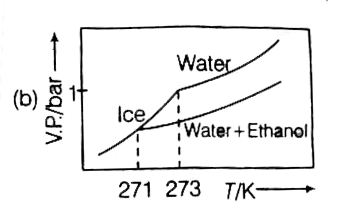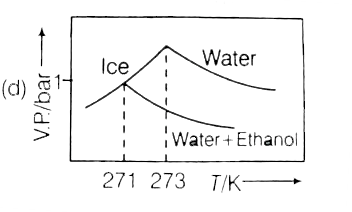Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Purification of aluminium by electrolytic refining is carried out by : |
|
Answer» Hoope process |
|
| 2. |
Purification of alumina is essential because : |
|
Answer» Impure ALUMINA is a very POOR conductor of electricity |
|
| 4. |
Purest form of silica is : |
|
Answer» Quartz |
|
| 9. |
Pure water is kep in a vessel and it remains exposed to atmospheric CO_(2) which is absorbed, then its pH will be |
|
Answer» GREATER than 7 |
|
| 10. |
Pure water is saturated with pure solid AgCl, a silver electrode is placed in the solution and the potential is measured against normal calomel electrode at 25^(@)C. This experiment is then repeated with a saturated solution of Agl. If the difference in potential in the two cases is 0.177V. What is the ratio of solubility product (solubility) of AgCl and Agl at the temperature of the experiment? |
|
Answer» `10^(3)` `E_(1)+E_("oxid")+E_("calomel")` `=E'-(0.0591)/(1)logK_(sp_(1))+E_("calomel")` `E_(2)=E'-(0.0591)/(1)logK_(sp_(2))+E_("calomel")` `E_(2)-E_(1)=0.177=0.0591log((K_(sp_(1)))/(K_(sp_(2))))` `(K_(sp_(1)))/(K_(sp_(2)))=10^(3)` |
|
| 12. |
Pure water is saturated with pure solid AgCl, a silver electrode is placed in the solution and the potential is measured agains normal calomet electrode at 25^(@)C. This experiment is then repeated with a saturated solution of AgI. If the difference in potential in the two cases is 0.177V. What is the ratio of solubility product (solubility) of AgCl and AgI at the temperature of the experiment? |
| Answer» Answer :A | |
| 13. |
Pure water is kept in a vessel and it remains exposed to atmospheric CO_2 which is absorbed, then its pH will be……. |
|
Answer» greater than 7 |
|
| 14. |
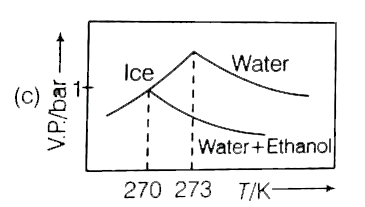
Pure water freezes at 273 K and 1 bar. The addition of 34.5g of ethanol to 500g of water changes the freeing point of the solution. Use of f.p. depression constant of water as 2Kg mol^(-1). The figures shown below represents plot of vapour pressure (V.P.) versus temperature (T) (mol wt. of ethanol =46 g mol^(-1)). Among the following the option representing change in the f.p. is |
|
Answer»
|
|
| 15. |
Pure water freezes at 273 K and 1 bar. The addition of 34.5 g of ethanol to 500 g of water changes the freezing point of the solution. Use the freezing point depression contant of water as 2 K kg mol^(-1). The figures shown below represent polts of vapour (V.P.) versus temperature (T). [Molecular weight of ethanol is 46g mol^(-1)] Among the following, the option representing change in the freezing point is |
|
Answer»
|
|
| 16. |
Pure water freezes at 273 K and 1 bar. The addition of 34.5 g of ethanol to 500 g of water changes the freezing point of the solution. Use the freezing point depression constant of water at "2 K kg mol"^(-1). The figures shown below represent plots of vapour pressure (V.P.) versus temperature (T) [Molecular weight of ethanol is "46 g mol"^(-1)]. Among in the freezing point is |
|
Answer»
`DeltaT_(F)=K_(f)xxm` `Delta_(f)^(@)-T_(f)=2xx(34.5//46)/(0.5)` `273-T_(f)=3 or T_(f)=270`. Hence, only option (a) is correct. |
|
| 17. |
