Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Pyrolusite is the main ore of manganese in which it is present asIts Mn content is determined by reducing it under acidic condition to Mn^(2+)with the help of oxalate (C_(2)O_(4)^(2-)) ion which in turn gets oxidized to CO_(2) The analytical determination is carried out by adding a known excess volume of (C_(2)O_(4)^(2-)) solution to a suspension of the pyrolusite and digesting the mixture on a hot water bath until all the MnO_(2) has been reduced. The excess unreacted oxalate solution is then titrated with standardized KMnO_(4) solution. Thereby Mn content of ore can be calculated. KMnO_(4)solution is also standardized under acidic condition against oxalate ion wherein MnO_(4)^(-) ion is reduced to Mn^(2+) and (C_(2)O_(4)^(2-)) ion is oxidized to CO_(2)Q If a student prepared a standard solution of Na_(2)C_(2)O_(4) by dissolving 3.2 g of dry anhydrous salt into distilled water and making the solution upto 500 mL. The Normality of oxalate solution is |
|
Answer» 0.085 `THEREFORE E_(Na_(2)C_(2)O_(4))=(134)/(2)=67g`m Normality=`(3.2/67)/(500)xx1000`=0.096N |
|
| 2. |
Pyroligneous acid (one of the fraction of destructive distillation of wood), does not contain |
|
Answer» STEARIC acid |
|
| 3. |
Pyroligeneous acid obtained during destructive distillation of wood contains : |
|
Answer» ACETONE and methyl ALCOHOL |
|
| 4. |
pyroligneous acid contains: |
|
Answer» `2%` ACETIC ACID |
|
| 5. |
Pyroligneous acid cntains : |
|
Answer» `CH_3COOH(10%),CH_3OH(2.5%)CH_3COCH_3(0.5%)` |
|
| 6. |
Pyridinium chlorochromate (PCC) is a selective oxidising agent to oxidise a 1^@ alcohol to corresponding aldehyde. |
|
Answer» |
|
| 7. |
Pyridoxine is _____. |
|
Answer» VITAMIN `B_(6)` |
|
| 8. |
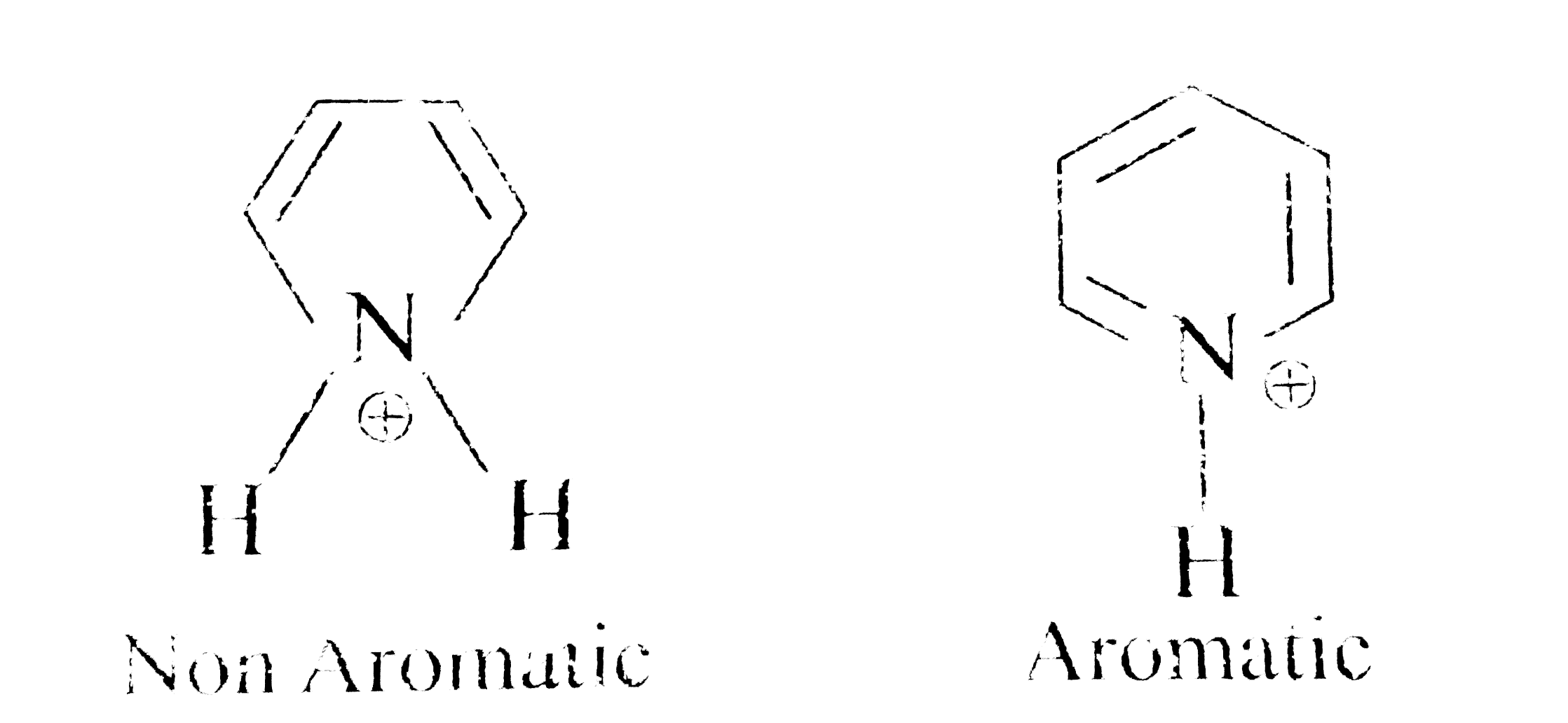
Pyridine like benzene, has six pi-electrons in delocalized pi - orbitals, but unlike benzene the orbitals will be deformed are more attractedtowardsthe nitrogen atom because of its high elecrtonegativity. This is reflected in the dipole of pyridine which has the negative end on Nand the positive end on the nucleus. Pyridine is thus reffered to as a pi - deficient hetrocycle. By analogy with a benzene ring that carries an electron-withdrawing substituent, NO_(2) one would expect it to be deactivated towards electrophilic attack with the help of pi delocalized molecular orbital. We can compare the respective properties of benzene, pyridine, pyrrole, furan and thiophene, which very frequently give aromatic electrophilic and nucleophilic substitution reactions in different positions. The simplest of six membered heterocyclic nitrogen compound is |
|
Answer» pyrrole |
|
| 9. |
