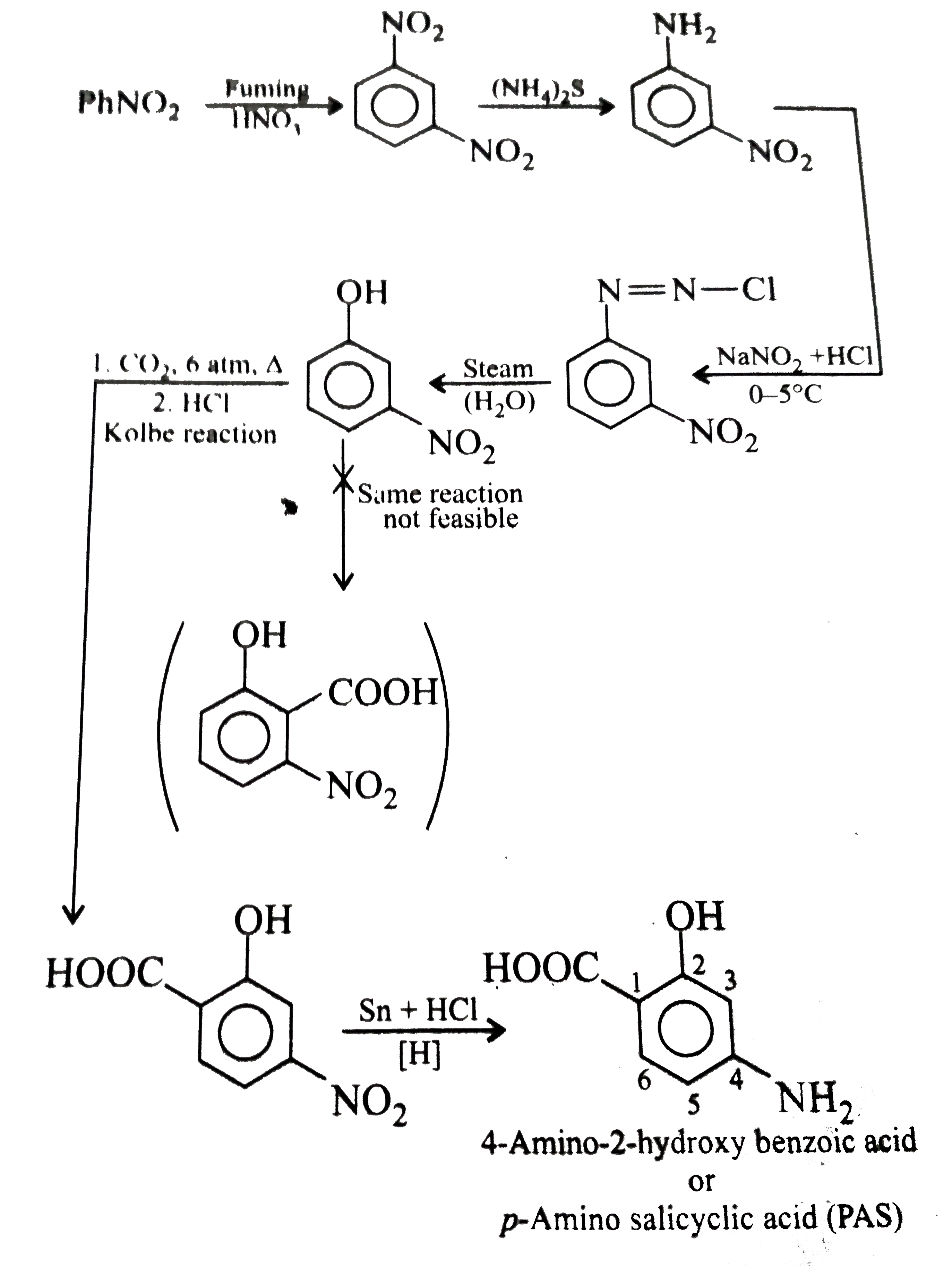 d. 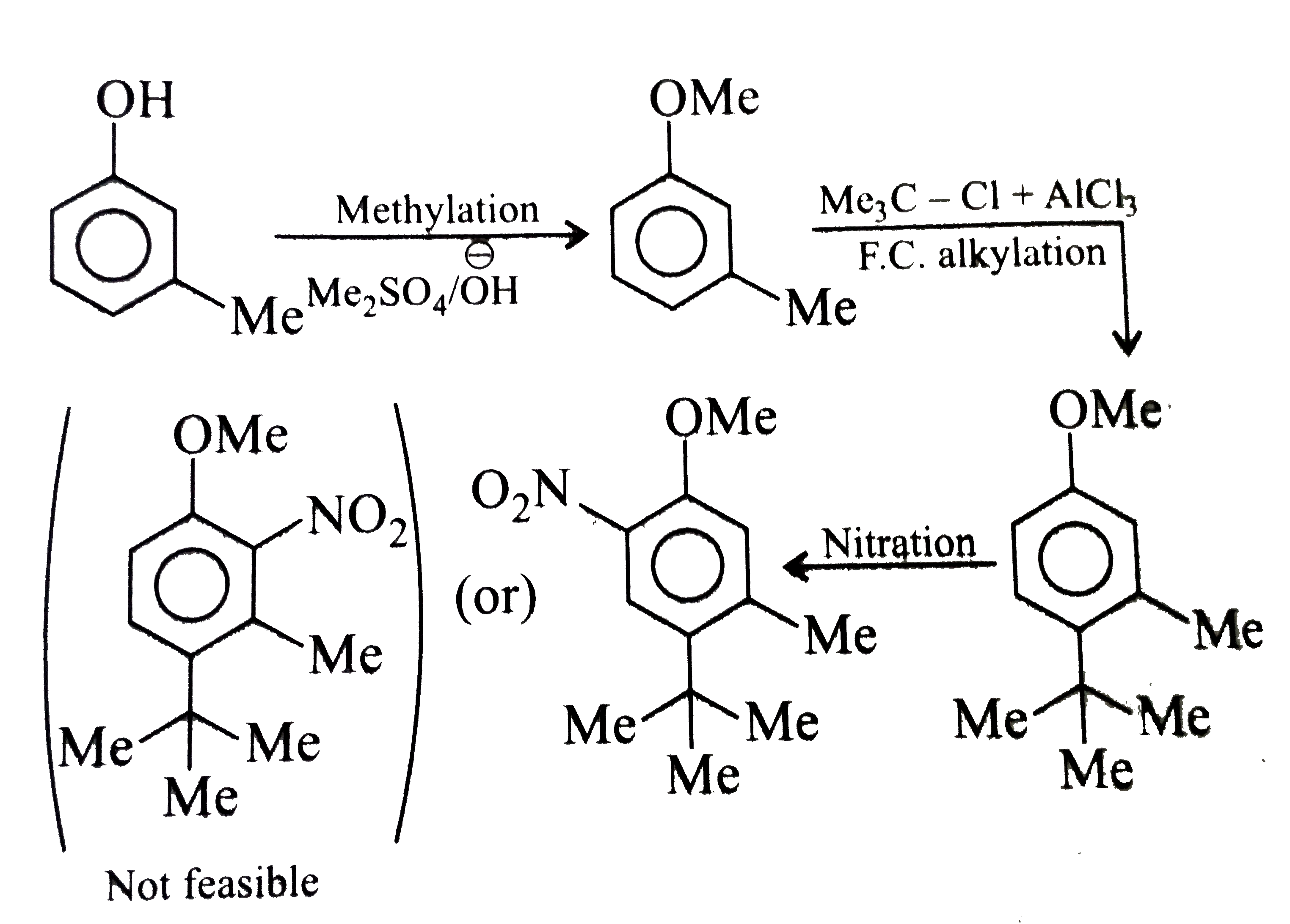 e. 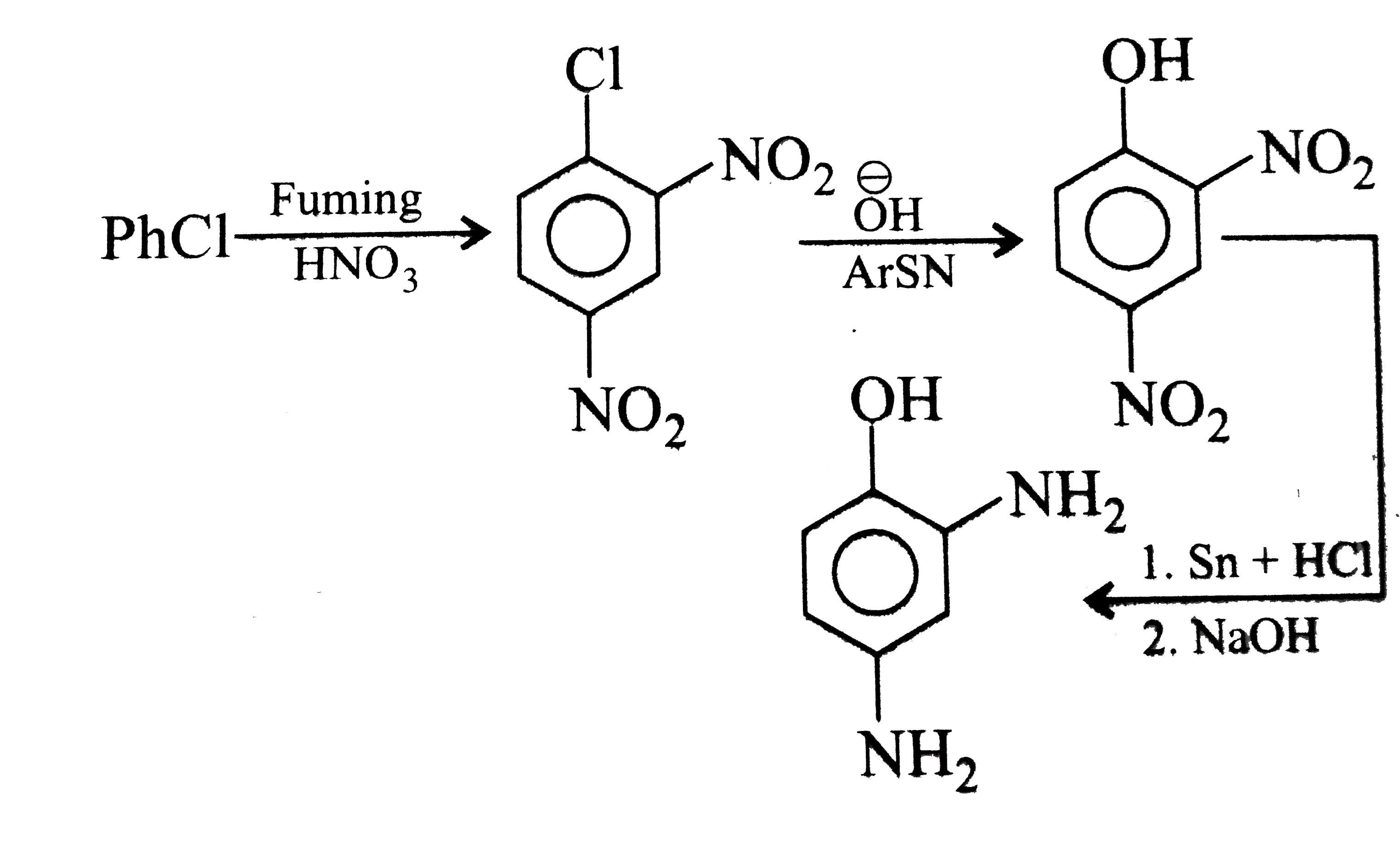 f. 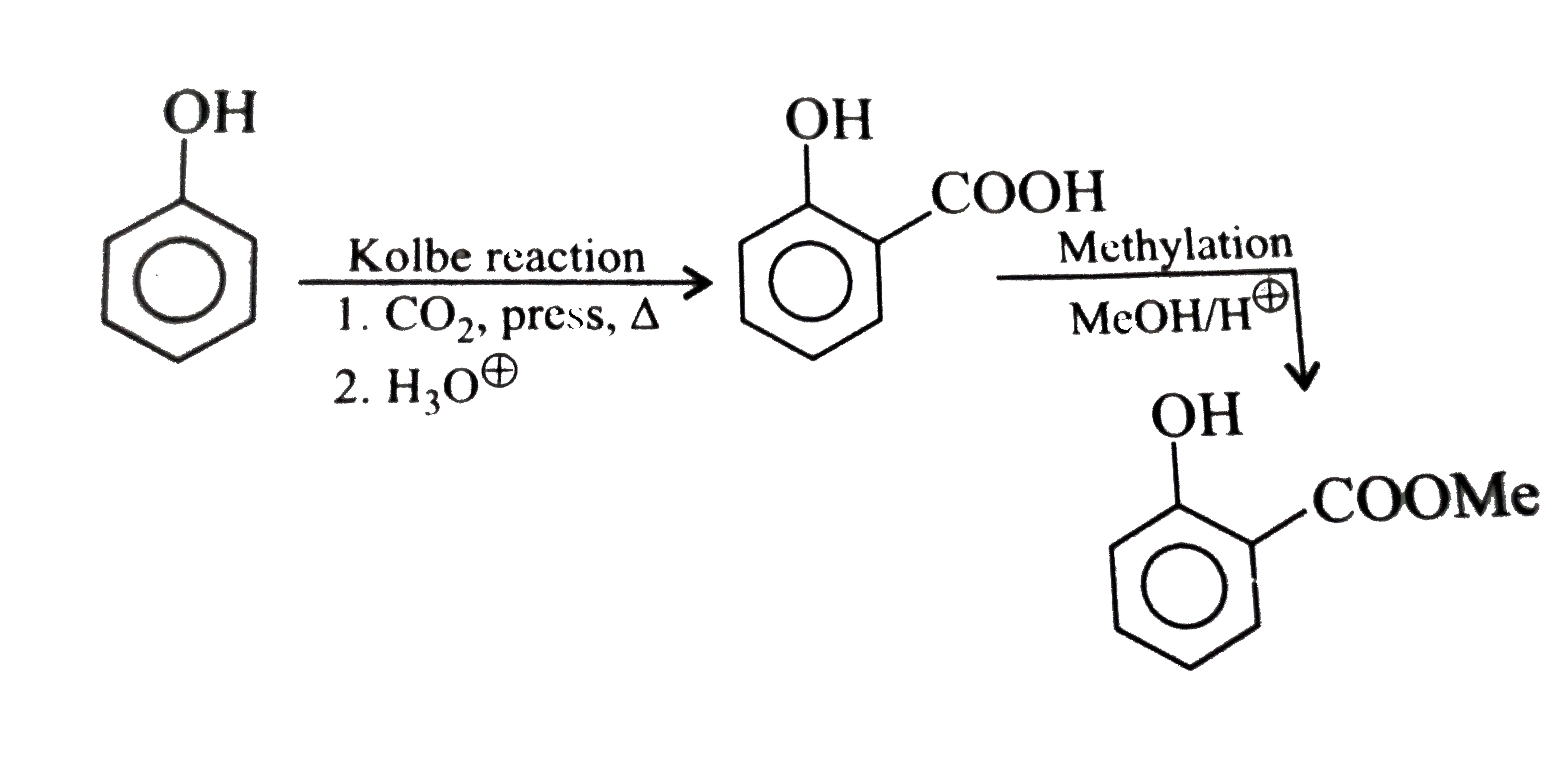 g. 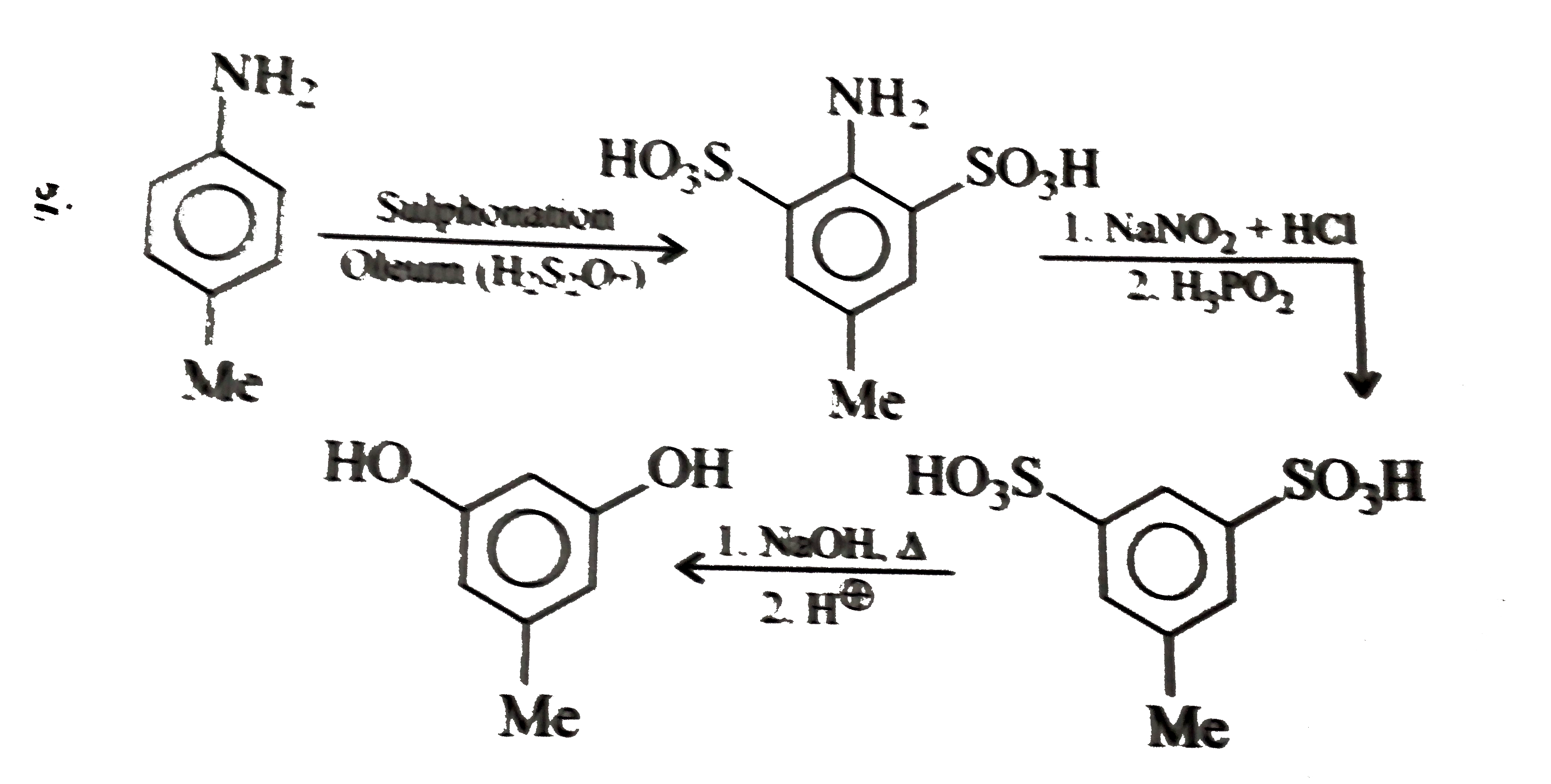 H. 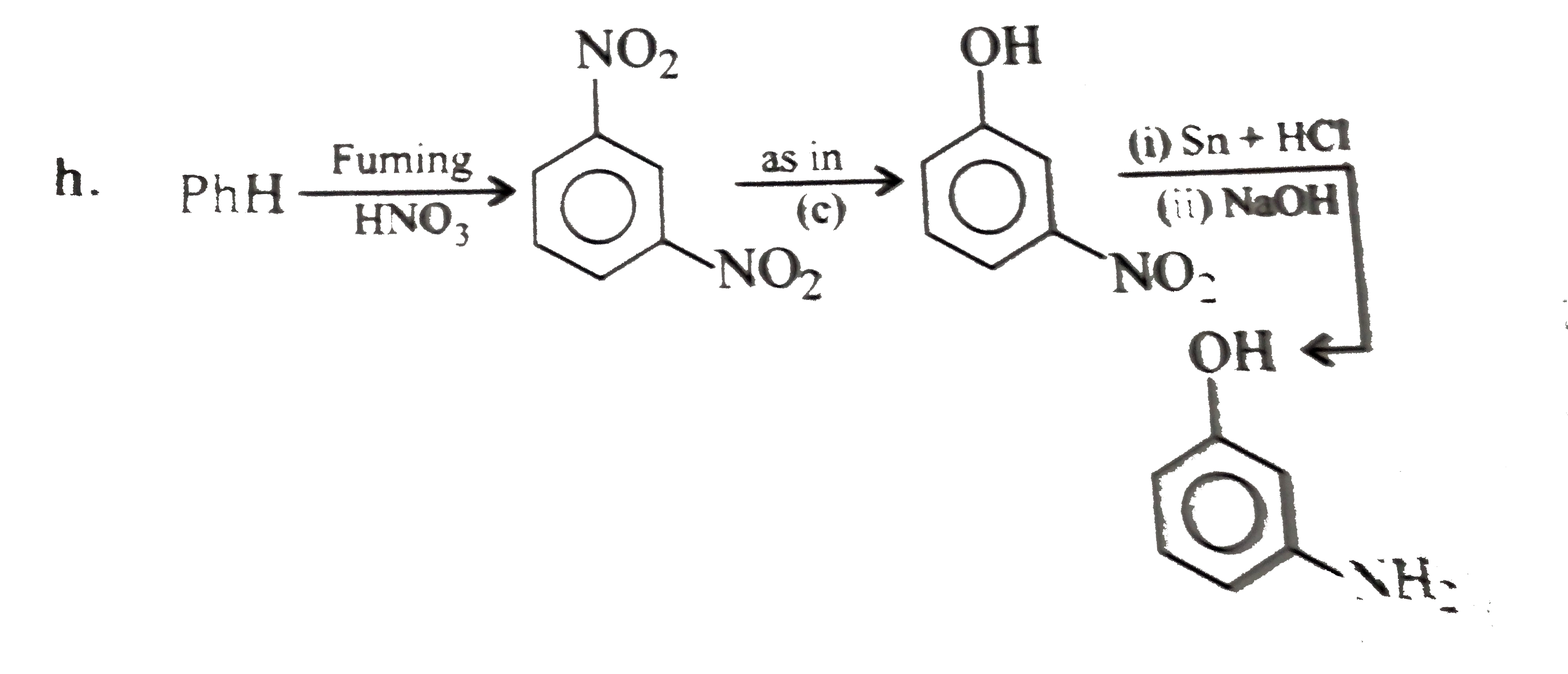
|
|
| 17. |
Synthesise the following : a.Butene to butanol and butan-2-ol 1-Chloro propane to pentanol and pentan-2-ol c.Benzene to 3-(4-chorophyl propan-1-ol) e. Ethyne to butanol Propane to allyl alcohol f. Propane to allyl acohol g. Propane to propanol and propan-2-ol |
Answer» Solution :a.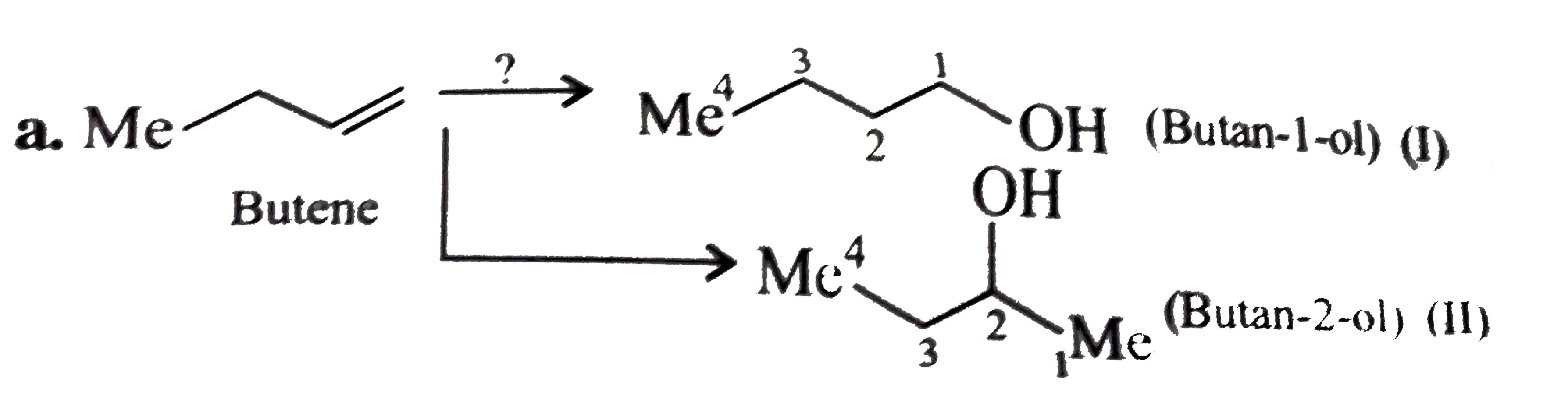 Hydrobration oxidation PROCEEDS with anti-Markovnikov addition, so it WOULD give (I), while acide-cataylsed hydration and mercuration-demercuration reaction proceed with Merkovnikov addition, so it would give (II). Synthesis: i. 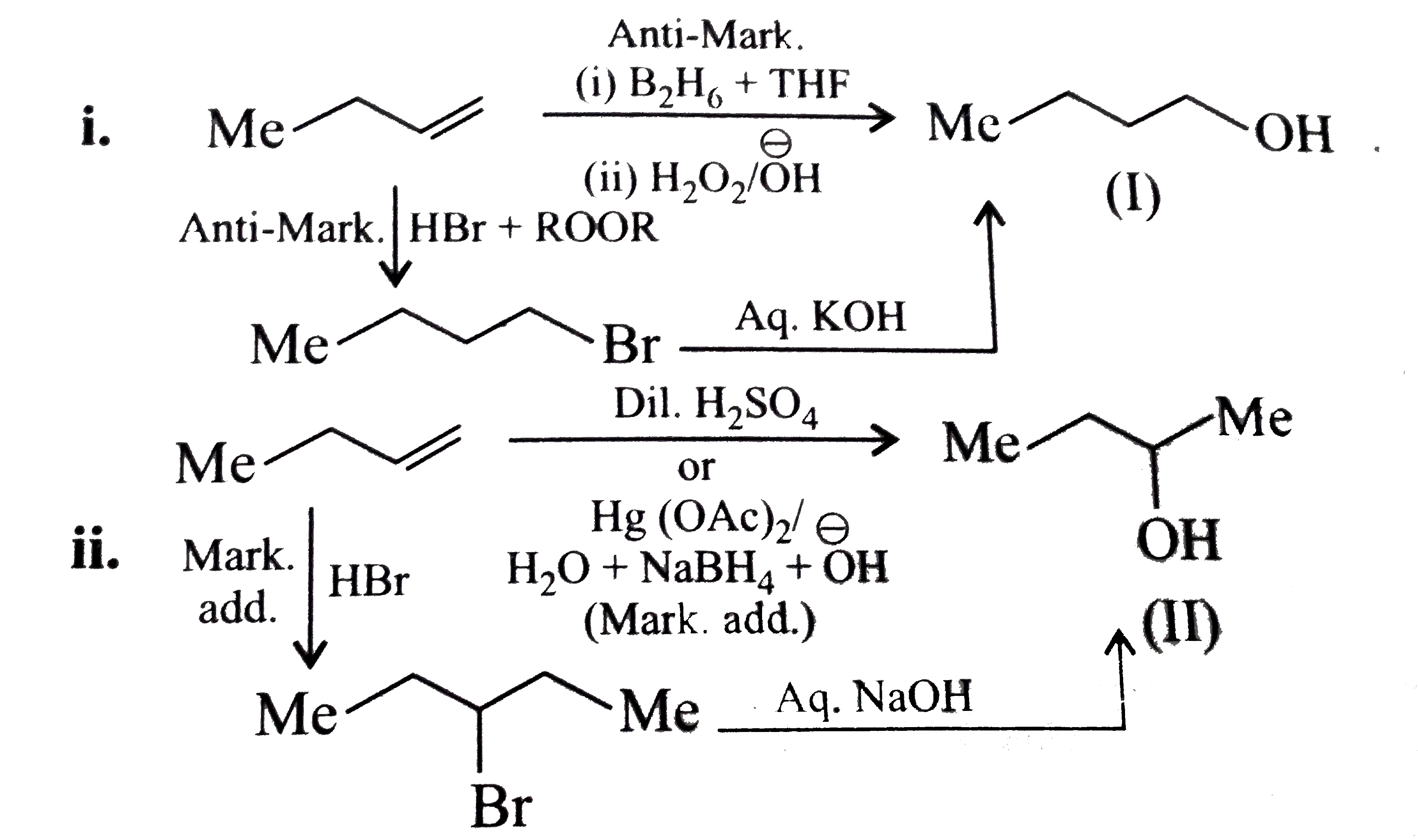 ii. 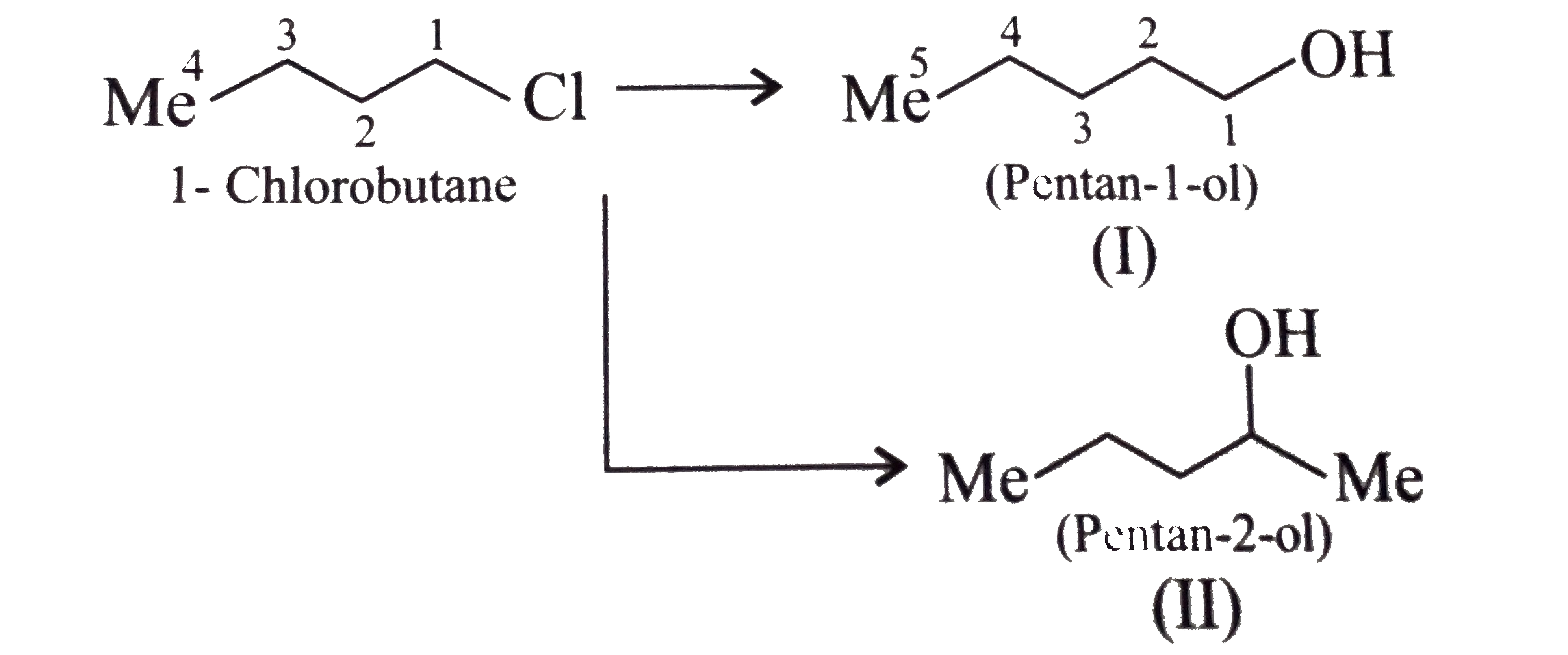 b. 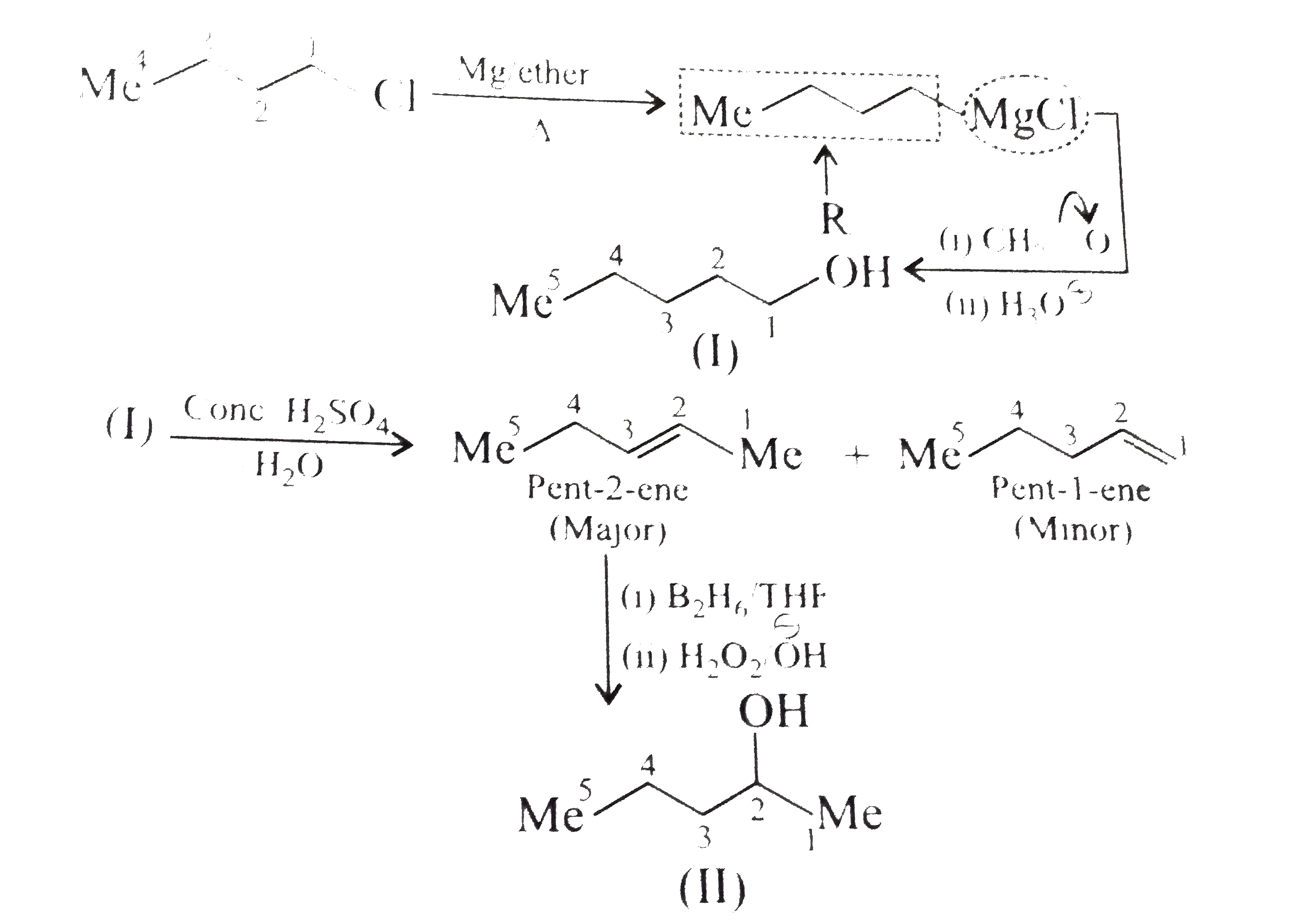 The 4C-atom chain has to be increased to 5C-chain by a GR. with `CH_(2)=O`. 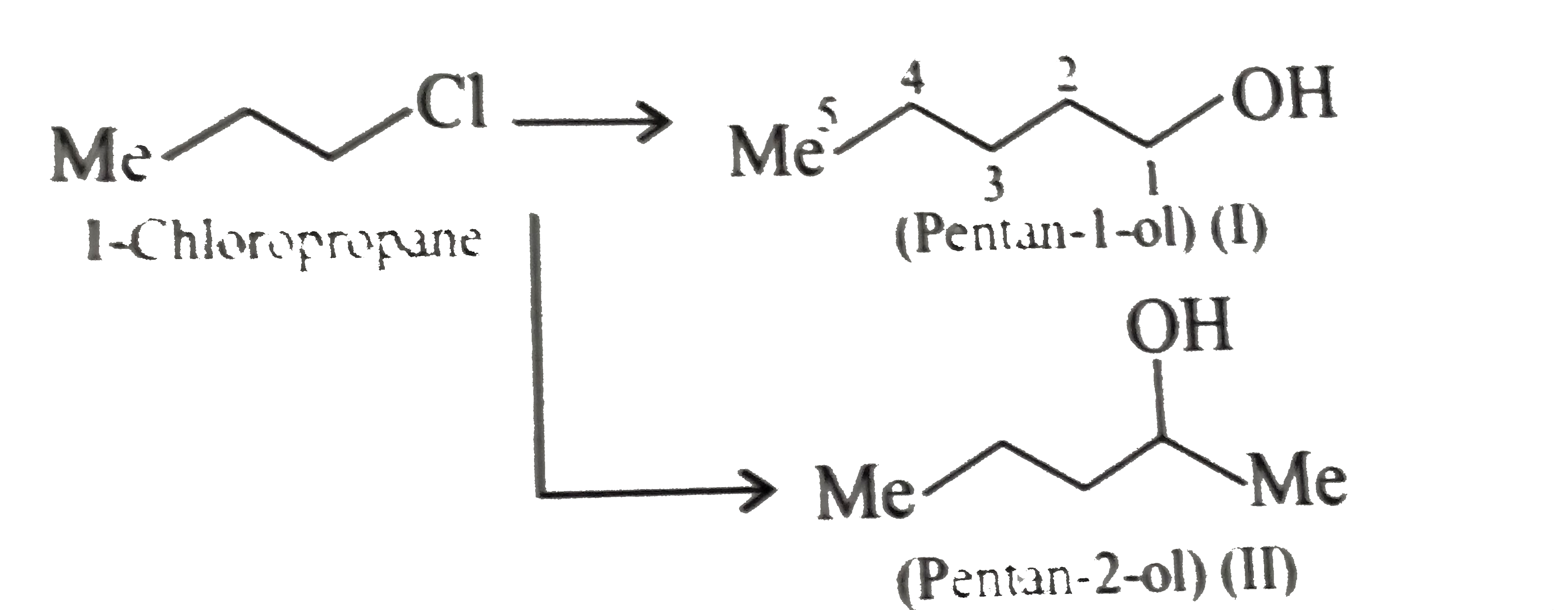 (I) 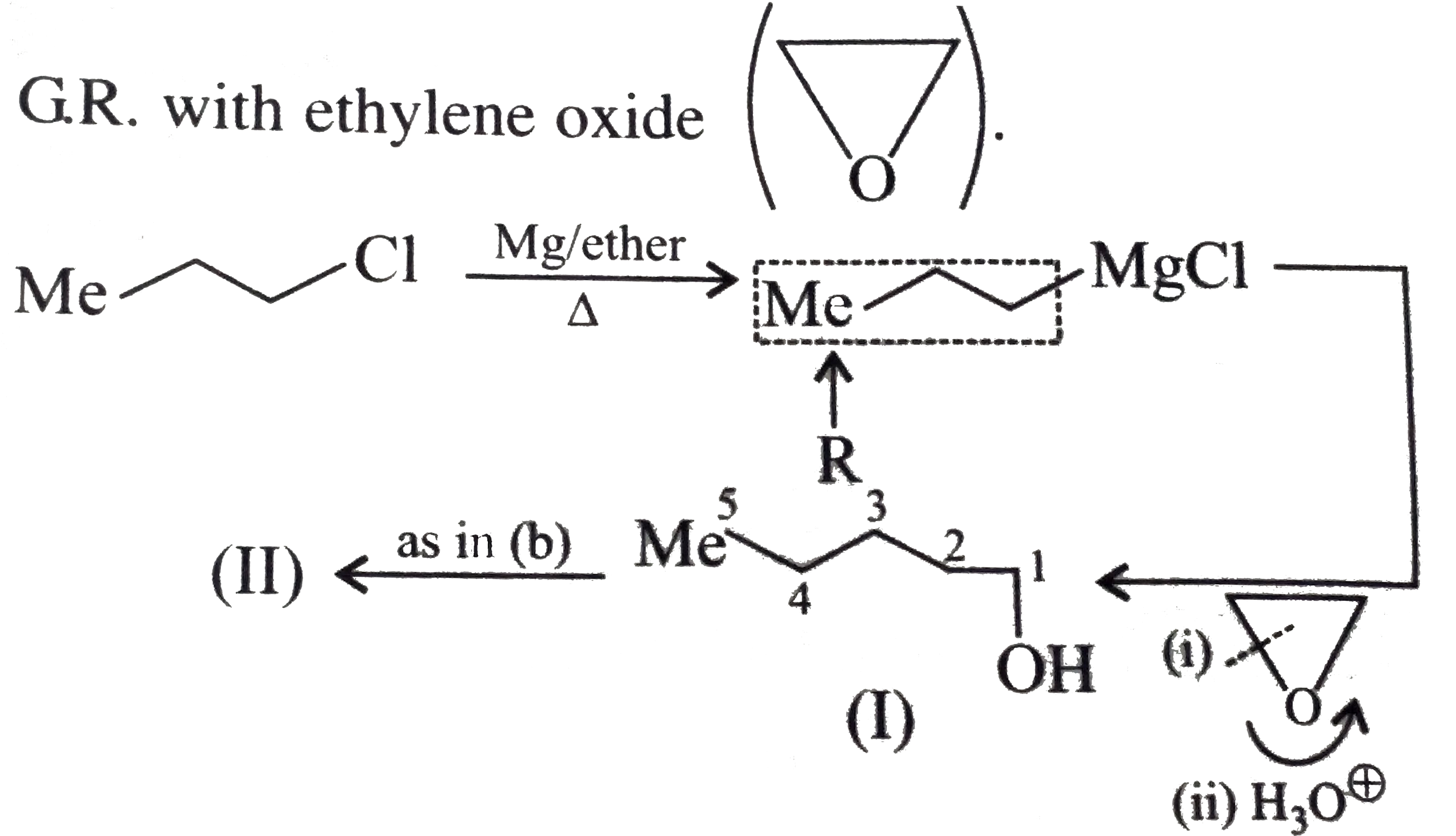 c. 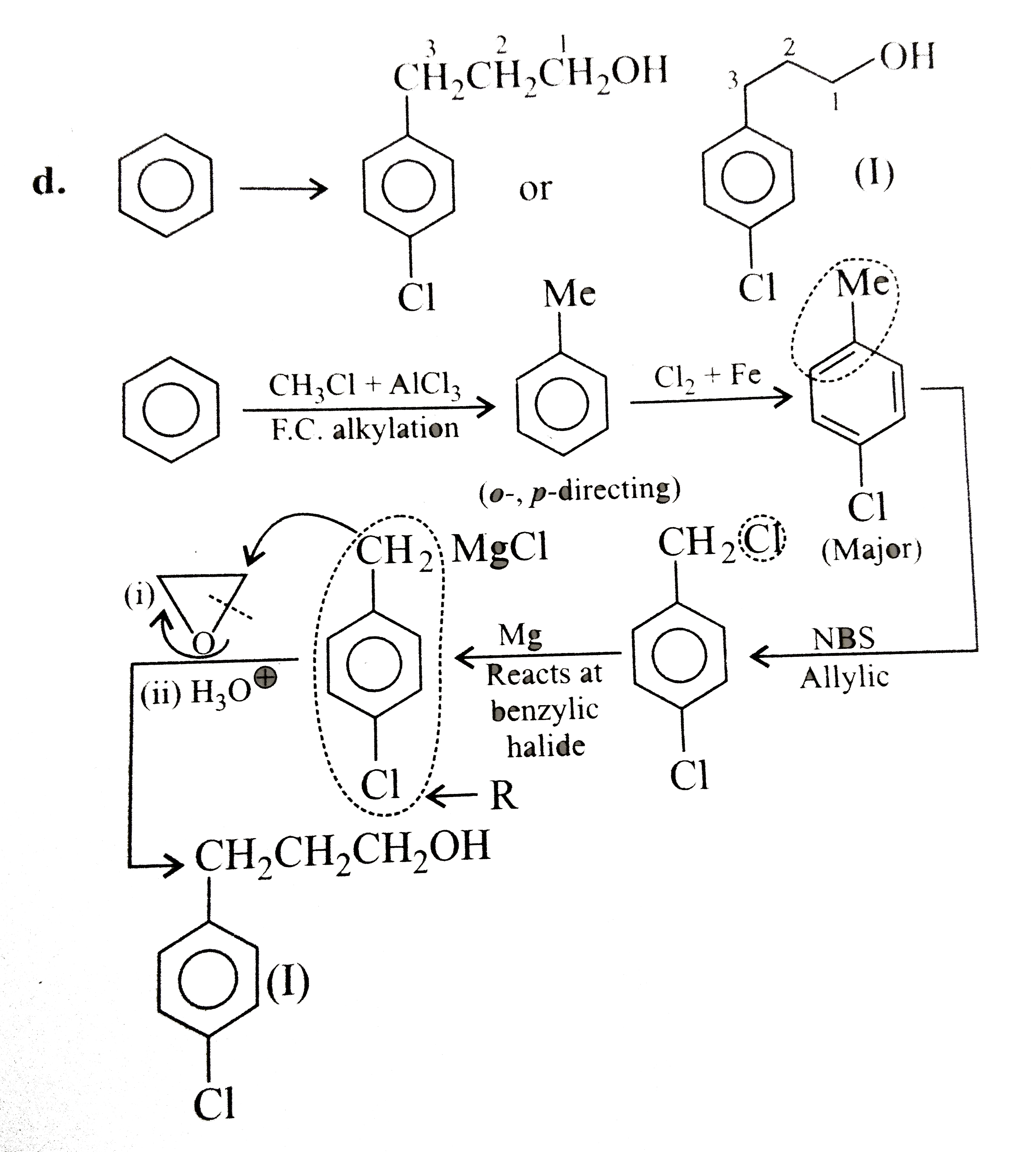 The 3C-atom chain has to be increased to 5C-chain by a 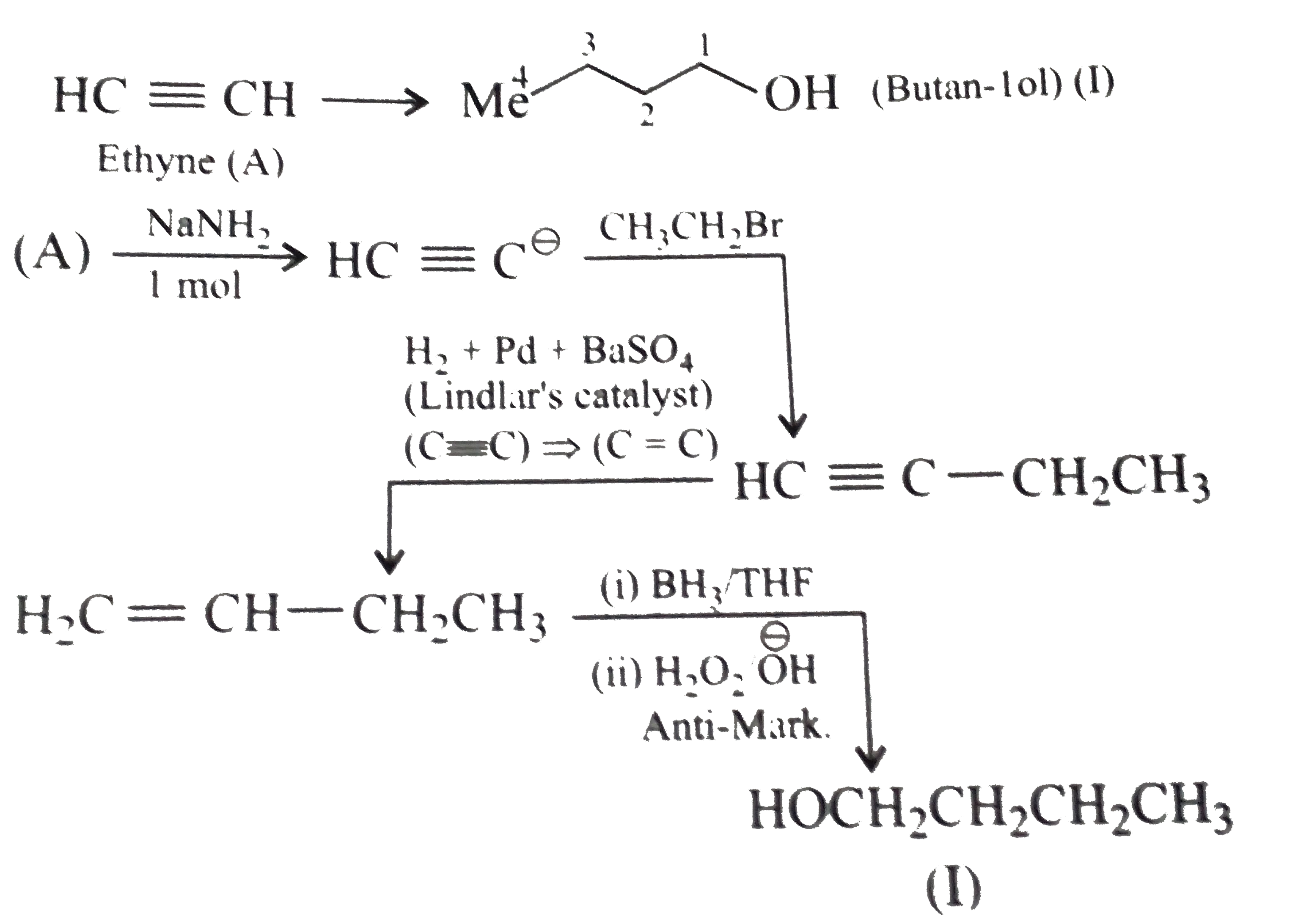 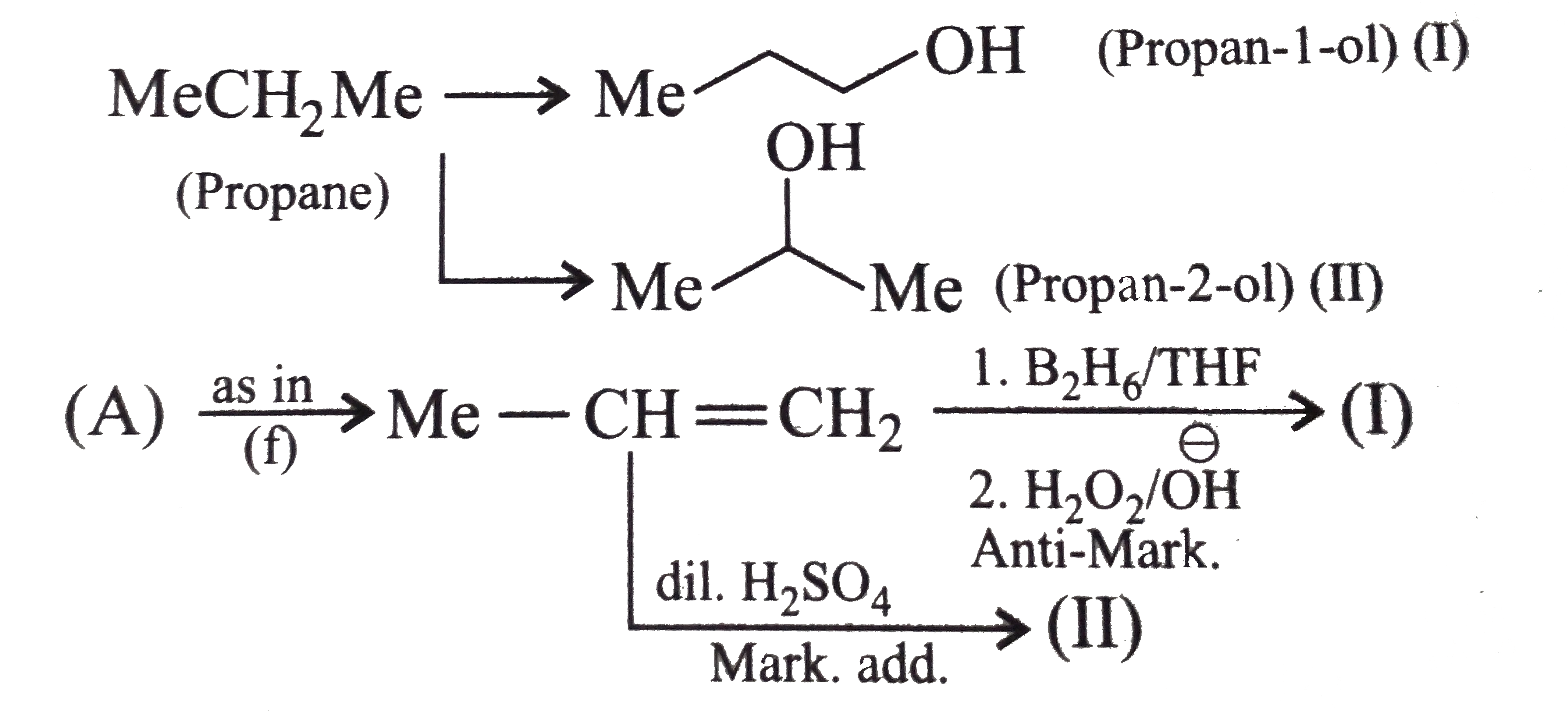 E. 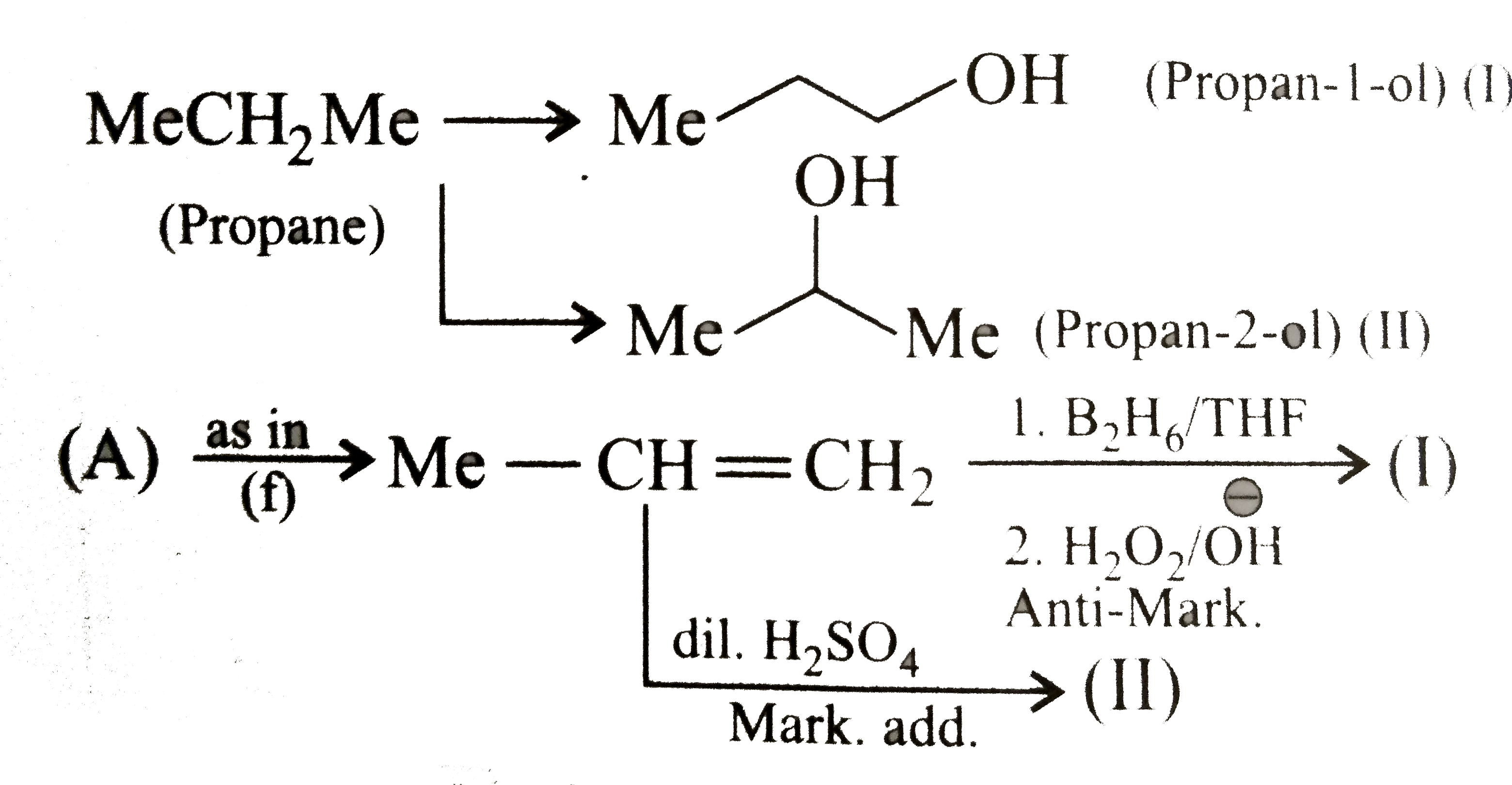 F.`overset (MeCH_(2)Me) underset (Propane (A))``rarr H_(2)C` == `overset (CH--CH_(2) OH) underset (Allyl aclohol (I))` |
|
| 18. |
Synthesis of propellane takes placed by the following route. Q. The product (w) is: |
|
Answer»
(B) `CU(a)+Cu^(2+)TOCU^(2+)+Cu(c)` (c). `MgCl_(2)+2H_(2)OtoMg(OH)_(2)+H_(2)+Cl_(2)` partically `O_(2)` will (d). `MgCl_(2)toMg+Cl_(2)` |
