Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Surcose decomposes in acid solution into glucose and fructose according to the first order rate law .with t_((t)/(2))=3.00 hours.What fraction of sample of surcose remains after 8 hours? |
|
Answer» Solution :`"surcose"_((AQ))overset(H^(+))to"Glucose"_((aq))+"Fructose"_((aq))` The reaction is FIRST order where ,`t_((1)/(2))=3.00` hours `therefore` So, `K=(0.693)/(t)=(0.693)/(300h)=0.231 hr^(-1)` CALCULATE fraction of sample of sucroso REMAINS after 8 hours. Suppose the initial con.of sucrose=`[R]_(0) mol L^(1)` Remaining concentration of sucrose after 8 hours =`[R]_(t) mol L^(-1)` So,`("Sucrose in initial")/("Sucrose after t time")=([R]_(0))/([R]_(t))` k=`(2.303)/(t)` log `([R]_(0))/([R]_(t))` For first order reaction , `therefore 0.231 hr^(-1)=(2.303)/(8 hr) log ([R]_(0))/([R]_(t))` `therefore log ([R]_(0))/([R]_(t))=(0.231 hr^(-1)xx8 hr)/(2.303)=0.8024` Remaining sucrose after 8 hour =`[R]_(t)` and Fraction of remaining sucrose `([R]_(0))/([R]_(t))` `therefore` Fraction of remaining sucrose =`(1)/(([R]_(0))/([R]_(t)))=(1)/(6.3445)` |
|
| 2. |
Suppose you have to determine the percentage of carbon dioxide in a sample of a gas available in a container. Which is the best absorbing material for the carbon dioxide? |
|
Answer» COLD, SOLID CALCIUM hydroxide |
|
| 3. |
Suppose you have four test tubes labelled as 'A' , 'B' , 'C' and 'D' ,'A' contains plain water 'B' contains solution of an alkali 'C' contains solution of and 'D' contains solution of sodium chloride which one of these solutin will turn phenoplhthalein solutionpink ? |
| Answer» Answer :B | |
| 4. |
Suppose we have cube of 1.00 cm length. It is cut in all three directions, so as to produce eight cubes, each 0.50 cm on edge length. Then suppose these 0.50 cm cubes are each subdivided into eight cubes 0.25 cm on edge length, and so on. How many of these successive subdivisions are required before the cubes are reduced in size to colloidal dimensions of 100 nm. |
|