Pure water freezes at 273 K and 1 bar. The addition of 34.5 g of ethanol to 500 g of water changes the freezing point of the solution. Use the freezing point depression constant of water as 2 K kg mol^(-1).The figures shown below represent plots of vapour pressure (V.P.) versus temperature (T). [molecular weight of ethanol is 46 g mol^(-1) Among the following, the option representing change in the freezing point is |
|
Answer»
`m=34.5/46xx1000/500`  `DeltaT_(f)=34.5/46xx1000/500xx2=3` `T_(f)^(@)-T_(f)=273-T_(f)=3` `T_(f)=273-3=270K` |
|
| 18. |
Pure water does notconduct electricity because it is |
|
Answer» BASIC |
|
| 19. |
Pure water dissociates to a small extent as per equilibrium 2H_2O_((i)) hArr H_3O_((eq))^(+) +OH_((eq))^(-) The pH of pure water at 298 K is 7, what will be pH of pure water at 310 K? |
| Answer» Answer :B | |
| 20. |
Pure water does not conduct electricity because it |
|
Answer» |
|
| 21. |
Pure water does not conduct electricity because it is : |
|
Answer» NEUTRAL liquid |
|
| 22. |
Pure sulphur was burnt. the gaseous products are SO_(2)=60% (mol), SO_(3)=20% (mol) and O_(2)=20% (mol). If initially 50 moles of sulphur was taken then how many moles of O_(2) should be taken. |
|
Answer» 110 `n_(a)=50=xxx0.6+x xx0.2-0.8x` `x=(50)/(0.8)=(500)/(8)=62.5` `n_(o_(2))` remain=62.5xx0.2-12.5 `n_(o)=2xxn_(SO_(2))+3xxn_(SO_(3))=2xx x xx0.6+3xx x xx0.2` `n_(o)=1.2x+0.6x=1.8x=1.8xx62.5` `n_(o_(2))=(1.8xx65)/(2)=0.9xx65=56.25` `n_(o_(2))` TOTAL require `=56.25+12.5=68.75` |
|
| 23. |
Pure solvent A has freezing point 16.5^(@)C. On dissolving 0.4 g of B in 200 g of A, the solution freezing at 16.4^(@)C and on dissolving 2.24 g of C in 100 g of A, the solution has freezing point of 16.0^(@)C. If the molar mass of Bis "74 g mol"^(-1), what is the molar mass of C? |
|
Answer» `DELTA T_(F)=(1000K_(f)w_(2))/(w_(1)M_(2)),"i.e., 0.1"=(1000xxK_(f)xx0.4)/(200xx74)or K_(f)="3.7 K kg MOL"^(-1)` For solute C dissolved in the same solvent A, `M_(2)=(1000K_(f)w_(2))/(w_(1)DeltaT_(f))=(1000xx3.7xx2.24)/(100xx0.5)=165.8` Alternatively,`""((DeltaT_(f))_(B))/((DeltaT_(f))_(C))=(w_(B))/(w_(A)M_(B))xx(w_(A)'xxM_(C))/(w_(C))` |
|
| 24. |
Pure silicon is an insulator. Silicon doped with phosphorus is a semi-conductor. Silicon doped with gallium is also a serni-conductor. What is the difference between two doped silicon semi- conductors ? |
|
Answer» Solution :Pure silicon has a network or lattice in which all the four valence electrons are bonded to four other atoms. Therefore, it is an insulator. However, when silicon is doped with phosphorus (`NS^(2) NP^(3)`) having five valence electrons, the impurity leads to an EXCESS of electron after forming four covalent bonds like silicon. The extra electrons SERVE to conductivity and silicon doped with phosphorus behaves as semi-conductor. When silicon is doped with gallium (`ns^(2) np^(1)`) having three valence electrons, the bonds formed create electron deficient sites called holes. Under the influence of applied electric field, one electron from neighbouring atoms moves to fill hole but creates another hole at its own place. Therefore, the electrical conductance is due to the movement of positive hole. The basic DIFFERENCE between the two doped semi- conductors is that phosphorus doped silicon semi- conductor is n-type semi-conductor while gallium doped silicon-conductor is p-type. In n-type, the conduction is due to movement of electrons while in p-type, the conduction is due to movement of positive hole. |