|
| 19. |
Synthesis of identical copies of DNA is called |
|
Answer» TRANSCRIPTION |
|
| 20. |
Synthesis of ester involves the reaction of alcohol with |
| Answer» Answer :D | |
| 21. |
Synthesis of each molecule of glucose in photosynthesis involves : |
|
Answer» 18 molecules of ATP |
|
| 22. |
Synthesis ofcyclohexane-1,2diolfromcyclohexenemay beaccomplished in twoways : I . MnO_(4) dilute OH, O^(@)C dihydroxylation II . Peracid epoxidationfollowedby NaOH openingof theepoxidering . Whichof thefollowingstatementabout theproductsfromthesereactionsis correct? |
|
Answer» MethodsI and IIgivesameproduct |
|
| 23. |
Synthesies the following ethers, selec-ting the given methods. (I) Williamson's synthesis (II) Alkoxy mercuration-demercuration (III) Intermolecular dehydration Given reasons for the selection of the method. a.Divinyl ether b.Dicyclohoexyl ether c.Propyl sec-butyl ether d.1-Propoxy-2-methyl propane e.1-(3-Mrthyl butoxy)-3-methyl butane |
Answer» Solution :a.Write the structures of ether: 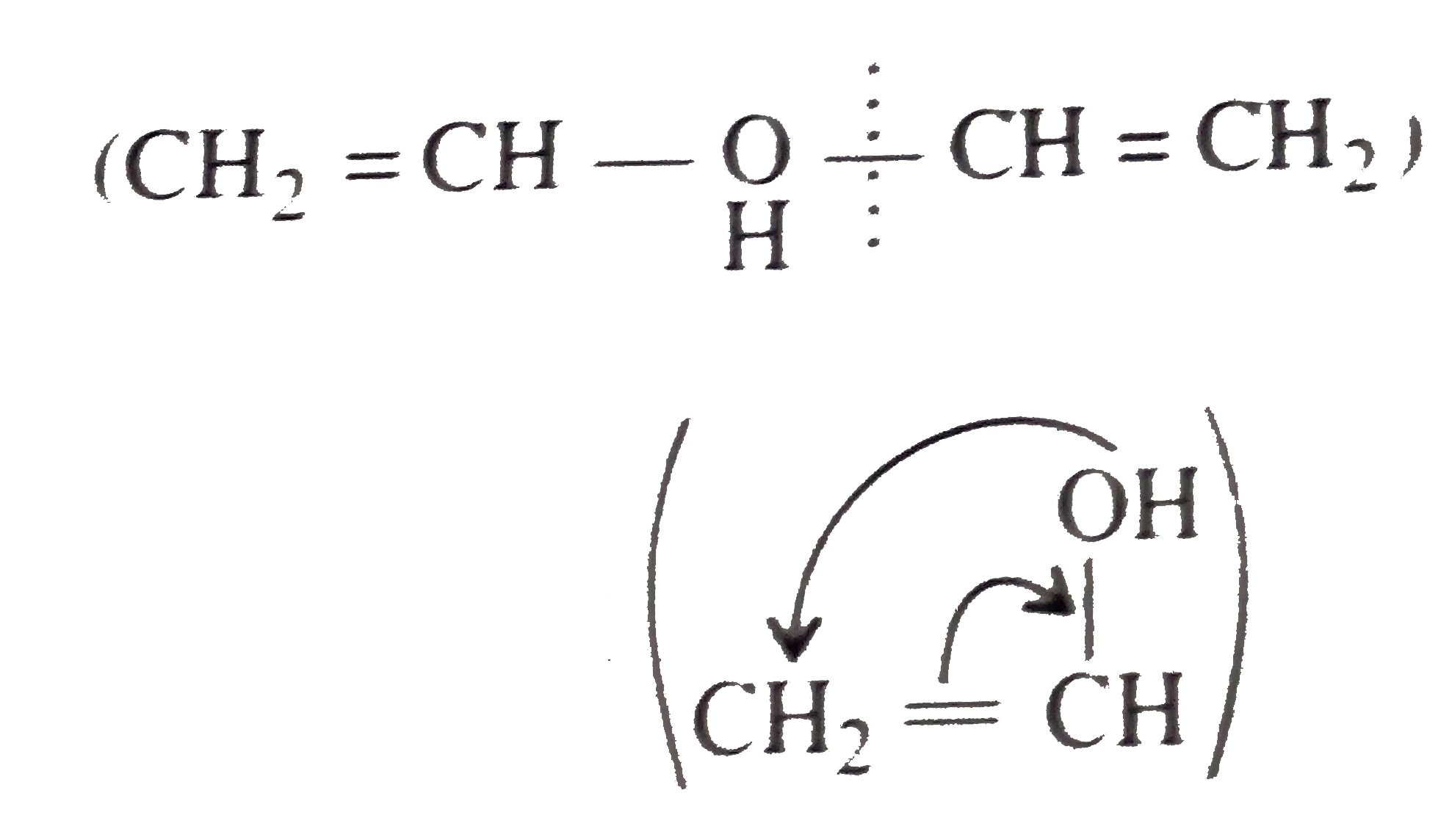 since it is an unstable compopund and tautomerise to `H_(3)C--CH=O`. Double BOND is introduced after the formation of ether by method (III). 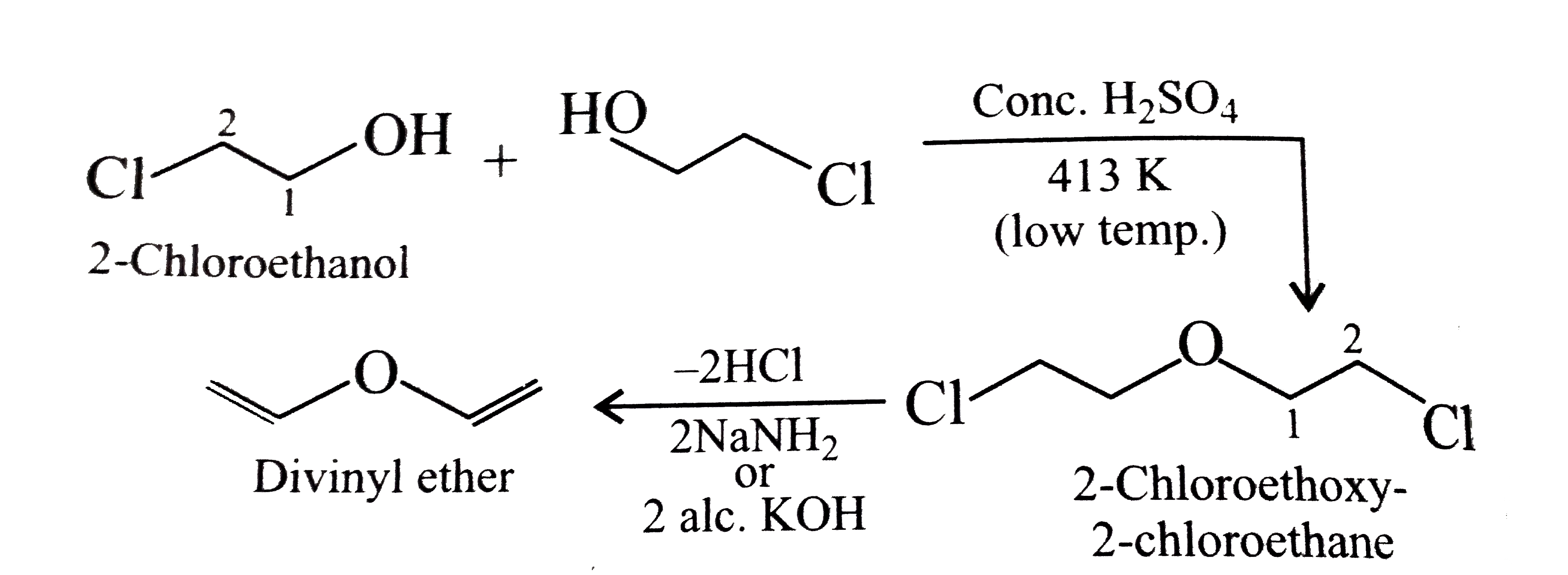 Method (I) cannot be used because of unreactive vinyl halide 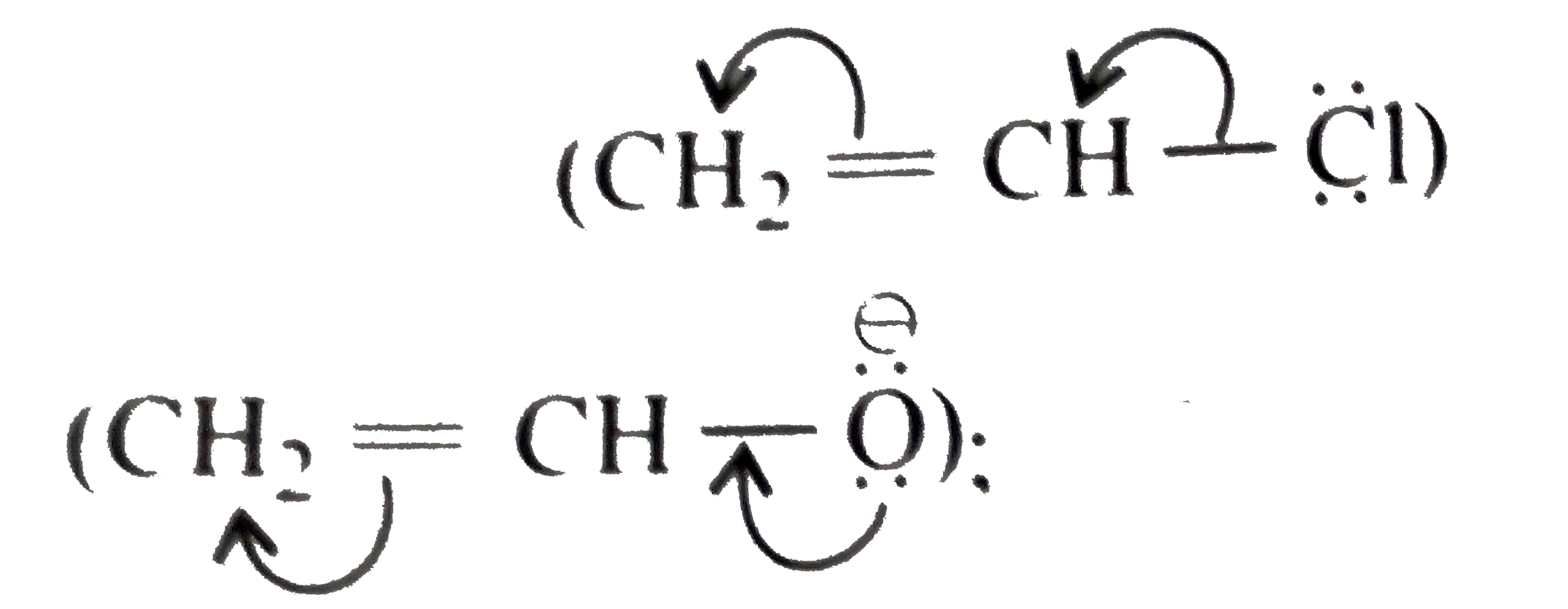 and vinyl oxide and vinyl oxide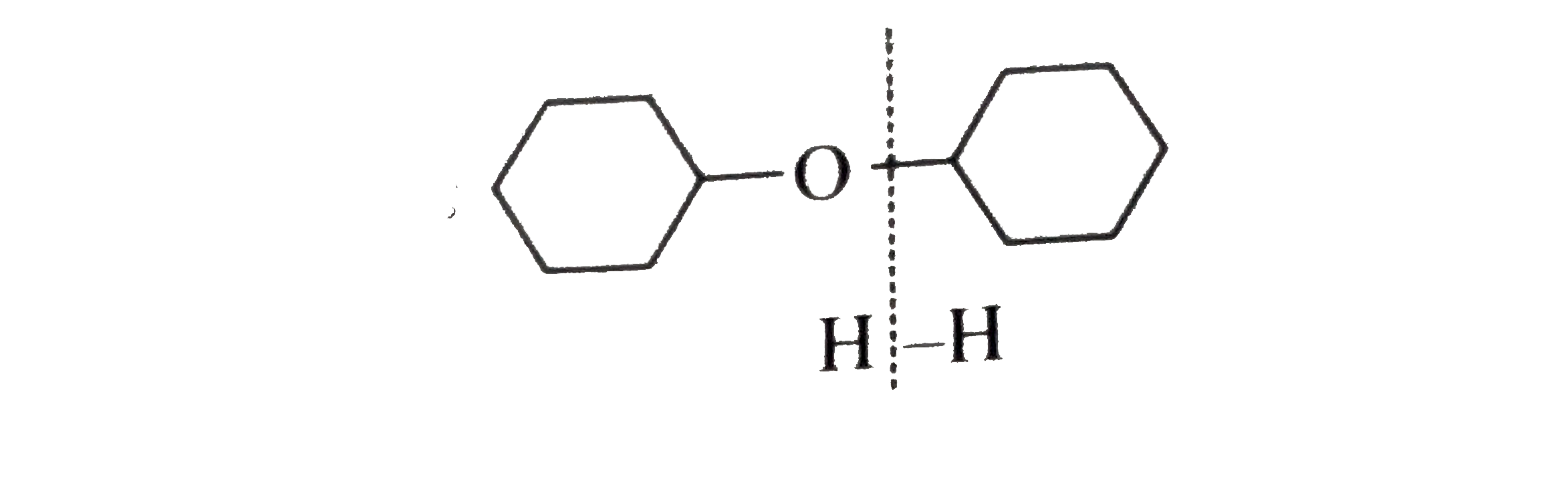 both are stabilised by reasonance.Method (II) cannot be used because of unreactive vinyl alcohol. both are stabilised by reasonance.Method (II) cannot be used because of unreactive vinyl alcohol.b.Structure of