Answer» Solution :We find that every division in two equal halves also reduces the size of edge LENGTHS to one half. In first subdivision 1 cm is reduceds to `0.5 cm = (1)/(2) cm.` In second subdivision 0.5 cm is reduced to `0.25 cm = (1)/(4) cm = ((1)/(2))^(2) cm` In n subdivision 1 cm is reduced to `((1)/(2))^(n)`. Size of colloidal particles lies between 1 to 1000 mm. Thus to make n subdivision required PARTICLE size may be ATTAINED. `((1)/(2))^(n) = 100 nm = 100 xx 10^(-9) m = 100 xx 10^(-7) cm.` n log 2 = 5 `n xx 0.3010 = 5.` `n = (5)/(0.3010) = 16.61 = 17` subdivisions are required for dimension of 100 nm. |
|
| 5. |
Suppose two elements X and Y combines to form two compounds XY_(2) and X_(3)Y_(2). When 0.1 mol of XY_(2) weighs 10 g and 0.05 mol of X_(3)Y_(2) weighs 9 g, the atomic weights of X and Y are |
|
Answer» 40, 30 |
|
| 6. |
Suppose uniusulated copper or aluminium wires are used for flow of electricity from pole to pole in the street. Do you expect a better flow in summer or winter and why? |
| Answer» Solution :In a metal, the electrical conduction decreases with increase of temperature. Hence, we expect a better flow of CURRENT through the WIRE in winter when the temperature is LOW than in summer when the temperature is HIGH. | |
| 7. |
Suppose the universe were different than it is and that the electron spin quantum number could only have of a value of +(1)/(2) . Assume that the Pauli's exclusion principle still holds and that the value of other quantum numbers were the same, which atomic number could correspond to the "noble gases" in this universe? |
|
Answer» 1,5,9... |
|
| 8. |
Suppose the mass of a single Ag atom is m. Ag metal crystallizes in fcc lattice with unit cell of length a.The density of Ag metal in terms of a and m is |
|
Answer» `(4m)/(a^(3))` `=("No. of atoms" xx "Mass of each ATOM")/("Volume of the unit cell")` `=(4xxm)/(a^(3))` (`:.` in fcc, no of atoms =4) |
|
| 9. |
Suppose the reaction PCl_(5(g))hArr PCl_(3(s))+Cl_(2(g)) is a closed vessel tat equlibrium stage. What is the effect on equlibrium concentration of Cl_(2(g)) byadding PCl_(5) at constant temperature |
|
Answer» <P>DECREASES |
|
| 10. |
Suppose the elements X and Y combine to form two compounds XY_(2) and X_(3)Y_(2). When 0.1 mole of XY_(2) weights 10 g and 0.05 mole of X_(3)Y_(2) weights 9 g, the atomic weight of X and Y are |