|
| 25. |
Pure (R) Mandelic acid has specific rotation of 150. Ifa sample contains 60% of the R and 40% of its enantiomer, the [alpha] of his solution is. |
|
Answer» |
|
| 26. |
Pure silicon is: |
|
Answer» Insulator |
|
| 27. |
Pure primary amines underline("can be prepared ")by the action of ammonia on alkyl halides. |
| Answer» SOLUTION : PURE primary amines cannot be PREPARED by the ACTION of ammonia on alkyl halides. | |
| 29. |
Pure ozone is ………………. . |
| Answer» Solution :Pale blue gas | |
| 30. |
Pure phosphoric acid is very viscous, because : |
|
Answer» It is a strong acid |
|
| 31. |
Pure phosphine is not combustible while impure phosphine is combustible, this combustibility is due to presence of: |
|
Answer» `P_2H_4` |
|
| 32. |
Pure ozone is …………… |
| Answer» Solution :Pale blue gas | |
| 33. |
Pure nitric acid is colourless, on standing it becomes yellow. Justify your answer. |
|
Answer» Solution :Nitric ACID DECOMPOSES on exposure to sunlight or on being heated, into nitrogen dioxide, water and oxygen. `4HNO_(3)rarr4NO_(2)+2H_(2)O+O_(2)` Due to this reaction pure acid or its CONCENTRATED solution becomes yellow on standing. |
|
| 34. |
Pure NaCl is prepared by saturating a cold saturated solution of common salt in water with HCl gas. The principle used is |
|
Answer» Le-Chatelier principle |
|
| 35. |
Pure O_(2) instead of air is used to oxidise the pig iron because: |
|
Answer» MOLTEN metal took up small amount of nitrogen which makes the steel brittle |
|
| 36. |
Pure nitrogen gas is obtained from |
|
Answer» `NH_(3)+NaNO_(2)` |
|
| 37. |
Pure N_(2) gas is obtained from |
|
Answer» `NH_(3)+NaNO_(2)` `NH_(4)NO_(2) overset("HEAT")tounderset("(g)")(N_(2))+underset("(l)")(2H_(2)O)` |
|
| 38. |
Pure m-bromotoluene can be obtained by |
|
Answer» Bromination of toluene 
|
|
| 40. |
Extra pure N_2 can be obtained by heating |
|
Answer» `NH_(4)NO_(3)` |
|
| 42. |
Pure HF does not attack: |
|
Answer» Glass |
|
| 43. |
Pure HBr gas may be obtained by heating sodium bromide with syrupy phospheric acid and not with concentrated sulphuric acid because concentrated sulphuric acid is: |
| Answer» Answer :D | |
| 44. |
Pure H_2S gas can be obtained by the action of water on : |
|
Answer» CuS |
|
| 45. |
Pure conc. HNO_(3) makes iron passive as the surface is covered with protective layer of |
|
Answer» `Fe_(2)O_(3)` |
|
| 46. |
Pure CO can be obtained from : |
|
Answer» SODIUM oxalate |
|
| 47. |
Pure H_2 is obtained by the action of: |
|
Answer» Alluminium over KOH |
|
| 48. |
Pure Cl_(2) is prepared by |
|
Answer» `PtCl_(4)` `PtCl_(4) overset( 374^(@))(rarr) PtCl_(2) + Cl_(2) + CuCl_(2) + H_(2) O overset( 582^(@))(rarr) Pt+Cl_(2)` |
|
| 49. |
Pure chloroform may be prepared by : |
|
Answer» chlorination of methane |
|
| 50. |
pure benzene freezes at 5.3^(@)C. A solution of 0.233 g of phenyl acetic acid (C_(6)H_(5)CH_(2)COOH) freezes at 4.47^(@)C. Form this observation, one can conclude that pbenyl acetic acid |
|
Answer» exists as such in benzene `"Molaliy (m)"=((0.223 g)//(0.0044 kg))/((136 g MOL^(-1)))` `=0.373 mol//kg` `DeltaT_(f)=iK_(f)m` `i=(DeltaT_(f))/(K_(f)m)=((0.83K))/((5.12 K mol^(-1))xx(0.373 mol kg^(-1)))` =0.435. Since I isclose to 0.5, this means that the acid dimerises in benzene. |
|