ether : 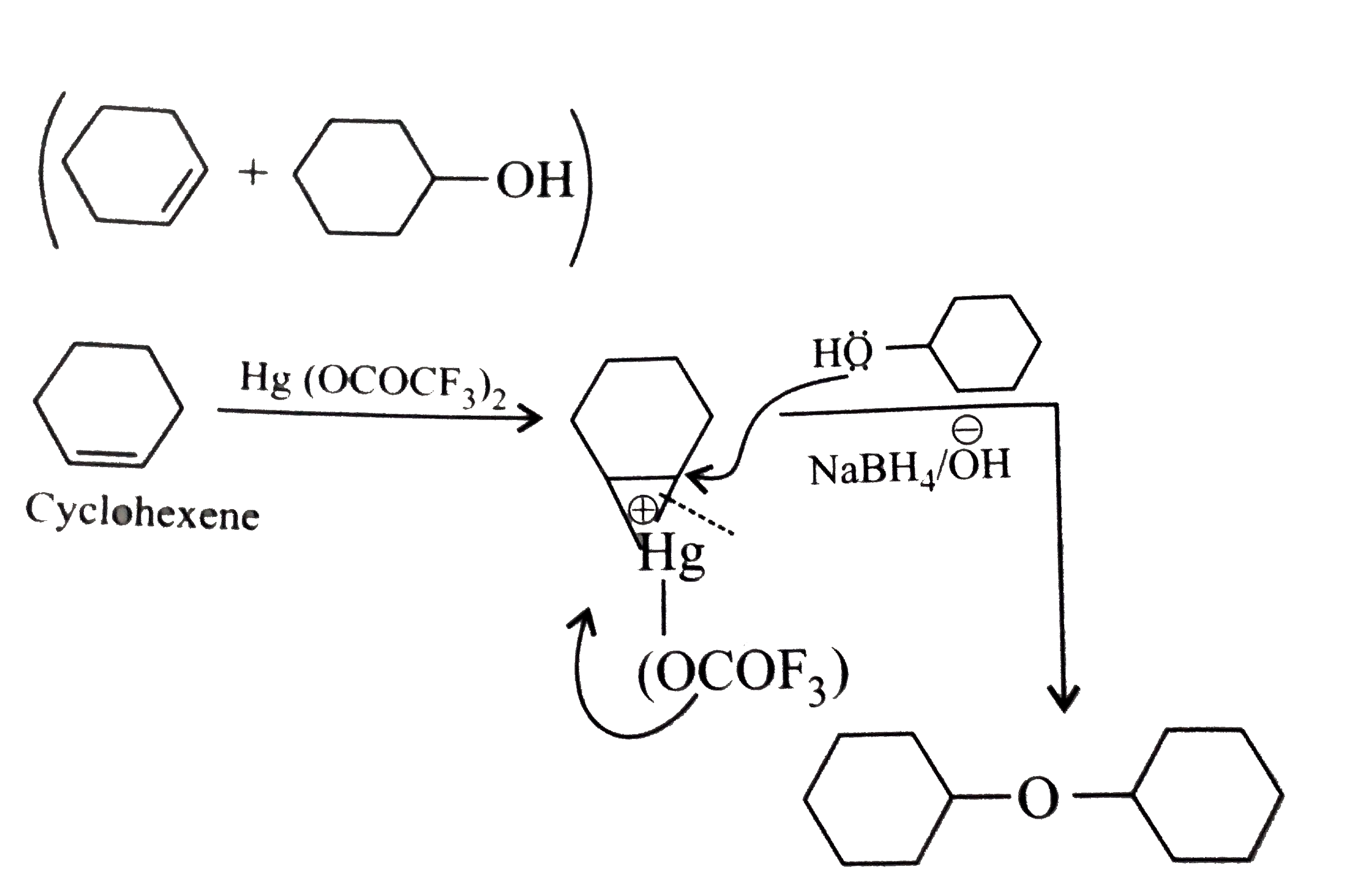 Method (II) will give desired result with 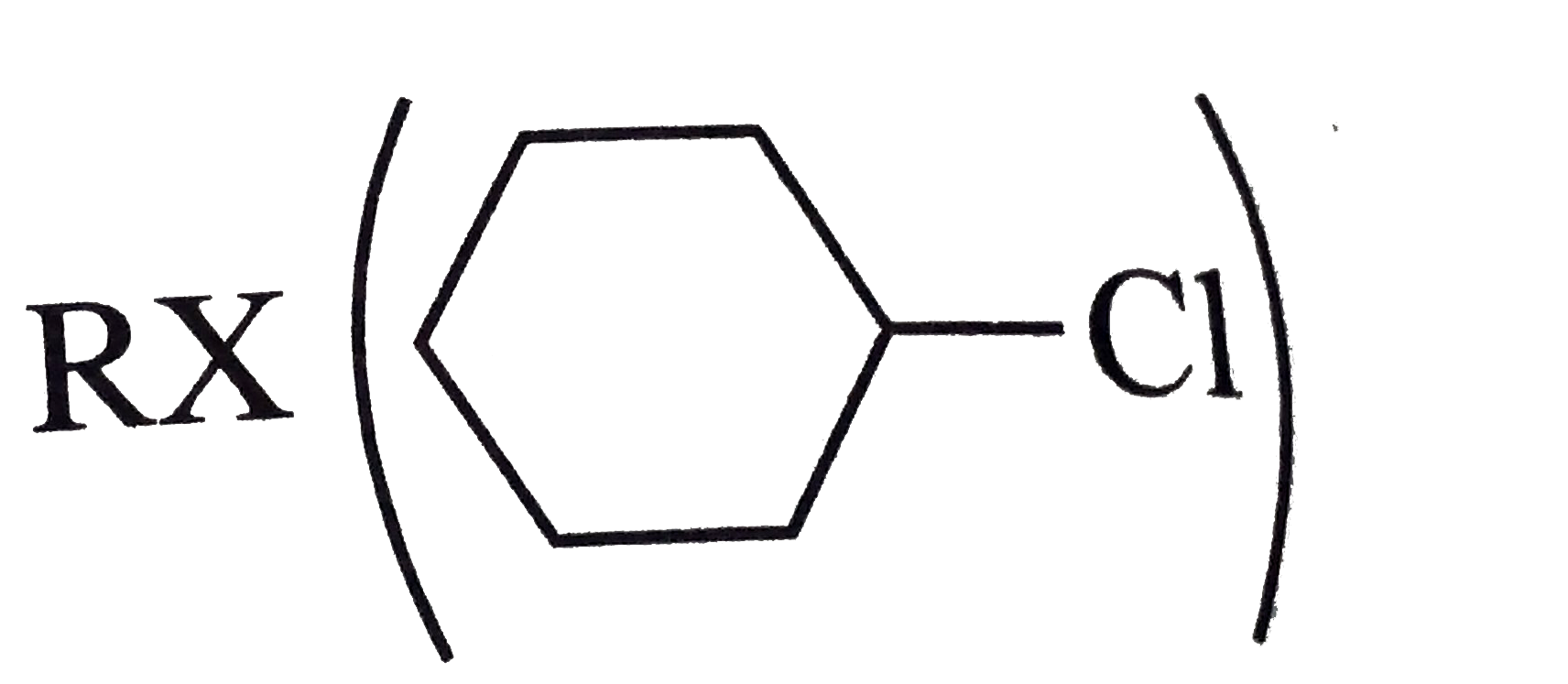 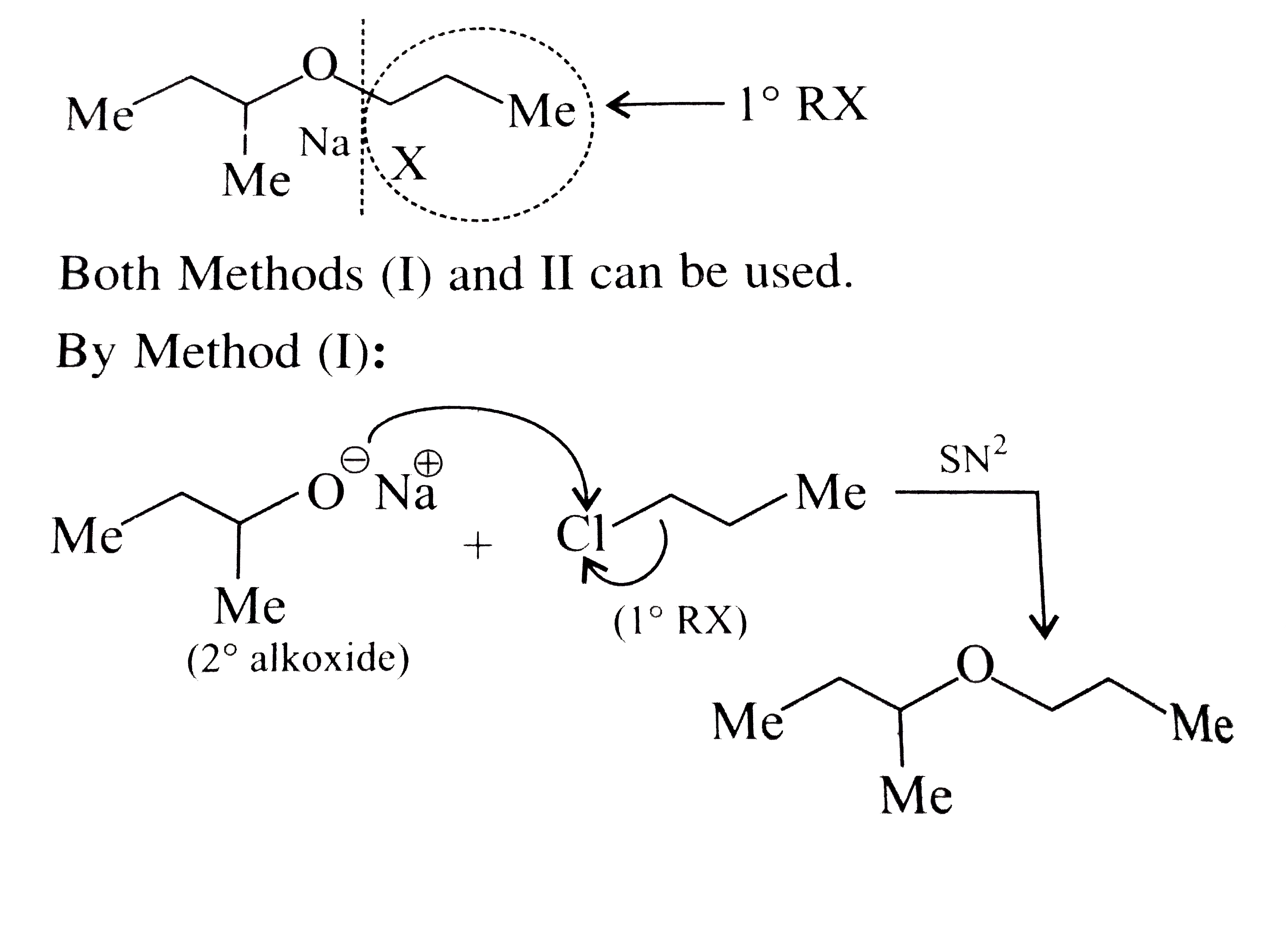 Although it is symmetrical ether, but method (III) cannot be used because intromlecular dehydration of `2^(@)` ROH would give mainly cyclohexene.Method (I) cannot be used because `2^(@)` RX 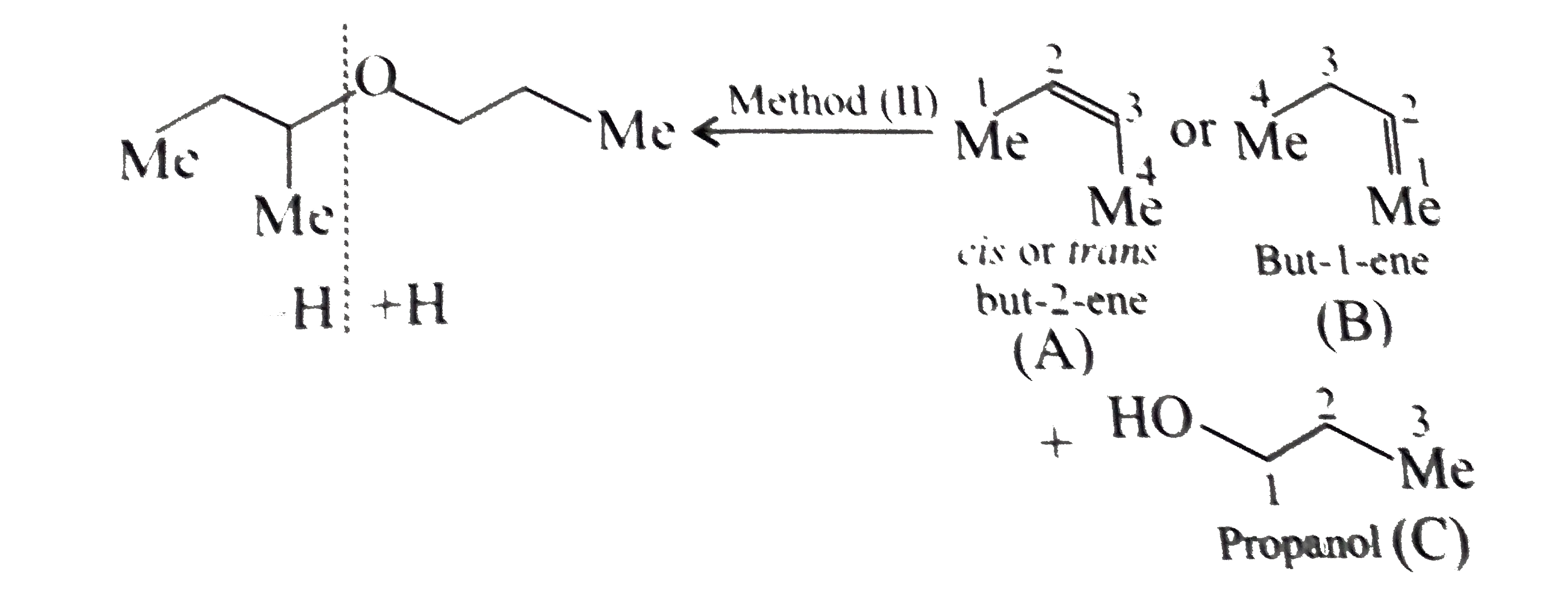 does not favour the reaction. does not favour the reaction. c.Structure of ether.  By method (II): i.  ii. 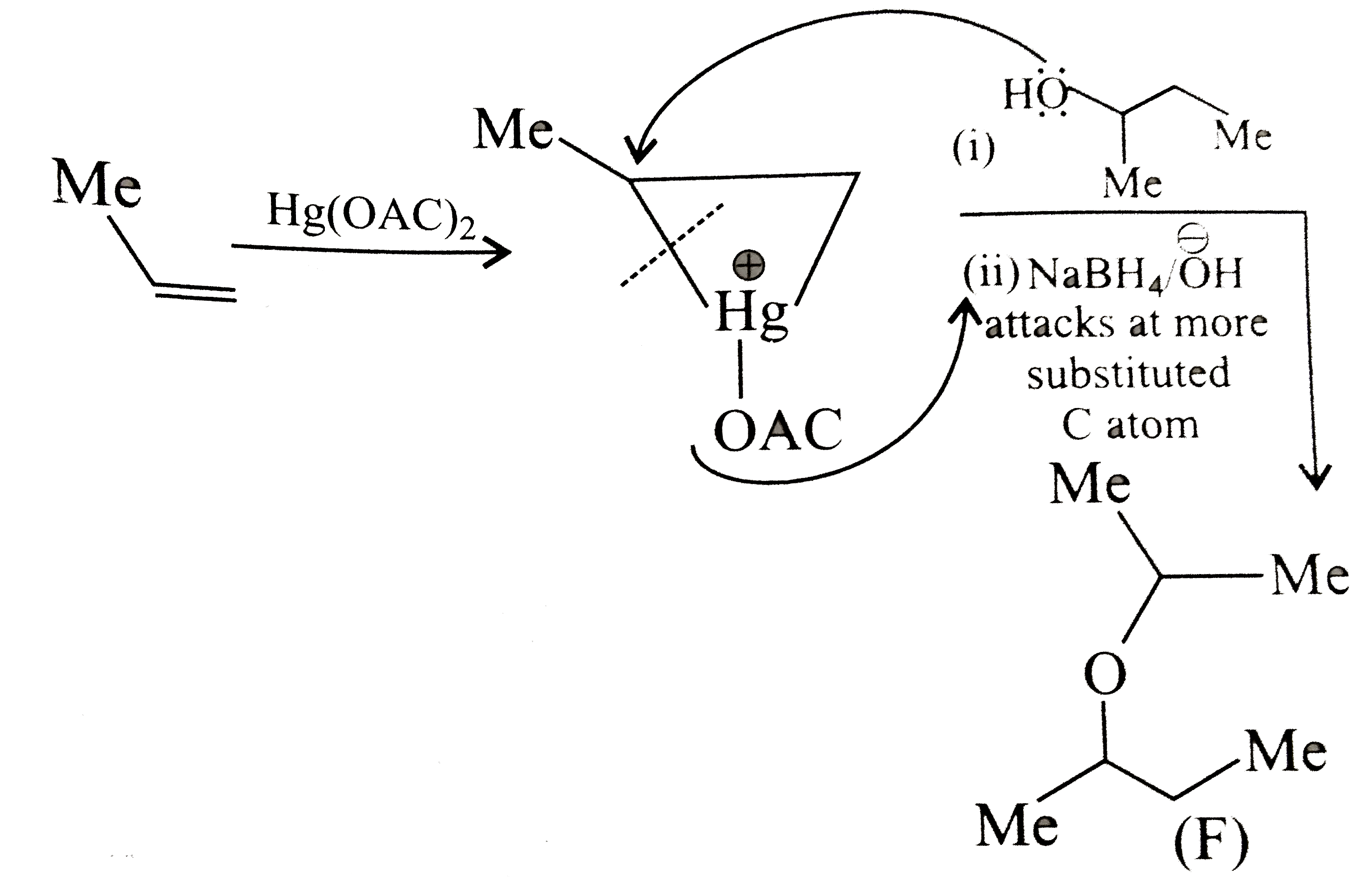 Path (ii) is not feasible because it would give different ether (isopropyl-sec-butyl ether) (F). 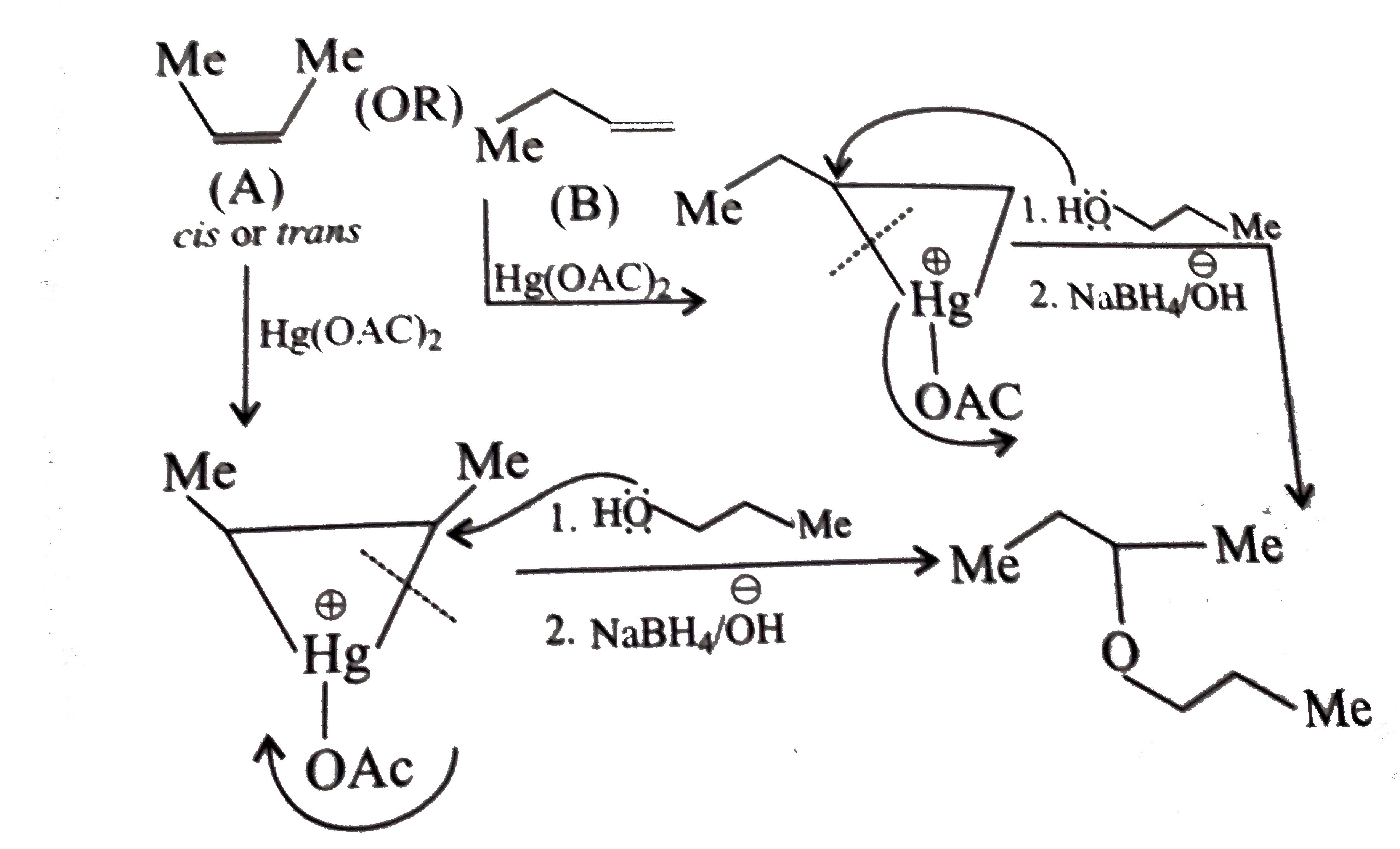 Path (i) would give the required ether: 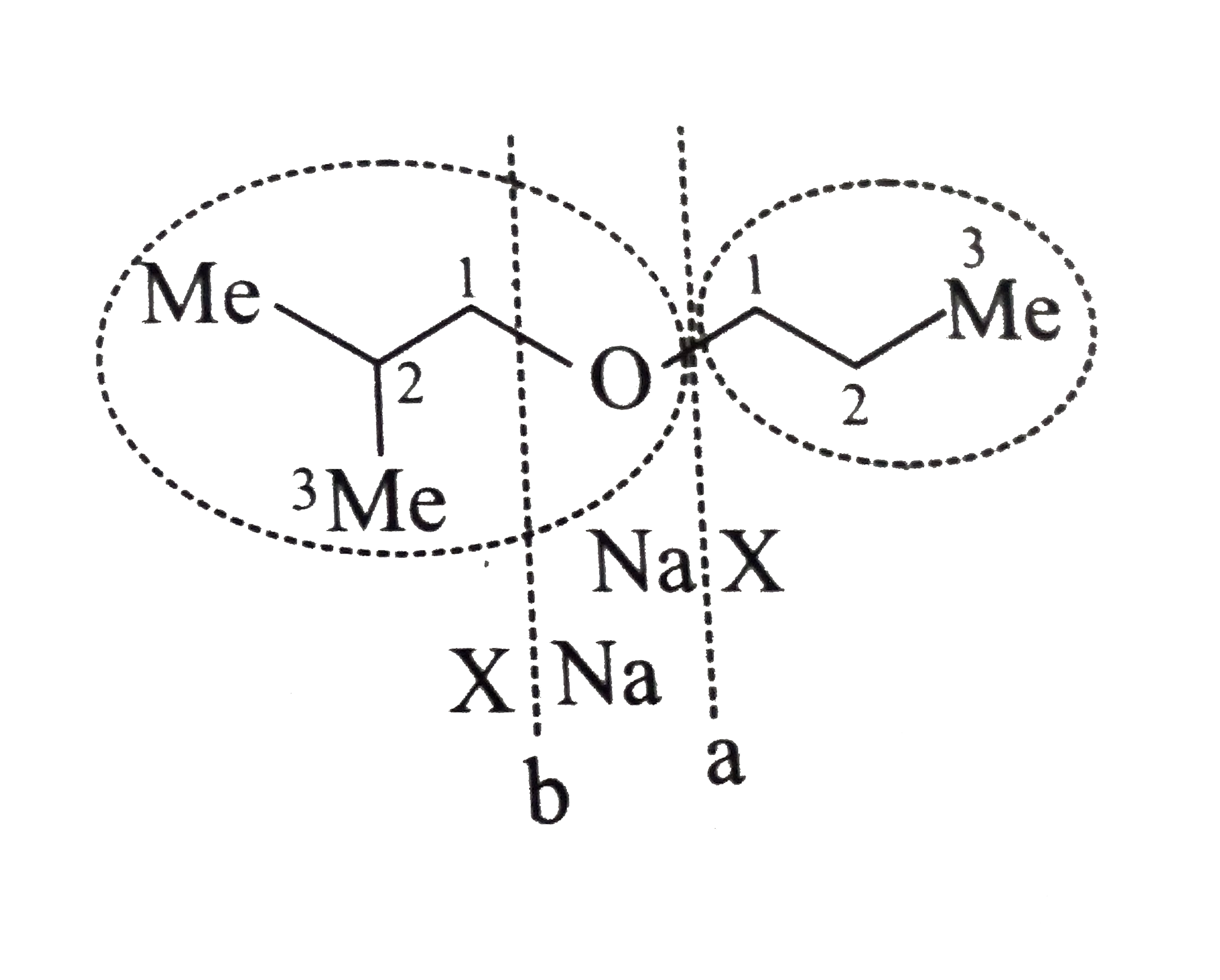 d.Structure of the ether: 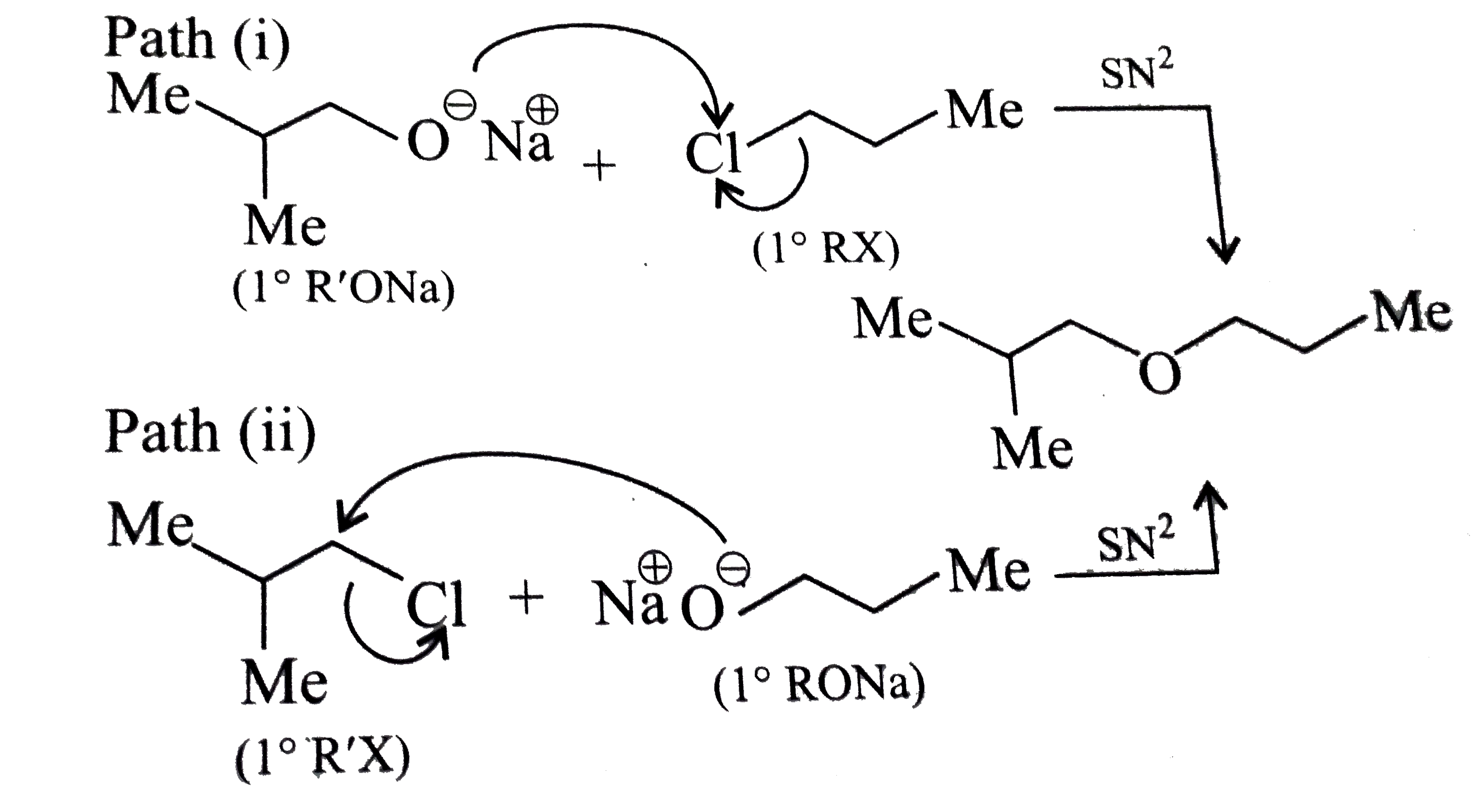 Method I is feasible: 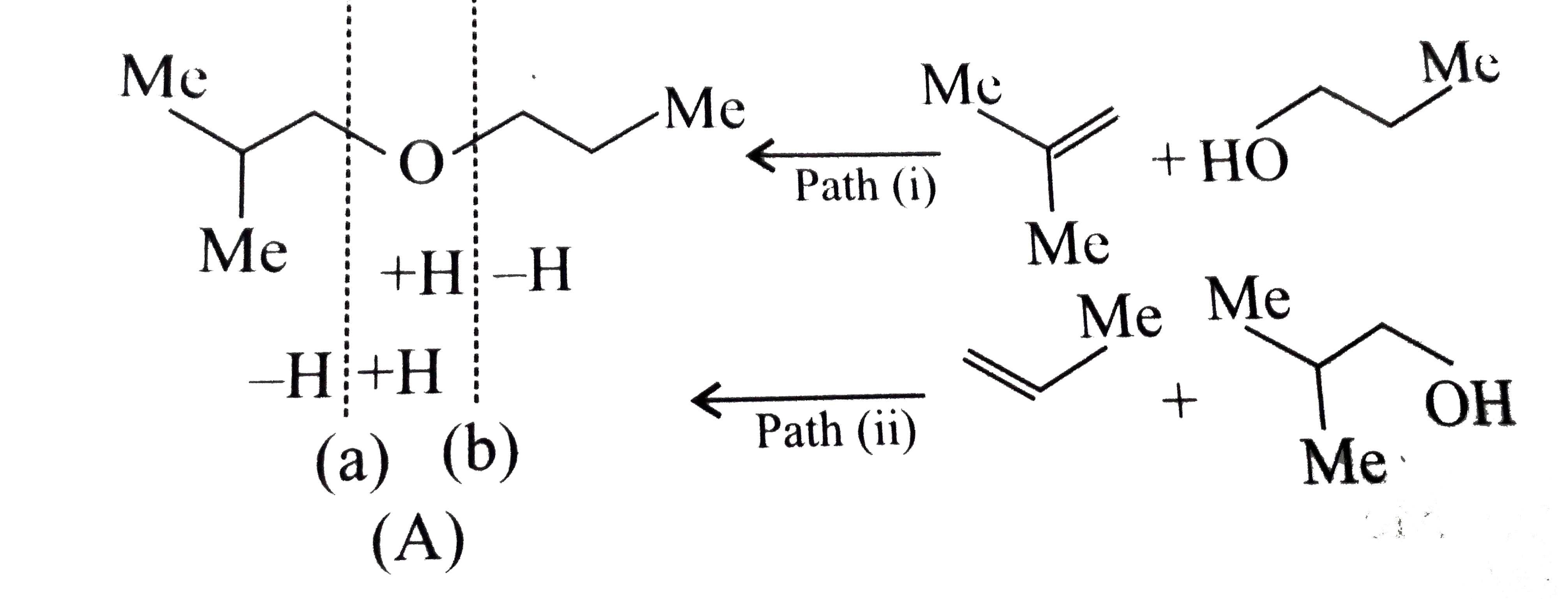 Methods II and III are not feasible and method II would give different isomers. 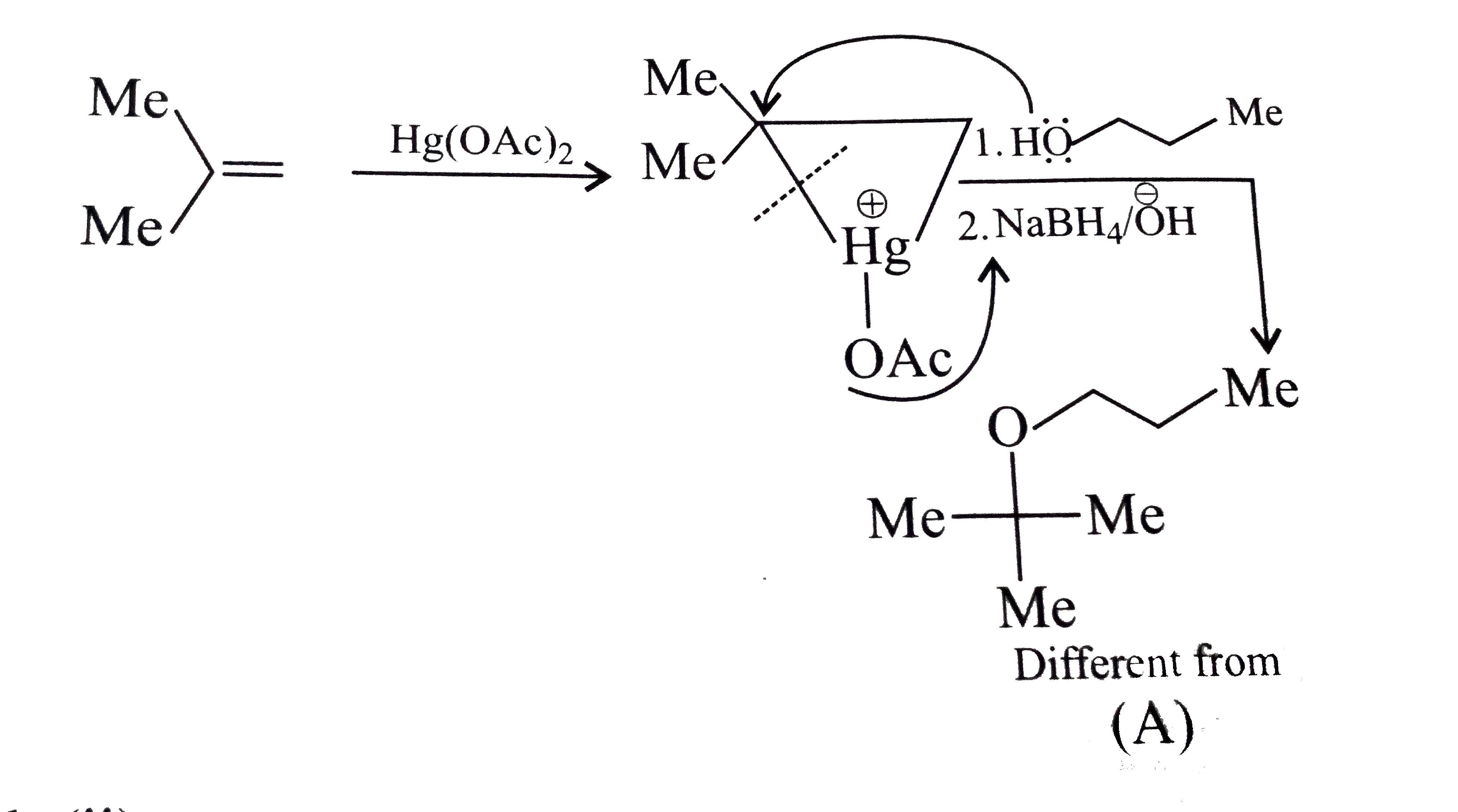 Path (i): 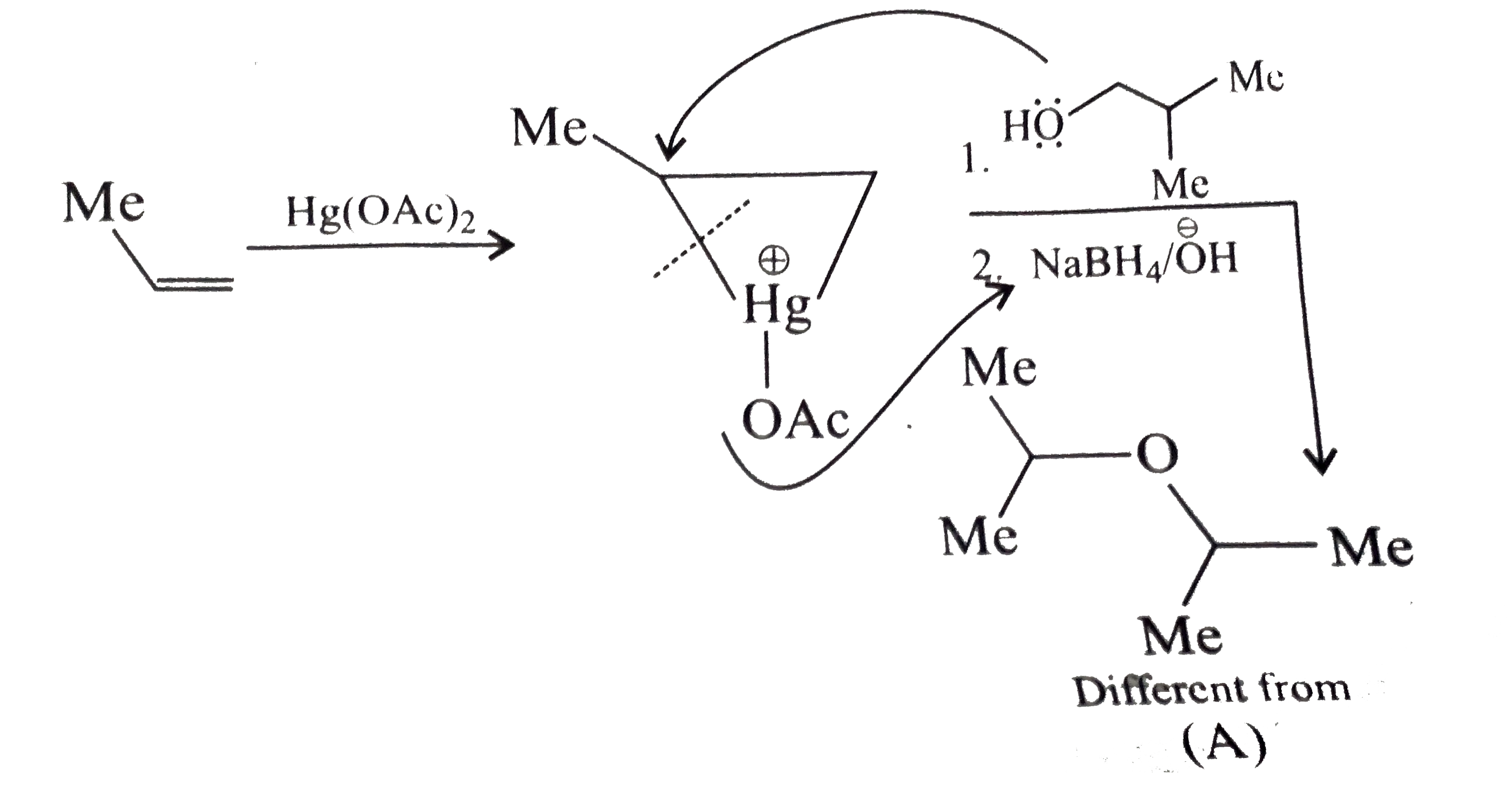 Path (ii): 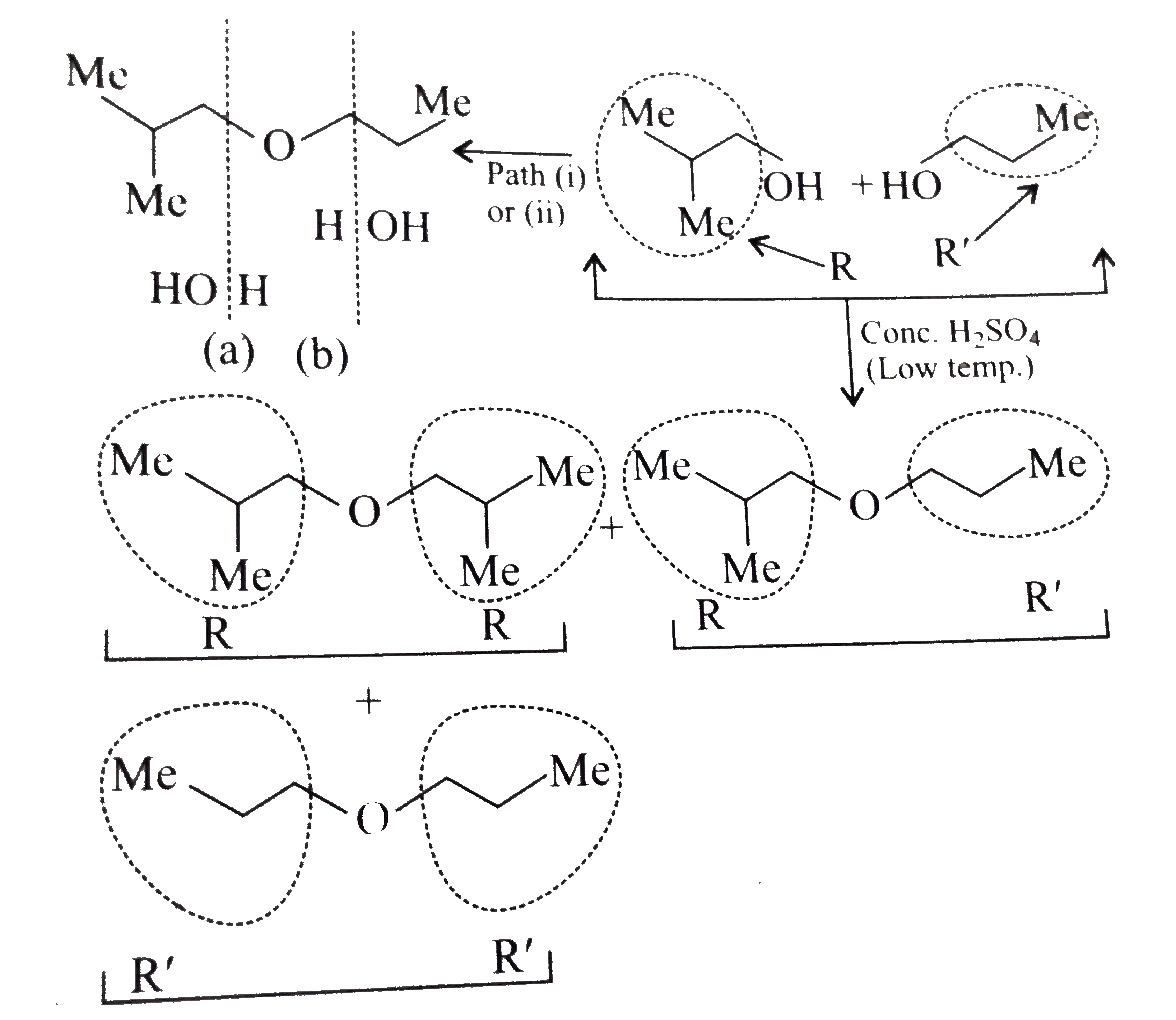 Method III is not feasible. It is a non-symmetrical ether, dehydration gives mixtures and both alkyl groups are `1^(@)` . `R--O--H + R'O---H OVERSET (Conc.H_(2)SO_(4)) underset (Low TEMP) rarr` `R--O--R' + R--O--R + R'--O--R` 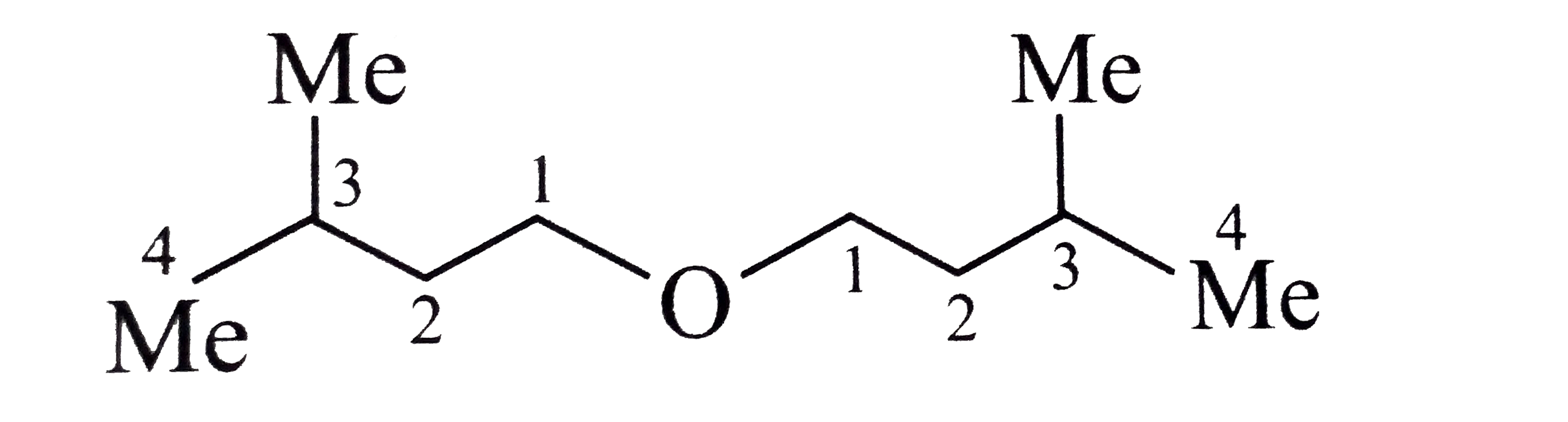 e.Structure of ether: 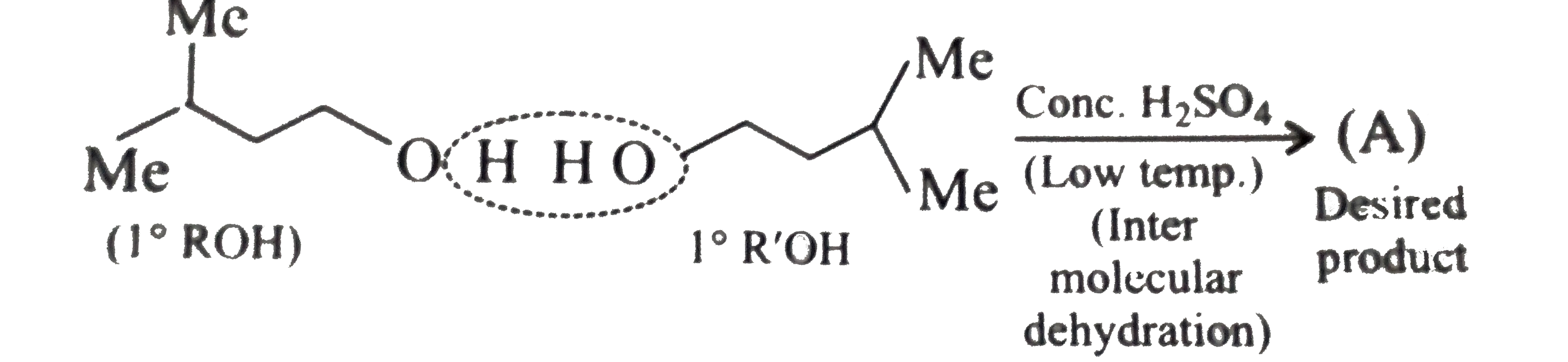 Method III is feasible. It is a symmetrical ether and with `1^(@) (R---)` groups. 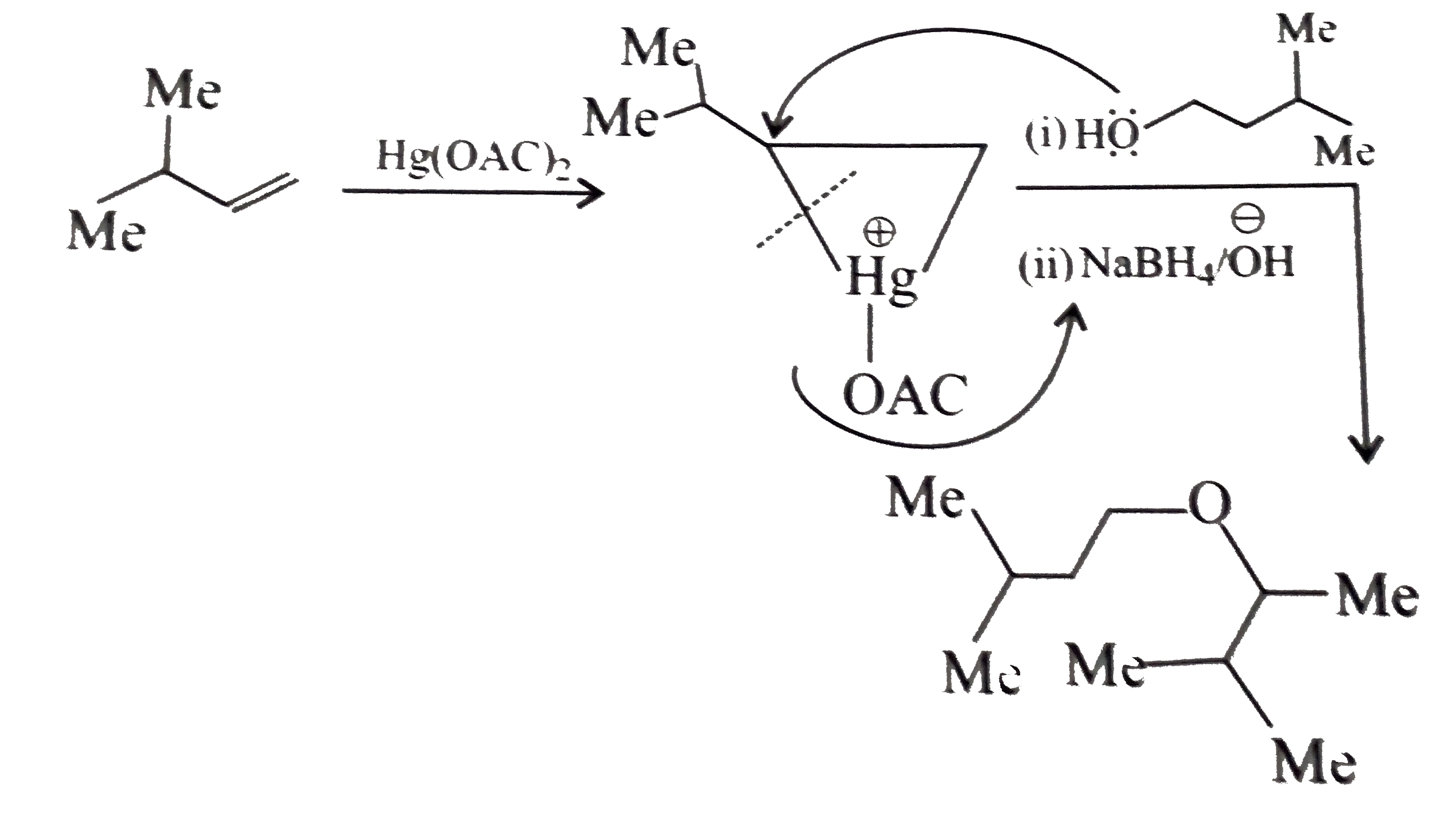 Method I is not feasible. Both RX and R'ONa wold be `1^(@)`. Better yield of ether is OBTAINED FORM `1^(@)` RX and `3^(@)` RONa.Method II is not feasible, it would give a different ether. |