|
Answer» 30, 20 `0.1=10/(X+2Y)=X+2Y=100` ...(i) For `X_(3)Y_(2)""n=W/M` `0.05=9/(3X+2Y)=3X+2Y=180` ...(ii) From (i) and (ii) `2X=80` `X=40 and 2Y=100-40=60` `=Y=30` |
|
| 11. |
Suppose the equilibrium constant for the reaction, 3M^(3+) rarr 2M^(2+)(aq) +M^(5+) (aq) |
|
Answer» `E_(M^(5+)//M^(2+))^(@) = 0.6V` `E_(M^(3+)//M^(2+))^(@) = 0.8V` `(2.303RT)/(F) = 0.06V` |
|
| 13. |
Suppose a solid solution is formed between two substances, one whose particles are very large and the other whose particles are very small. What type of this solid solution is likely to be ? |
| Answer» SOLUTION :INTERSTITIAL SOLID solution. | |
| 14. |
Suppose an orbital may accomodate 3 electrons then the number of elements in IV period. |
|
Answer» 18  no. of ORBITAL = 9 (ONE orbital OCCUPY 3electron) then no. of elements `=9xx3=27` |
|
| 15. |
Suppose a fully charged battery containes 1.50 litre of 5.0 M H_(2)SO_(4). What will be the concentration of H_(2)SO_(4) left in battery after 2.50 ampere current is drawn from the battery for 6.0 hour? Assume volume of solution ramains constant. |
|
Answer» |
|
| 16. |
Suppose 60% w//w aqueous solution of glucose ( C_(6)H_(12) O_(6) ) and 20% w //w aqueous solution of urea ( NH_(2)CONH_(2) )have equal molarity, then which solution has higher density : |
|
Answer» Both have EQUAL density |
|
| 17. |
Suppose 250 mL of a 0.433 M solution of CuCl_(2) is electrolysed. How long will a current of 0.75 A have to run in order to reduce the concentration of Cu^(2+) to 0.167 M? What mass of Cu(s) will be deposited on the cathode during this time ? |
|
Answer» Solution :Mole of `Cu^(2+)` reduced `= 0.433 XX 0.250 - 0.167 xx 0.250` = 0.0665. Wt. of Cu deposited `= 0.0665 xx 63.5g` Let the time to DEPOSIT this amount of Cu be t seconds `THEREFORE` no. of F = no. of EQ. `= (0.75 xx t)/(96500) = 0.0665 xx 2` `t = 1.71 xx 10^(4)` seconds. |
|
| 18. |
Support the statement given below with relevent examples. |
|
