Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Pure benzene freezes at 5.3^(@)C. A solution of 0.223 of phenylacetic acid (C_(6)H_(5)CH_(2)COOH) in 4.4 g of benzene (K_(f) = 5.12 Kg mol^(-1)) freezes at 4.47^(@)C. From this observation, one can conclude that |
|
Answer» Phenylacetic ACID exists as such in benzene molality of solvent `m = (0.223)/(136)xx(1000)/(4.4)=0.373` (Molecular weight of phenylacetic acid = 136) `K_(f)=5.12` `Delta T_(f) =i. K_(f)m` `i=(Delta T_(f))/(K_(f)m)=(0.83)/(5.12xx0.373)=0.45` i is NEARLY equal to 0.5 it MEANS phenylacetic acid undergoes dimerization in benzene. |
|
| 2. |
Pure benzene freezes at 5.3^(@)C. A solution of 0.223 g of phenylacetic acid (C_(6)H_(5)CH_(2)COOH) in 4.4 g of benzene (K_(f)="5.12 K kg mol"^(-1)) freezes at 4.47^(@)C. From this observation, one can conclude that |
|
Answer» phenylacetic acid exists as such in benzene `=(1000xx5.12xx0.223)/((5.3-4.47)xx4.4)=312.6` Calculated molecular mass of `C_(6)H_(5)CH_(2)COOH` `=72+5+12+2+12+32+1=136` As observed molecular mass is nearly double of the theoretical value, it dimerizes in benzene. |
|
| 3. |
Pure aniline is a |
|
Answer» Colourless SOLID |
|
| 4. |
Pure aniline is a : |
|
Answer» colourlesssolid |
|
| 5. |
Pure ammonia is placed in a vessel at temperature where its dissociation constant (alpha) is appreciable. At equlibrium |
|
Answer» `K_(p)` does not CHANGE significantly with pressure |
|
| 6. |
Pure and dry Air is passed over soda lime and potash solution and then through a long tube containing red hot copper. The remaining air is passed over heated magnesium ribbon Which of the following are removed when air is passed through soda lime and potash |
|
Answer» `O_2` `3Mg+ N_2 to Mg_3 N_2` for COMPLETE REMOVAL of `O_2 and N_2` the AIR obtaned after removal of `CO_2 and H_2O` is repeatedly passed over heated Cu and Mg. `C + O_2 to CO_2` |
|
| 7. |
Pure ammonia is placed in a vessel at a temperature where its dissociation appreciable. At equilibrium |
|
Answer» `K_p` does not CHANGE significantly with pressure |
|
| 8. |
Punderset(2.H_(3)O^(+))overset(1.CH_(3)MgBr)toR underset(2.Delta)overset(1.dil.NaOH)to4-methylpent-3-en-2-one P is |
|
Answer» PROPANONE |
|
| 9. |
Pure (+) 2- chlorobutane gives how many different dichlorbutanes? |
Answer» 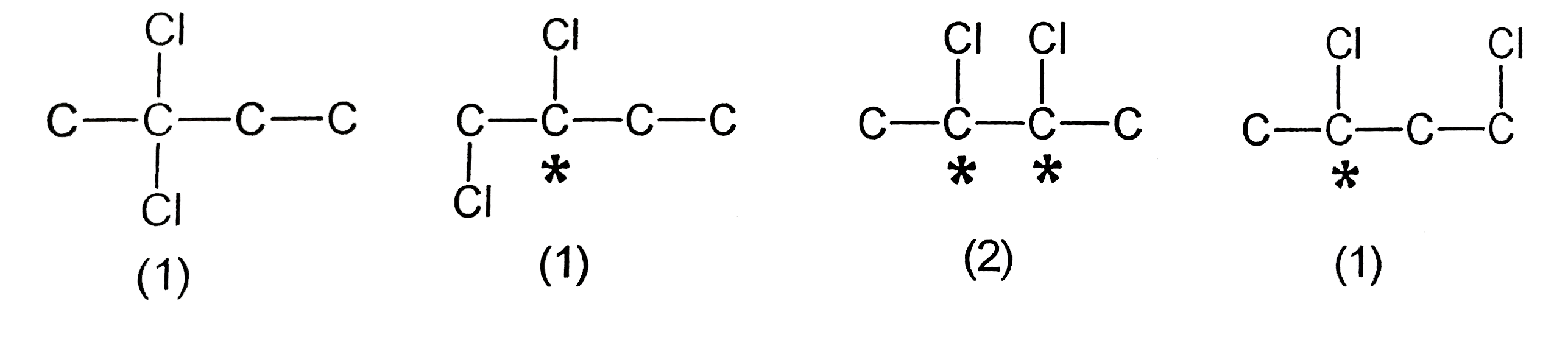
|
|
| 10. |
Pure alky chloride can be obtaned from alkanol by the eaction of |
|
Answer» `PCl_(5)` |
|
| 11. |
Pure acetylene has sweet ethereal smell while impure smells like garlic due to presence of: |
|
Answer» `NH_3` |
|
| 12. |
Pudding process is used in the manufacture of: |
|
Answer» Steel |
|
| 13. |
[Pt(Ox)(py)_(2)(O_(2))(H_(2)O)] Select correct statement about this complex : |
|
Answer» Oxidation state of `O_(2)` is `-1` |
|
| 14. |
[Pt(NH_(3))(NH_(2)OH)(NO_(2))(py)]^(+) will form how many geometrical isomers ? |
|
Answer» 2 |
|
| 15. |
[Pt("trien")]^(2+) is a ……. Ligand on the basis of its denticity. |
| Answer» SOLUTION :tetradentate | |
| 16. |
[Pt(NH_(3))_(4)][PtCl_(4)] is called as …………….. |