|
| 24. |
Synthesis gas is a mixture of : |
|
Answer» STEAM and CARBON MONOXIDE |
|
| 25. |
Synthesies the following alcohols by using: a.Grignard reagent (G.R.) b.Not using G.R. (any other method). (I) (II) (III) (IV) (V) |
|
Answer» SOLUTION :a. SYNTHESIS by using G.R. (I) 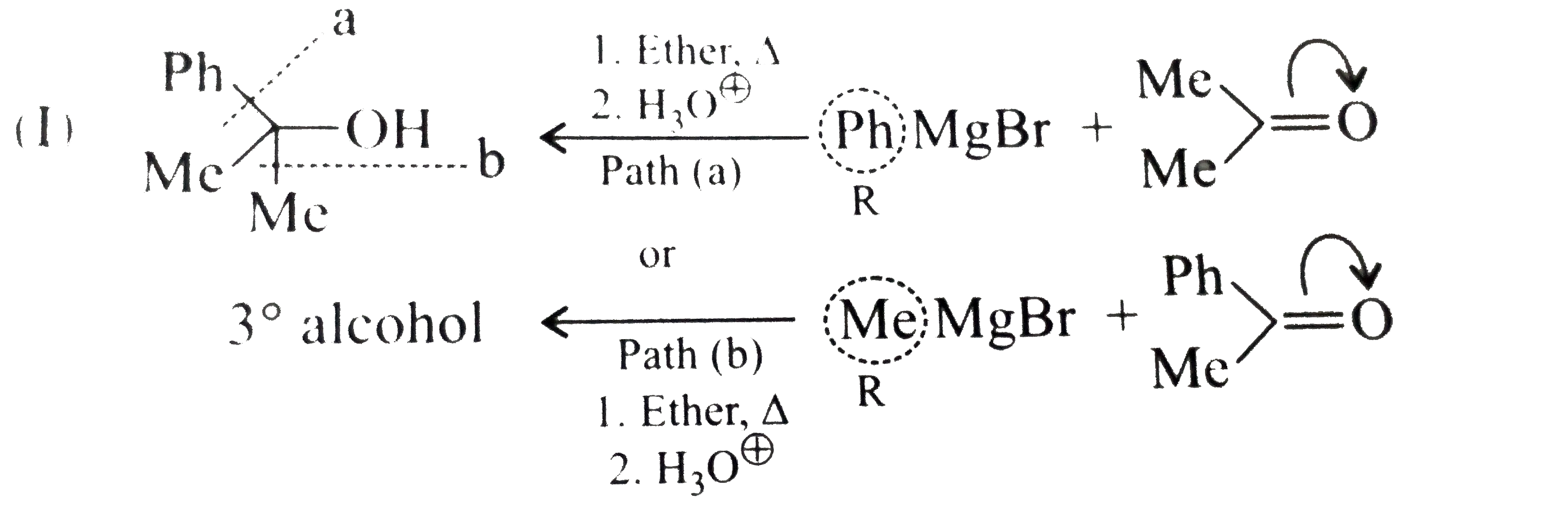 (II) 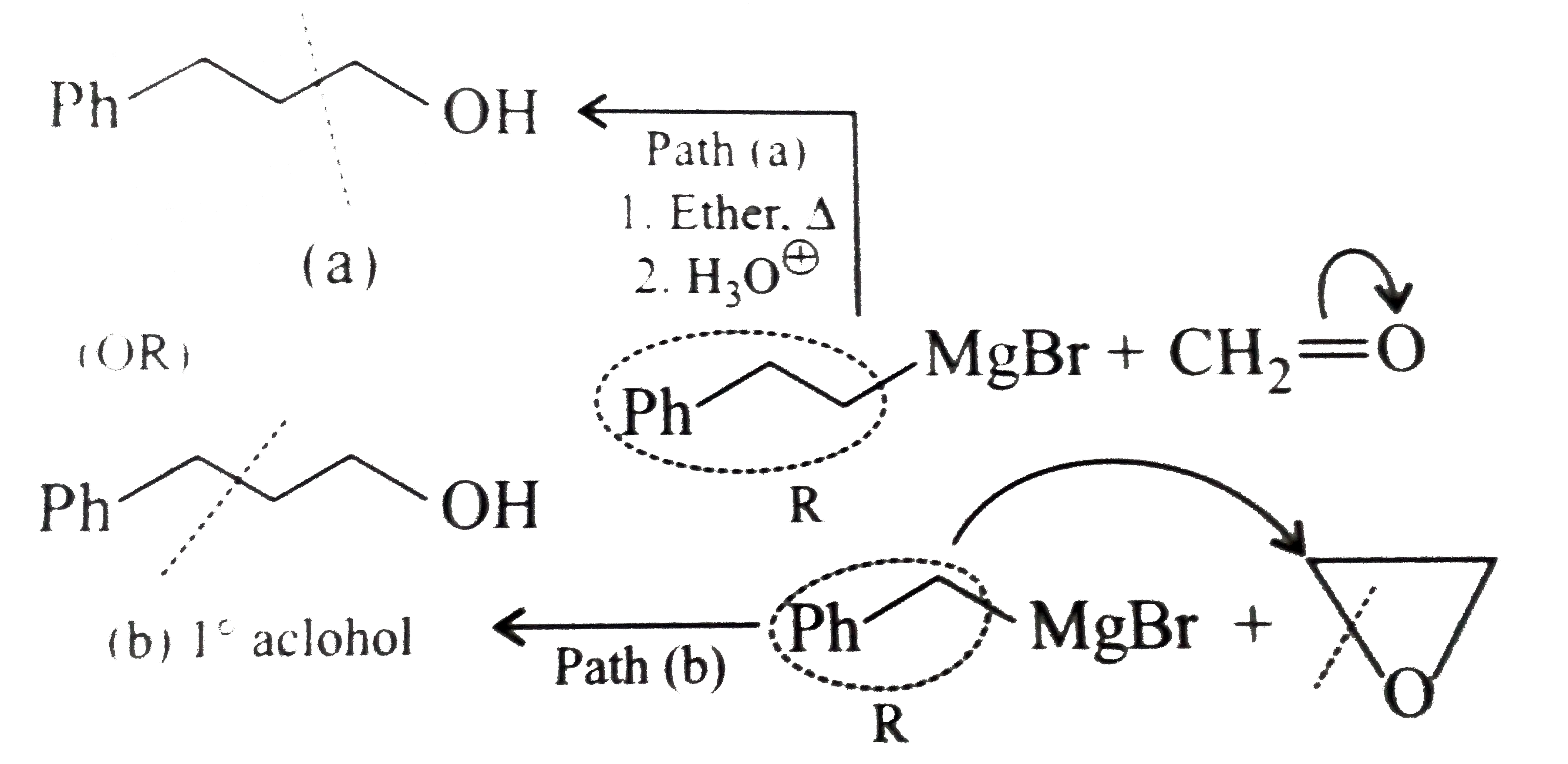 (III)  (IV) 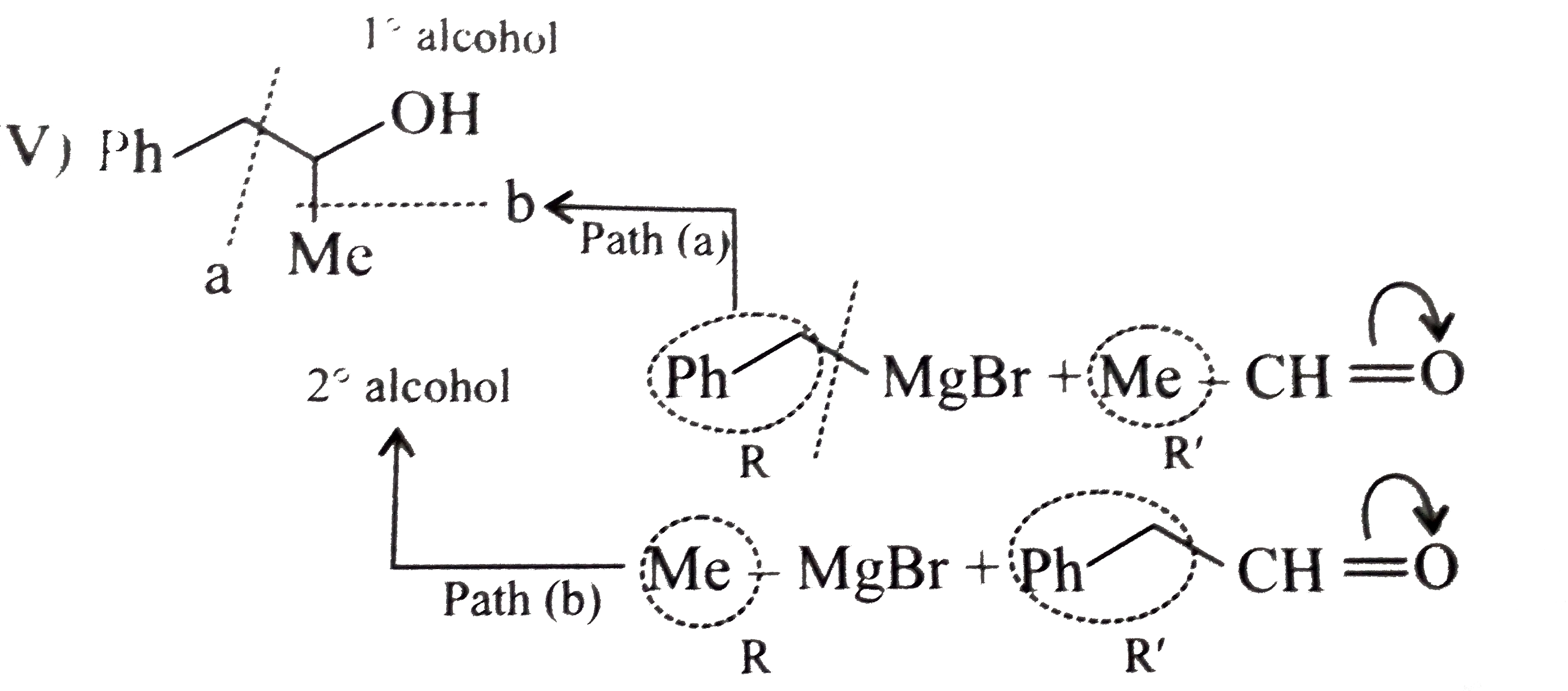 (V) 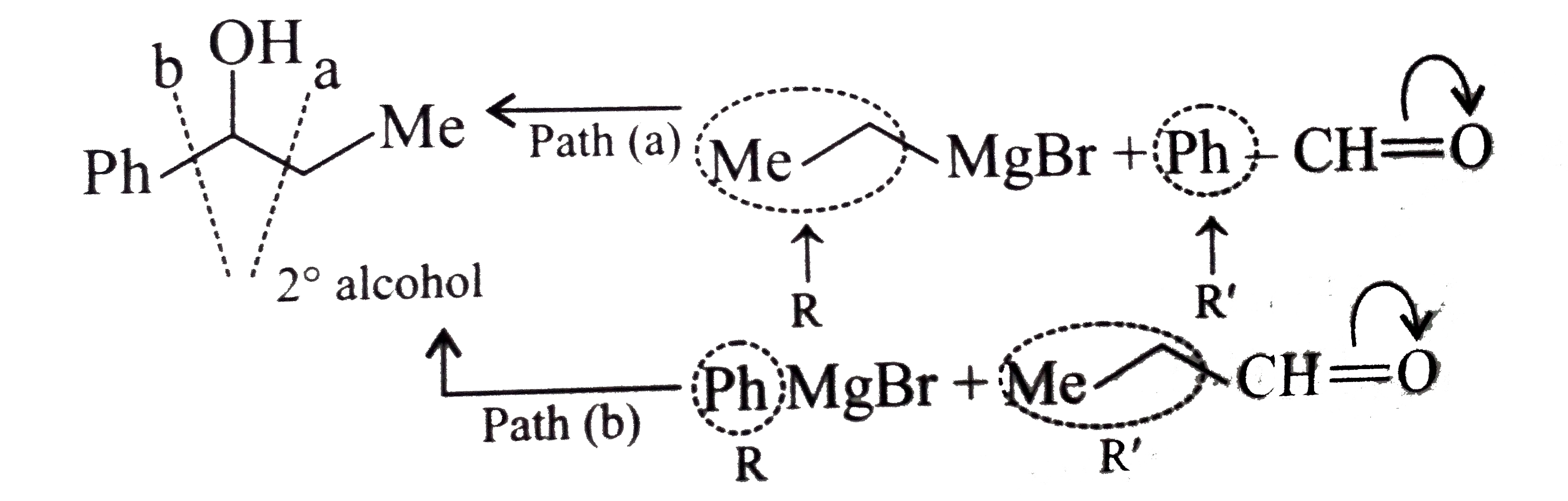 (B) Synthesis of ALCOHOLS without using G.R. (I) 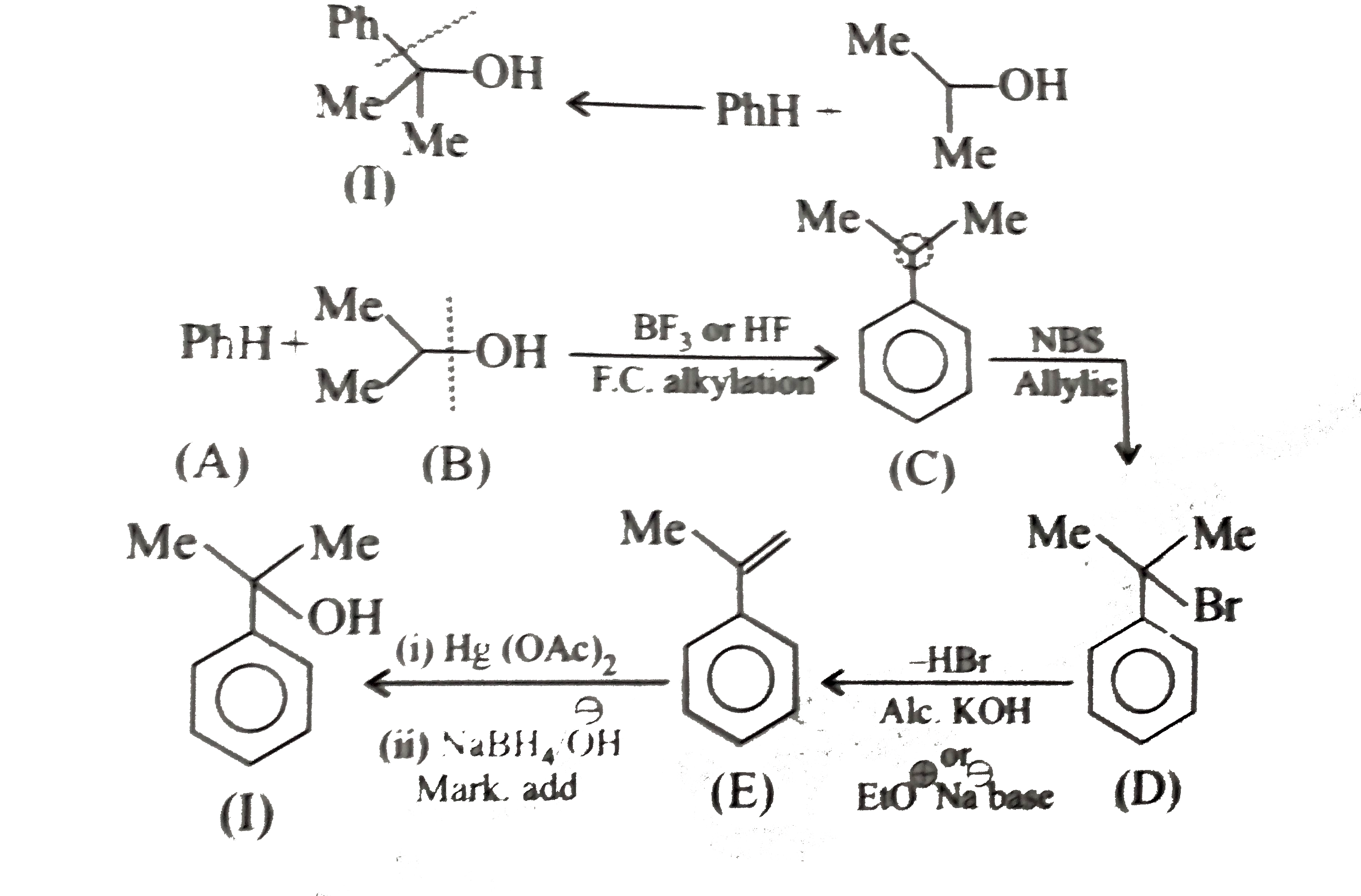 (II) 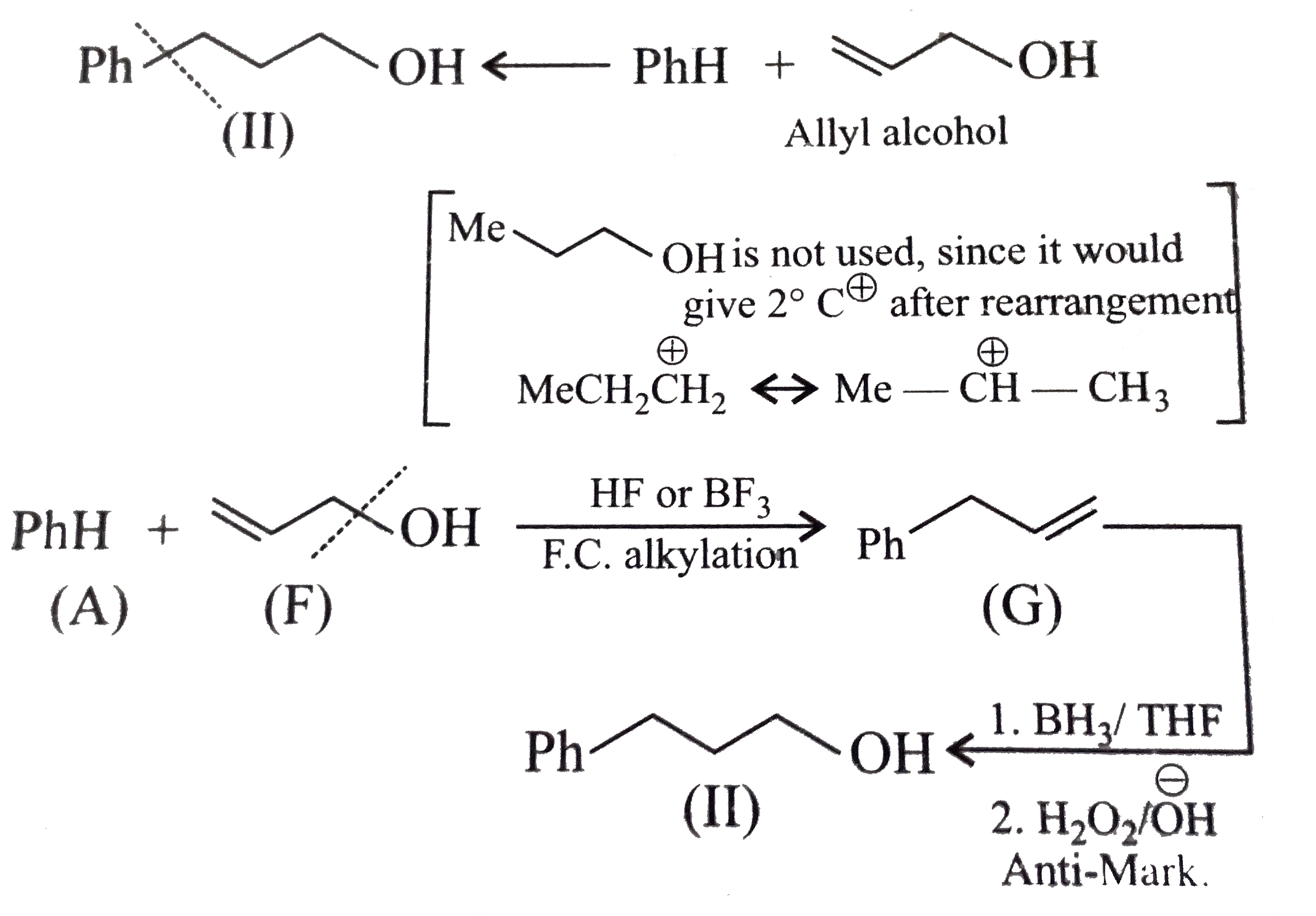 (III) 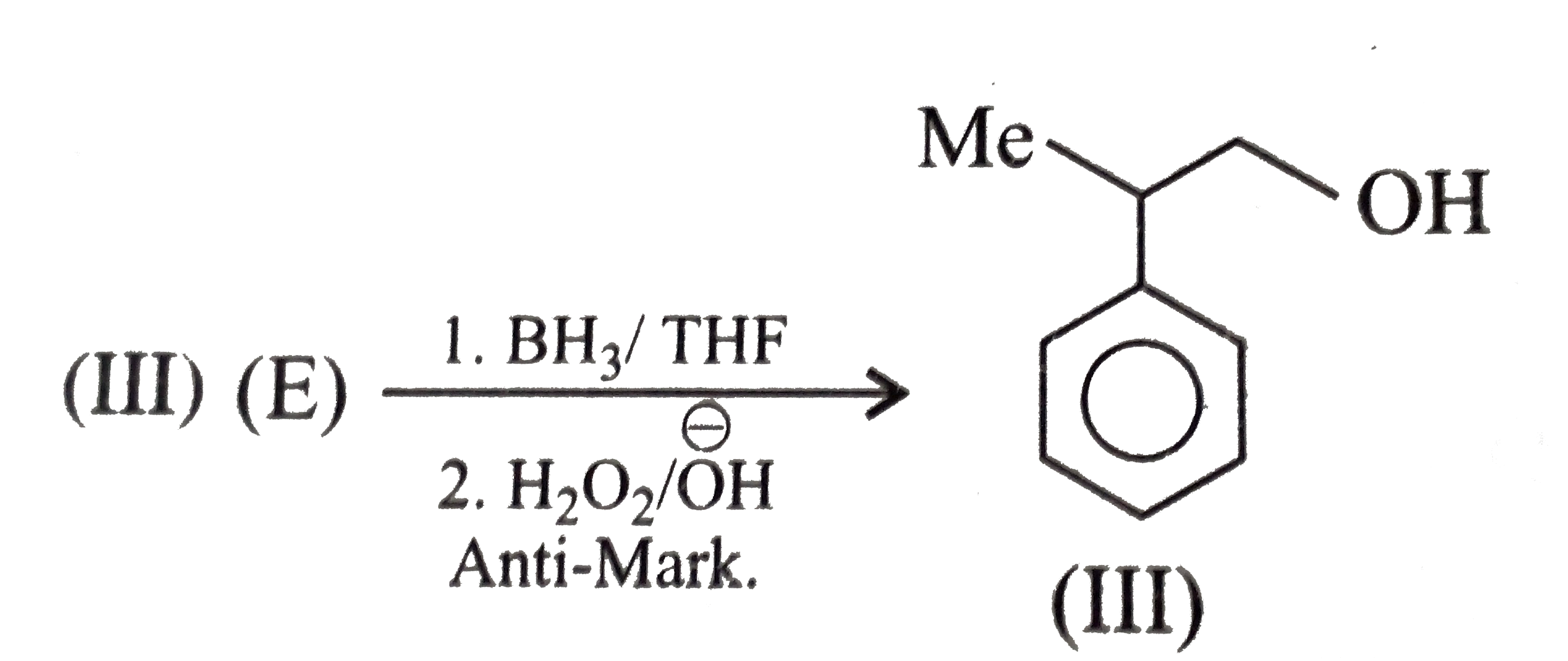 (IV) 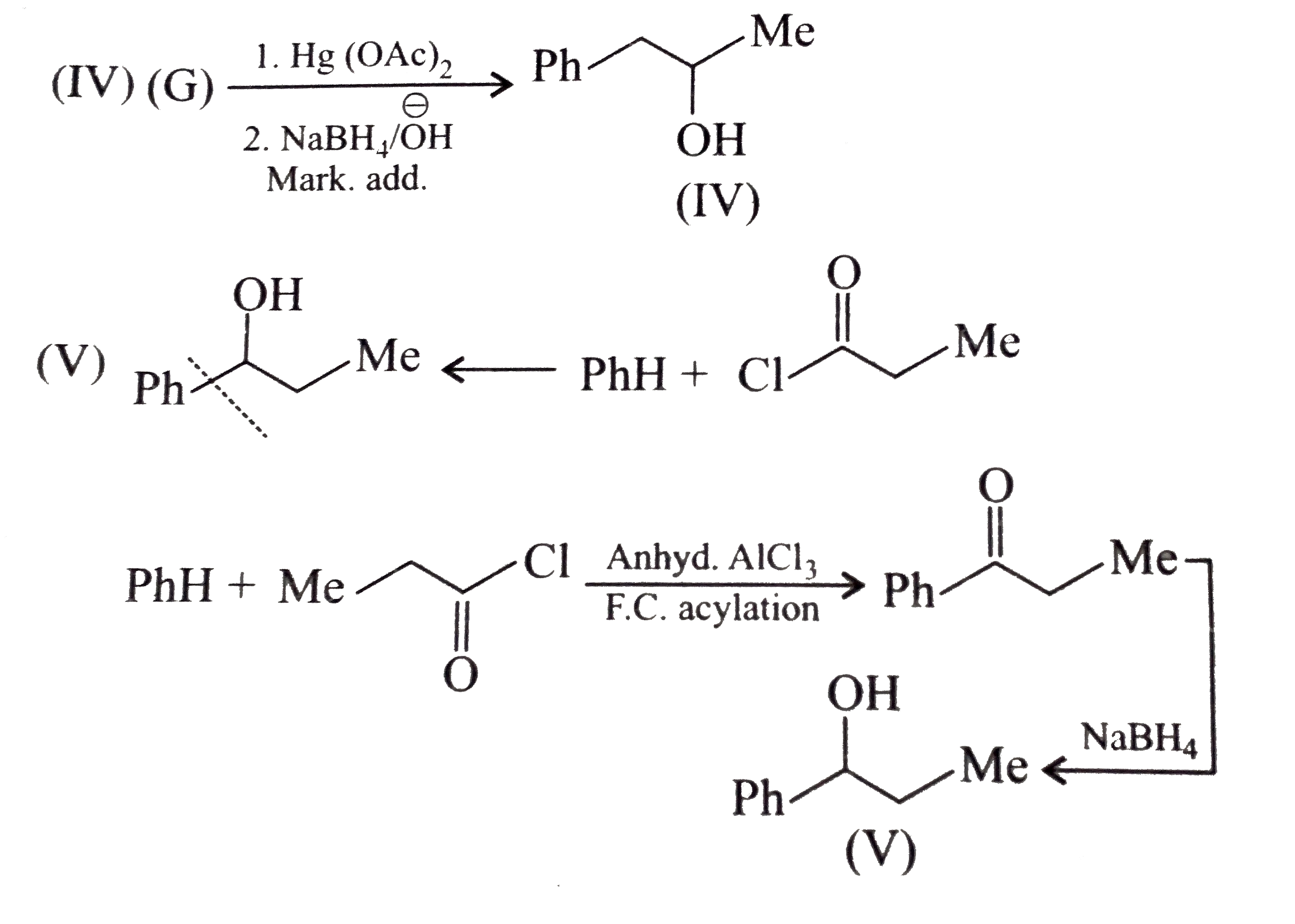
|
|
| 26. |
Symmetrically subsituted epoxides give the same products in both the acid catalysed and base catalyzed ring opening. An unsymmetrical epoxide gives different products under acid catalysed and base catalysed conditions. Under basic contions, the alkoxide ion simply attacks the less hindered carbon atom in an SN^(2) displacement. Under acidic conditions, the alcohol, attacks the protonated epoxide. Structure II and III show that the oxtrane carbon share part of positive charge. The tertiary carbon bear a larger part of positive charge and it is more strongly electrophilic. The bond between teritiary carbon and oxygen is weaker implying a lower transition state energy for attack at the teritary carbon. Attack by the weak nucleophilic is sensitive to the strength of electrophilic is sensitive to the strength of electrophile. Center attack takes place at more electrophilic carbon which is usually the more substituted carbon because it can better support the positive charge. |