Answer» Solution :"The choce of a reducing agent in a particular case depends on the thermodyanmics factor". The STATEMENT is correct. We consider both relations, `DELTA G = Delta H -T Delta S` (or) `Delta G^(@) = -RT "inK"` For a particular reducing agent to WORK with a metallic OXIDE. (i) the value of `Delta G` should be negative. (II) `Delta S` should be positive. |
|
| 19. |
Supply structures for H through K. Given : " An aldohexose" overset(NH_(2)OH"/base")(to)H overset(Ac_(2)O"/"NaOAc)(to)I overset(-HOAC)(to)J overset(NaOMe"/"MeOH)(to)K. Explain the last step. What is net structural change. Name this overall method. Discuss the possibility of epimer formation. |
|
Answer» Solution :H is an oxime `HOCH_(2)(CHOH)_(4)CH=NOH`, I is the COMPLETELY acetylated oxime, `AcOCH_(2)(CHOAc)_(4)CH=NOAc` that loses 1 mole of HOAc to form J, `AcOCH_(2)(CHOAc)_(4)C EQUIV N, K` is an aldopentose, `HOCH_(2)(CHOH)_(3)CHO`. The acetates undergo TRANSESTERIFICATION to give methyl acetate freeing all the sugar OH.s. This is followed by reversal of HCN addition. There is loss of one C from the carbon chain. Wohl degradation. The `alpha-CHOH` becomes the `-CH=O` without any configurational changes of the other chiral carbons. Thus no epimers are formed. |
|
| 20. |
Supply the missing reagents. |
| Answer» Solution :The overall transformation over TWO steps involves replacing an alcohol HYDROXYL group by a cyano group with inver sion of configuration. To accomplish this, we need to convert the alcohol hydroxyl to a GOOD leaving group in the first step, which we do by making it a methanesulfonate ester (a mesylate) using methanesulfonyl chloride in pyridine. The second step is an `S_(N)2`substitution of the methanesulfonate (mesyl) group, which we do using potassium or sodium cyanide in a POLAR aprotic solvent such as dimethylformamide (DMF). | |
| 21. |
Supply structures for H through K. Given : " An Aldohexose "overset (MH_2OH"/base ")(to)H overset (Ae_2O"/"NaOAC)(to)I overset (-HOAC)(to)J overset (NaOMe"/"MeOH)(to)K. Name this overall method. |
| Answer» SOLUTION :WOHL DEGRADATION. | |
| 22. |
Supply structures for H through K. Given : " An Aldohexose "overset (MH_2OH"/base ")(to)H overset (Ae_2O"/"NaOAC)(to)I overset (-HOAC)(to)J overset (NaOMe"/"MeOH)(to)K. What is net structural change. |