Pyridine is mixed with aqueous solution of NiSO_(4), NaNO_(2) solution is then added to mixture to get dark blue precipitate 'A'. Analyse show that A does not contain any SO_(4)^(2-) ion but produce three ions per molecule A is |
|
Answer» `[Ni(Py)_(2)[NO_(2))_(2)]` |
|
| 10. |
Pyridine is more basic than pyrrole. Which of these of following statememt explain this fact? |
|
Answer» In pyrrole lone pair is involved in armaticity, in pyridine lone pair is not involved in aromaticity it is FREE for donation |
|
| 11. |
Pyrexglassis obainedbyfusingtogether_____. |
|
Answer» 60 to 80%`Al_(2)O_(3)` 10to25%`SiO_(2)` andramininf amountof `B_(2)O_(3)` |
|
| 12. |
Pyridine is less basic than triethylamine because |
|
Answer» Pyridine has aromatic character Pyridine is less basic than triethylamine because lone pair of ELECTRON in nitrogen in pyridine is delocalised. |
|
| 13. |
'Pyrene' is the trade name of ....... which is used as fire extinguisher |
|
Answer» `CO_2` |
|
| 14. |
Pyrene is the trade name of …….. when used as fire extinguisher |
|
Answer» `C Cl_(4)` |
|
| 16. |
p_(y)-orbital can not form pi -bond by lateral overlap with : |
|
Answer» <P>`d_(XZ)`- ORBITAL |
|
| 17. |
Pyranose structure of glucose is |
|
Answer» HEXAGONAL |
|
| 18. |
PVC, polythene, LDPE, cellulose, HDPE, bakelite |
| Answer» SOLUTION :Cellulose. It is a NATURAL POLYMER WHEREAS others are synthetic polymers. | |
| 19. |
PVC is produced by the polymerisation of : |
|
Answer» VINYL acetate |
|
| 20. |
_______ is used to make non-stick cookware. |
|
Answer» PVC |
|
| 21. |
PVC is used for |
|
Answer» Manufactured of cosmetics |
|
| 22. |
PVC is prepared by the polymerisation of |
|
Answer» ETHYLENE `UNDERSET("1-chloroethene")(nCH_(2)=CH.Cl)overset("Polymerisation")tounderset(PVC)([-CH_(2)-overset(Cl)overset(|)(CH)-])_(n)` |
|
| 23. |
PVC is of which type polymer ? |
| Answer» Solution :Branch | |
| 24. |
PVC is obtained by polymerization of |
|
Answer» `CH_2=CH_2` |
|
| 25. |
Which is naturally occurring polymer? |
| Answer» SOLUTION :PROTEIN is a NATURALLY OCCURING POLYMER. | |
| 26. |
PVC is a polymer of _____. |
|
Answer» ethylene |
|
| 28. |
PVC is a polymer of: |
|
Answer» `CH_2=CH_2` |
|
| 29. |
PVC, bakelite and polythene are plastics Name the monometer units of PVC and bakelite . |
|
Answer» Solution :Monomers of PVC: vinyl chloride, `CH_2=CH-Cl` MONOMER of bakelite:PHENOL `(C_6H_5OH) and Formaldehyde (HCHO) |
|
| 30. |
PVC, bakelite and polythene are plastics. Classify the above plastics into thermoplastics and thermosetting plastics. |
| Answer» SOLUTION :Thermoplastics-PVC, POLYTHENE. Thermostting PLASTICS- BAKELITE | |
| 31. |
PVC. |
| Answer» SOLUTION :It is used in the MANUFACTURE of SHEETS, waterpipes hand BAGS, ETC. | |