|
Answer»
|
|
| 27. |
Symmetrically subsituted epoxides give the same products in both the acid catalysed and base catalyzed ring opening. An unsymmetrical epoxide gives different products under acid catalysed and base catalysed conditions. Under basic contions, the alkoxide ion simply attacks the less hindered carbon atom in an SN^(2) displacement. Under acidic conditions, the alcohol, attacks the protonated epoxide. Structure II and III show that the oxtrane carbon share part of positive charge. The tertiary carbon bear a larger part of positive charge and it is more strongly electrophilic. The bond between teritiary carbon and oxygen is weaker implying a lower transition state energy for attack at the teritary carbon. Attack by the weak nucleophilic is sensitive to the strength of electrophilic is sensitive to the strength of electrophile. Center attack takes place at more electrophilic carbon which is usually the more substituted carbon because it can better support the positive charge. |
|
Answer»
|
|
| 28. |
Symmetrically subsituted epoxides give the same products in both the acid catalysed and base catalyzed ring opening. An unsymmetrical epoxide gives different products under acid catalysed and base catalysed conditions. Under basic contions, the alkoxide ion simply attacks the less hindered carbon atom in an SN^(2) displacement. Under acidic conditions, the alcohol, attacks the protonated epoxide. Structure II and III show that the oxtrane carbon share part of positive charge. The tertiary