|
Answer» ZEIGLER NATTA Catalyst |
|
| 18. |
Pt|H_(2)(1 atm )|H^(+)(0.001M)||H^(+)(0.1M)|H_(2)(1atm)|Pt what will be the value of E_(cell) for this cell |
|
Answer» 0.1182V |
|
| 19. |
Pt(H_(2),"x atm")abs(0.0MH^(+))abs(0.1MH^(+))Pt(H_(2),"y atm"). ifE^(o) "" (cell) = 0.00V,then x/y is : |
|
Answer» 100 |
|
| 20. |
Pthalic acid overset(Delta)to underset(x)("product") . No of sp^2 carbons in 'x' are |
Answer» 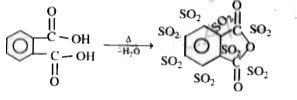
|
|
| 21. |
Pt, O_(2)(1atm.) OH^(-)_((aq)) 2OH_((aq))^(-) hArr H_(2)O_((l))+?+2e^(-) |
|
Answer» `1//2 O_(2)` |
|
| 22. |
Pt|Cl_(2)(P_(1)atm)|HCl(0.1M)|Cl_(2)(P_(2)atm)|Pt, cell reaction will be spontaneous if |
|
Answer» `p_(1)=p_(2)` if `P_(1)ltP_(2),E_(cell)=+ve` (SPONTANEOUS) |
|
| 23. |
[Pt (NH_(3))(NH_(2)OH) (NO_(2))(Py)]^(+) will form how many geometrical isomers :- |
|
Answer» 2 |
|
| 24. |
[PtBrCl(NO_(2))(NH_(3))] I on ionisation gives the ion |
|
Answer» `CL^(-)`<BR>`Br^(-)` |
|
| 26. |
Psycotherapeutic drugs are also called |
|
Answer» antacidis |
|
| 27. |
Pseudonitrol is formedfromnitrousacidand what ? |
|
Answer» `CH_(3) - CH_(2) - CH_(2) - NO_(2)` |
|
| 28. |
Pseudo halides are anions having resemblance with halide ions. Group I metals can form salts with pseudo halides. Pseudo halogens can acts as ligands and form coordinate complexes. Their hydrides are weakly acidic and can be prepared in analogous way as halogen hydrides are prepared. Azides, cyanides, selonocyanides are example of pseuda halides. When sodium pseudo halides are dissolved in water, it resembles with: |
| Answer» Answer :C | |
| 29. |
Pseudo first order rate for the reaction, A+B toProduct, when studied in 0.1M of B is given by -(d[A])/(dt)=k[A], where , k=1.85xx10^(4)sec^(-1), calculate the value of second orderrate constant. |
|
Answer» Solution :`A+B to `Product `-(d[A])/(DT)=K[A]` `-(d[A])/(dt)=1.85xx10^(4)XX[A]`…………………..`(i)` Assuming the reaction to be of second ORDER `-(d[A])/(dt)=k.[A][B]` `-(d[A])/(dt)=k.[A][0.1]` …………..`(ii)` Dividing Eq. `(i)` by `(ii)`, we get `1=(1.85xx10^(4))/(k.xx(0.1))` `:.k.=1.85xx10^(5)Lmol^(-1)s^(-1)` |
|
| 30. |
Prussionblue is formedwhen : |
|
Answer» Ferroussulphatereacts with `FeCI_(3)` |
|
| 31. |
Prussian'sblueisformed when Fe^(+2) ions are added to K_(4)[Fe(CN)_(6)]_(2) Turnbull'sblue is |
|
Answer» `Fe_(4)[Fe(CN)_(6)]_(3)` |
|
| 32. |
Prussian blue is obtained by mixing together aqueous solution of Fe^(3+) salt with |
|
Answer» Ferricyanide |
|
| 33. |
Prussian blue is a complex with the formula "…...............". |
|
Answer» |
|
| 34. |
Prussian blue is formed when |
|
Answer» Ferrous sulphate REACTS with `FeCl_(3) ` |
|
| 35. |
Prussian blue is |
|
Answer» `K_(3)[Fe(CN)_(6)]` |
|
| 36. |
Prussian blue is formed when : |
| Answer» Answer :C | |
| 37. |
Prussian blue is : |
|
Answer» `K_3[FE(CN)_6]` |
|
| 38. |
Provided detailed curved arrow mechanisms for the following reactions: |
Answer» Solution : In the fourth MECHANISM step, the nitrogen atom is protonated in preference to the oxygen atoms because nitrogen electronegative, and THUS more readily WILLING to share its LONE pair. |