| 32. |
Purpose of hydrogen -oxygen fuel cell is to |
|
Answer» GENERATE heat |
|
| 33. |
Purk alkyl bromide is formed from bromination of which alkane ? |
|
Answer» iso-butane |
|
| 36. |
Purity of butter is determined in terms of: |
|
Answer» SAPONIFICATION VALUE |
|
| 37. |
Purity of ether before using it as anaesthetic agent is tested by : |
| Answer» Answer :A | |
| 39. |
Purification or Refining of metals : Metals obtained by reduction processes still contain some objectionable impurities and have to be refined Numerous techniques are available , including the following : Physical methods:These methods include the following processes : (I) Liquation process (II)Fractional distillation process (III)Zone refining method(IV)Chromatographic methods Chemicalmethods : These methods include the following methods : (I)Oxidative refining(II)Poling process (III)Electrolytic refining(IV)Vapour phase refining Now answer the following questions . Select the correct statements : |
|
Answer» Q,r,s (s)Cupellation involves removal of Pb impurity from Ag metal. |
|
| 40. |
Purine derivative among the following bases is |
|
Answer» cytosine |
|
| 41. |
Purification or Refining of metals : Metals obtained by reduction processes still contain some objectionable impurities and have to be refined Numerous techniques are available , including the following : Physical methods:These methods include the following processes : (I) Liquation process(II)Fractional distillation process (III)Zone refining method(IV)Chromatographic methods Chemicalmethods : These methods include the following methods : (I)Oxidative refining (II)Poling process (III)Electrolytic refining(IV)Vapour phase refining Now answer the following questions . Select the INCORRECT statement : |
|
Answer» Poling process used in purification of COPPER & tin involves reduction of impurities present |
|
| 42. |
Purine and pyrimidine bases present in both DNA and RNA are |
|
Answer» URACIL |
|
| 43. |
Purification of Nickel by ............... Process involves formation .................. Which yields 99.5% pure Nickel on decomposition. |
|
Answer» |
|
| 44. |
Purification of silicon element used in semiconductors is done by : |
| Answer» Answer :A | |
| 45. |
Purification of colloids is done by: |
|
Answer» HEATING |
|
| 46. |
Purification of blood is carried out by.... |
|
Answer» coagulation |
|
| 47. |
Purification of bone black is carried out by |
|
Answer» `CH_(3)COOH` |
|
| 48. |
Purification of aluminium done by electrolytic refining is known as |
|
Answer» Serpeck's PROCESS |
|
| 49. |
Purification of alumina by electrorefining is known as : |
|
Answer» HALL's PROCESS |
|