carbon bear a larger part of positive charge and it is more strongly electrophilic. The bond between teritiary carbon and oxygen is weaker implying a lower transition state energy for attack at the teritary carbon. Attack by the weak nucleophilic is sensitive to the strength of electrophilic is sensitive to the strength of electrophile. Center attack takes place at more electrophilic carbon which is usually the more substituted carbon because it can better support the positive charge. |
| Answer» SOLUTION :N//A | |
| 30. |
Symmetrical alkane is prepared from which of the following reaction ? |
|
Answer» Resenmund's REACTION |
|
| 31. |
Symmetrically subsituted epoxides give the same products in both the acid catalysed and base catalyzed ring opening. An unsymmetrical epoxide gives different products under acid catalysed and base catalysed conditions. Under basic contions, the alkoxide ion simply attacks the less hindered carbon atom in an SN^(2) displacement. Under acidic conditions, the alcohol, attacks the protonated epoxide. Structure II and III show that the oxtrane carbon share part of positive charge. The tertiary carbon bear a larger part of positive charge and it is more strongly electrophilic. The bond between teritiary carbon and oxygen is weaker implying a lower transition state energy for attack at the teritary carbon. Attack by the weak nucleophilic is sensitive to the strength of electrophilic is sensitive to the strength of electrophile. Center attack takes place at more electrophilic carbon which is usually the more substituted carbon because it can better support the positive charge. What will be the products in following reactions |