| Answer» SOLUTION :There is loss of one C from the CARBON chain. | |
| 23. |
Supply structural formulae for missing compounds. (i) H_(2)C=CH_(2)+(A) rarr ClCH_(2)-CH_(2)OH underset(413K)overset(H_(2)SO_(4))(rarr) (B) underset(Delta)overset("alc. KOH")(rarr) (C ) (ii) CH_(3)CHBrCH_(3)overset("alc. KOH")(rarr)(A)underset("Peroxide")overset(HBr)(rarr)(B)overset(CH_(3)ONa)(rarr)(C ) (iii) Me_(3)CBroverset("alc. KOH")(rarr)(A) overset(HOCl)(rarr) (B) overset(NaOH)(rarr)(C ) |
|
Answer» `{:(A,B,C),(CH_3CH=CH_2,CH_3CH(BR)CH_2Br,CH_3CH(OH)CH_3):}` 
|
|
| 24. |
Supply structures for H through K. Given : " An Aldohexose "overset (MH_2OH"/base ")(to)H overset (Ae_2O"/"NaOAC)(to)I overset (-HOAC)(to)J overset (NaOMe"/"MeOH)(to)K. Discuss the possibility of epimer formation. |
| Answer» SOLUTION :The `alpha-CHOH` becomes the `-CH=O` WITHOUT any configurational changes of the other chiral CARBONS. Thus no EPIMERS are formed. | |
| 25. |
Supply structures for H through K. Given : " An Aldohexose "overset (MH_2OH"/base ")(to)H overset (Ae_2O"/"NaOAC)(to)I overset (-HOAC)(to)J overset (NaOMe"/"MeOH)(to)K. Explain the last step. |
| Answer» Solution :The acetates undergo transesterification to GIVE methyl ACETATE freeing all the SUGAR OH's. This is FOLLOWED by reversal of HCN addition. | |
| 26. |
Superphosphate of lime is obtained from the reaction of: |
|
Answer» Calcium CARBONATE with PHOSPHORIC acid |
|
| 27. |
Superphosphate of lime is used in: |
|
Answer» CEMENT industry |
|
| 28. |
Superphosphate of lime is an example of ........... phosphate |
| Answer» ANSWER :A | |
| 29. |
Superphosphate is the mixture of |
|
Answer» CALCIUM phosphate and dil. `H_(2)SO_(4)` |
|
| 30. |
Supercritical CO_(2) is used as |
|
Answer» DRY ice |
|
| 31. |
Super octet molecule is : |
|
Answer» `ClF_3` |
|
| 33. |
Super halogen is: |
|
Answer» `F_2` |
|
| 34. |
Super glue or crazy glue is |
|
Answer» POLY METHYL methacrylate 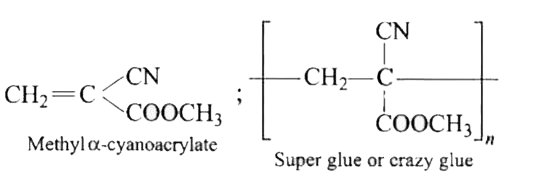
|
|
| 35. |
Super conductors are derived fTom compounds of |
|
Answer» p-Block elements |
|
| 36. |