|
| 39. |
Provided detailed curved arrow mechanisms for the following reactions: |
Answer» SOLUTION : Fragmentation of the tetrahedral INTERMEDIATE with an amide anion leaving group (in this case RNH) is the obvious RATE determining step, because an amide anion is a POOR leaving group (An amide anion is a poorer leaving group than a hydroxide ion.) One could combine this step with the nitrogen anion protonation to avoid having an amide anion leaving group. This is reasonable, as the solvent is WATER, and the amine will he hydrogen bonded before it departs. However, in this reaction, the amide anion leaving group problem cannot be solved by protenation with `H_(3) O` because `H_(3)O.` is simply not present. The reaction medium is strongly basic, so any strong acid such as hydronium ion will be immediately neutralized. |
|
| 40. |
Provided detailed curved arrow mechanisms for the following reactions: |
Answer» SOLUTION :
|
|
| 41. |
Provided detailed curved arrow mechanisms for the following reactions: |
Answer» Solution :Two reasonable mechanisms can be written for this reaction: a traditional FISCHER esterification or a carbonyl carbon cation capture mechanism. Both are ACCEPTABLE, although the traditional Fischer esterification mechanism is probably the minor pathway as an alcohol is more basic than a carboxylic acid. Fischer esterification mechanism is as follows:  Mechanism involving carbocation capture of carbonyl  The carbocation captures the carbonyl oxygen of the OC-CH, group INSTEAD of the OH oxygen, because it LEADS to the more stable oxonium ion intermediate. The resulting oxonium ion enjoys RESONANCE stabilization, whereas the oxonium ion formed by OH capture does not. This carbocation-carbonyl capture mechanism for acid-catalyzed esterification of a carboxylic acid can only be a possibility when the carbocation derived from the alcohol is sufficiently stable (secondary or better). |
|
| 42. |
Provided detailed curved arrow mechanisms for the following reactions: |
Answer» SOLUTION :
|
|
| 43. |
Provided detailed curved arrow mechanisms for the following reactions: |
Answer» SOLUTION : The last TWO stpes of this mechanism show loss of water in an `E_(1)` fashion, An `E_(2)`- type of DEHYDRATION is also ACCEPTABLE. As explaianed below.  |
|
| 44. |
Provide the structure of the major organic product which results in the following reaction. |
|
Answer» |
|
| 45. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 46. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 47. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 48. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 49. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|
| 50. |
Provide the organic product(s) of the reaction. Pay careful attention to stereochemistry. If more than one product is formed, indicate which product (if any) is the major product. If no reaction occurs, write "NR" |
Answer» SOLUTION :
|
|