|
Answer»
|
|
| 32. |
Symmetrical alkane can be prepared from alkyl halideby, |
|
Answer» Wurtzs REACTION |
|
| 33. |
Symbolic name for teflon is: |
|
Answer» PTFE |
|
| 35. |
Swimming pools are disinfected by bubbling through water in controlled quantity of: |
|
Answer» `Br_2` |
|
| 37. |
Sweet spirit of nitre is |
|
Answer» ETHYL NITRITE |
|
| 38. |
Sweet fruity smell is obtained by the reaction of ethanol with …….. |
|
Answer» `PCI_(3)` Esther is a compound having fruity smell. |
|
| 39. |
Swati's father wanted to go to the hospital to see his ailing friend. Swati insisted to accompany his father. On reaching the hospital, swati noticed a perculiar smell. after reading the above passage, answer the following questions: (i) Name the chemical which causes the hospital smell. (ii) What is the use of this chemical and how does it work? |
|
Answer» Solution :(i) the hospital smell is not that of a particular disease but is the smell of a chemical compound called iodoform `(CHI_(3))`. (ii) Iodoform is a mild disinfectant and is used for CLEANING the FLOORS of the corridors, WARDS, etc. it can also be used as an antiseptic for skin INFECTIONS, sores, bruises, BOILS, burns, etc. when applied on skin, iodoform decomposes to release iodine. it is iodine which acts as the actual antiseptic, killing bacteria and fungi. iodoform is also safer than other antiseptics if it is accidently swallowed. |
|
| 40. |
Swart reaction is used to prepare |
|
Answer» R-F |
|
| 41. |
Suspension of slaked lime in water is called : |
|
Answer» WASHING of lime |
|
| 42. |
Suspended impurities from a colloidal solution of gum in water can be removed by |