Sunita is of marriagable age. She wears spectables. She is afraid that the she may not find to suitable suggested her to wear contact lenses. After reading the above passage , answer the following questions : (i) What values are expressed by Manisha ? (ii) Name the polymer used in making contact lenses. (iii) How is this polymer prepared ? (iv) Besides contact lenses what are the other uses of this polymer ? |
|
Answer» Solution :(i) Manisha expressed the values about looking ATTRACTIVE by wearing contact lenses. (ii) Polymer USED in making contact lenses is polymethyl methacrylate (PMMA). (iii) it is prepared by free radical polymerization of METHYL methacrylate. 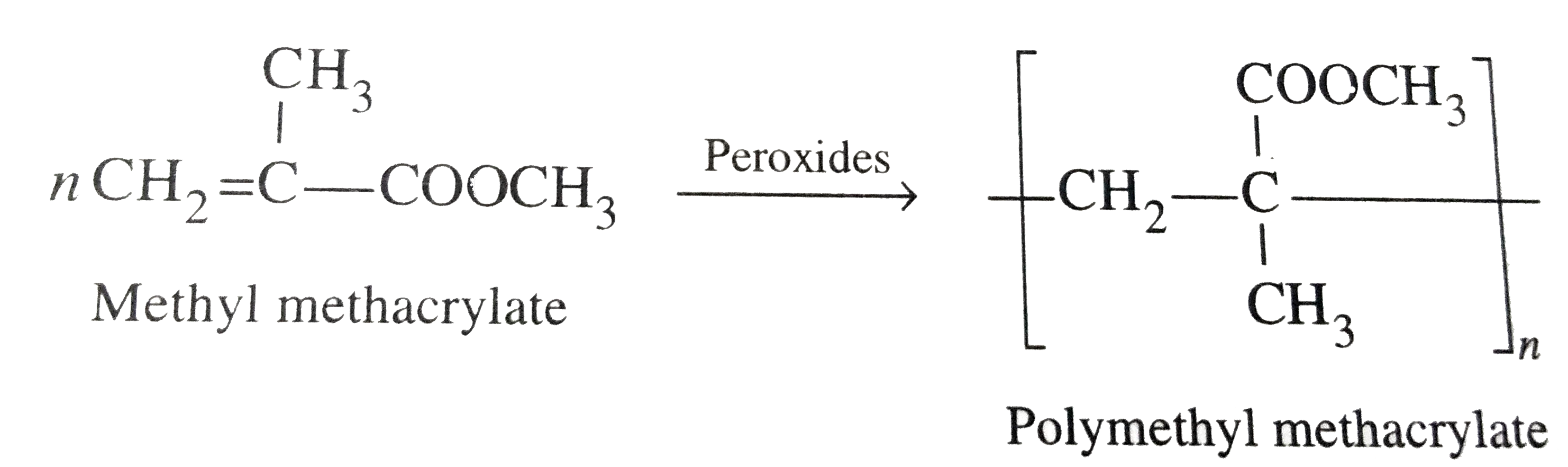 (iv) Besides contact lenses , PMMA is used for manufactering transport domes , light shades , sign boards , TRANSPARENT domes and sky domes , AIRCRAFT windows, dentures and plastic jewellery. |
|
| 37. |
Sum of x and y is |
|
Answer» 2 |
|
| 38. |
Sum X + Y = ? |
|
Answer» 1 |
|
| 40. |
Sum of total no. of stereoisomers (A) and total no. of fractions (B) for the compound is. |
Answer» 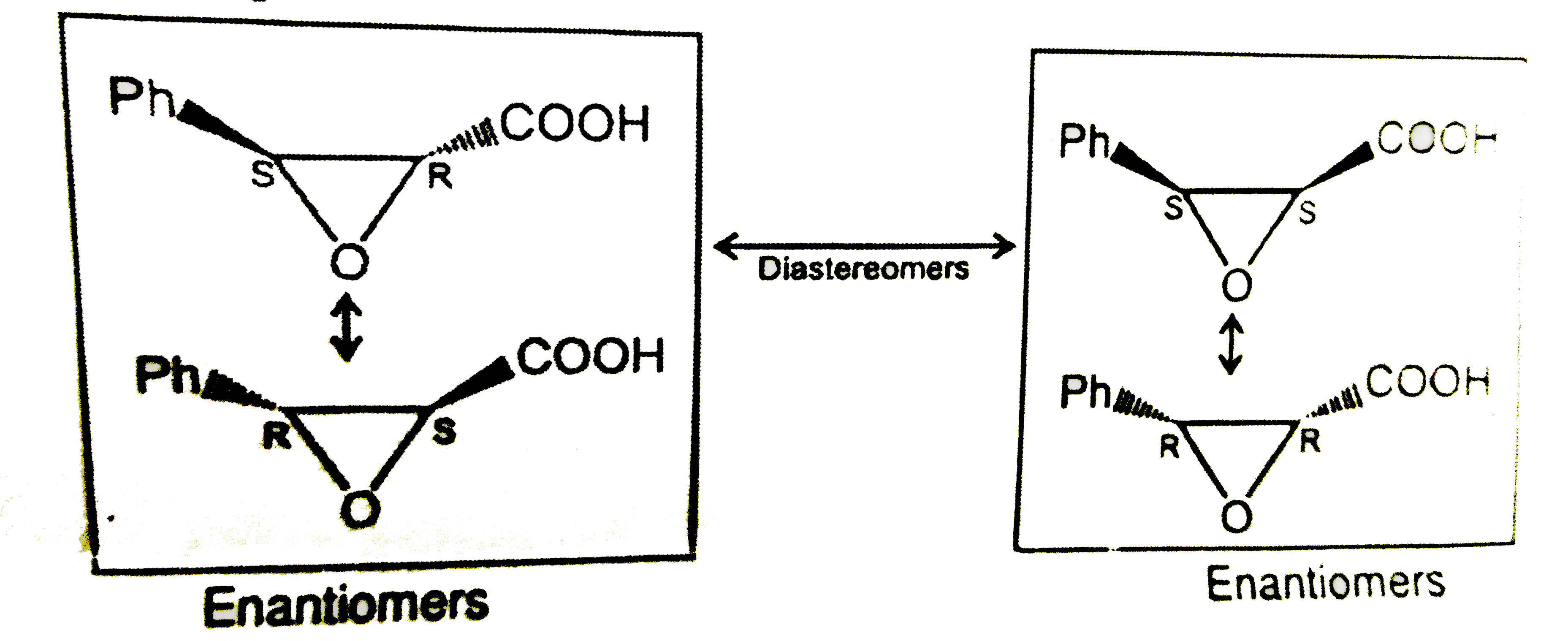
|
|
| 42. |
Sum of molecular mass of iodides produced in following reaction is : If answer of part ( a) is x, part ( b) is y and part( c) is z then present sum of x+y+z in the OMR sheet. For example : if answer of ( a) is 12, ( b) is 13 and (c ) is 3 you will fill 0028 in OMR sheet. |
Answer» 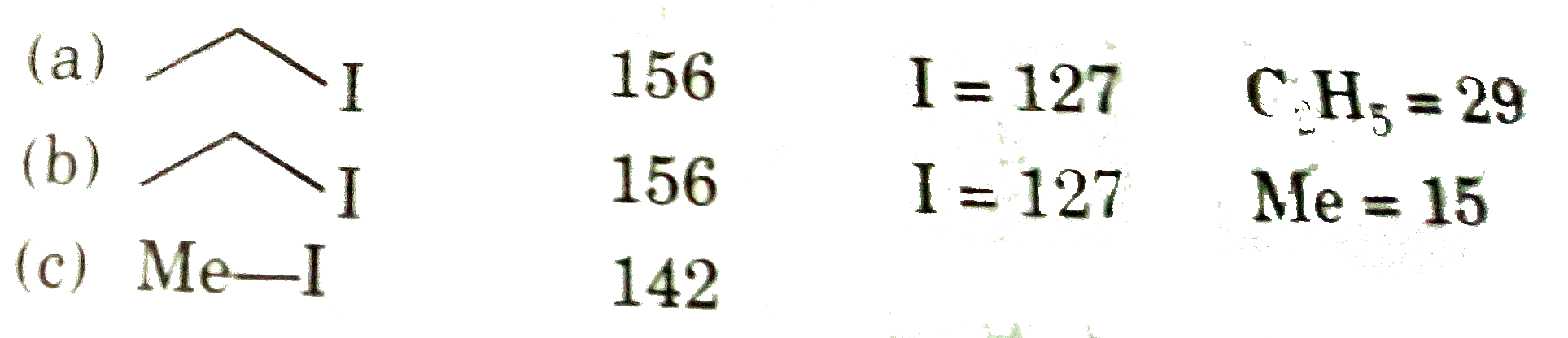
|
|
| 43. |
Sum of number of stereoisomer (C ) Degree of unsaturations in (D). |
Answer» 
|
|
| 44. |
Sum of number of N atoms present in all the products of the following reactions is……… |
|
Answer» |
|
| 45. |
Sulphuryl chloride (SO_(2)Cl_(2)) reacts with white phosphorous (P_(4)) to give |
|
Answer» `PCl_(5), SO_(2)` |
|
| 46. |
Sulphuryl chloride (SO_(2)Cl_(2)) reacts with white phosphorus (P_(4)) to give : |
|
Answer» `PCl_(5), SO_(2)` |
|
| 47. |
Sum of denticityof following ligands are Glycinaty ion, Oxalate ion, o-phenathroline, 2,2-bipyridyl, diethylenetriamine, ethylenediamine |
|
Answer» |
|
| 48. |
Sulphurous acid is reducing agent due to |
|
Answer» absence of hydrogen bonding `[O=underset(OH)underset|overset(* *)S-OH]` |
|