|
Answer» FILTRATION |
|
| 43. |
Surfactants and detergents have the same common property of_____ in them: |
|
Answer» Detergency |
|
| 44. |
Surfacetant molecules can cluster together as micelles, which are colloid sized cluster of molecules. Micellesfrom only above critical micelle concentration (CMC) and above centain temperature called K raft temperature. DeltaH of micelle formation can be positive or negative. Which is correct statement(s) about micelle formation? |
|
Answer» `DeltaS` of MICELLE formation is positive |
|
| 45. |
Surface tension of lyophobic sols is usually |
|
Answer» LOWER than DISPERSION medium |
|
| 46. |
Surface tension of lyophilic sols is: |
|
Answer» LOWER than that of `H_2O` |
|
| 47. |
Surface tension of lyophilic sol is |
|
Answer» LOWER than that of `H_(2)O` |
|
| 48. |
Surface layer of solid means |
|
Answer» atoms PERESENT in the upper LAYER of the solid . |
|
| 49. |
Surface layer of a solid means |
|
Answer» atoms PRESENT in the upperlayer of the solid |
|
| 50. |
Supposingthe energy of the energy levelsin the hydrogen atom is given under Energy level K L M N.. n=1 n=2n=3 n=4=n=infty Total energy of anat n =1 is 864 a.u the ecitation energy needed to raise the electron from M levelto n= infty wouldbe : |
|
Answer» 192 |
|
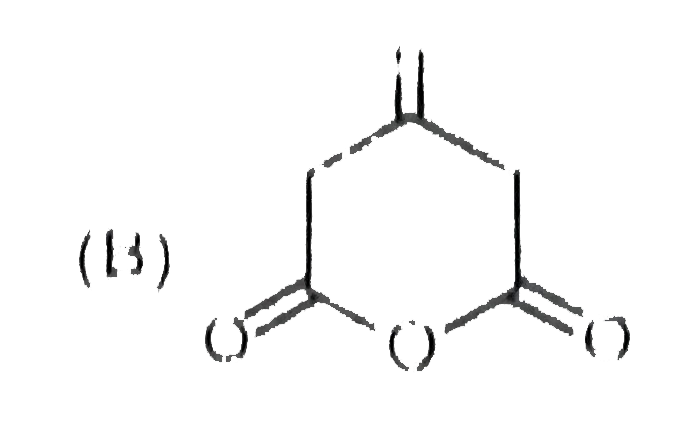
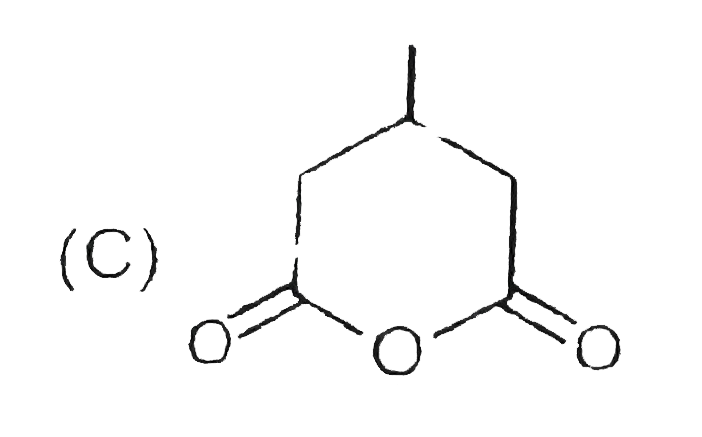
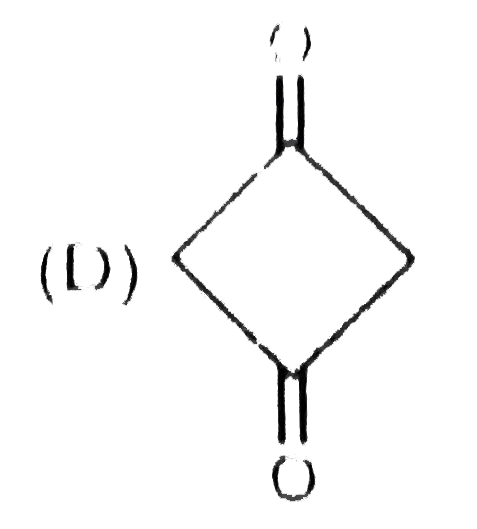
_S01_405_O01.png)
_S01_405_O02.png)
_S01_405_O03.png)
_S01_405_O04.png)
_S01_403_O01.png)
_S01_403_O02.png)
_S01_403_O03.png)
_S01_406_O01.png)
_S01_406_O02.png)
_S01_406_O03.png)
_S01_402_O01.png)
_S01_402_O02.png)
_S01_402_O03.png)
_S01_402_O04.png)