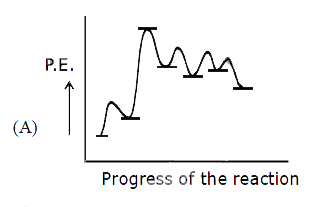Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Ph -underset(darr Cao//Delta)(CO_(2))H + CH_(3) CO_(2)H |
|
Answer» `PH-overset(O)overset(||)(C)-PH` |
|
| 2. |
Ph-underset(14)overset(O)overset(||)(C)-CHN_2-underset(H_2O)overset(Ag_2O)(rarr)(P). Product (P) is |
|
Answer» `Ph-overset(14)(C)H_2-CO_2H` |
|
| 3. |
pH scale was introduced by |
|
Answer» Arrhenius |
|
| 4. |
pH of water is 7. When a substance Y is dissolved in water, the pH becomes 13. The substance Y is a salt by |
|
Answer» STRONG acid and strong BASE |
|
| 5. |
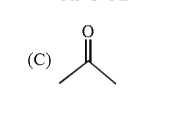
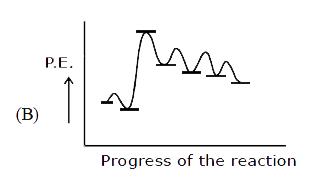
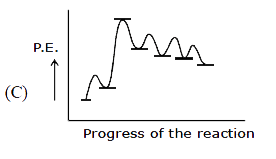
Ph-overset(Ph)overset(|)underset(H)underset(|)C-overset(H)overset(|)underset(CH_3)underset(|)C-CH_2-OH overset(H^+)underset(Delta)to Product Select the correct P.E. graph for major product. |
|
Answer»
|
|
| 6. |
Ph-overset(O)overset("||")C-Phoverset(PhCO_(3)H)rarrAoverset(LiAlH_(4))rarrB+C B and C are respectively : |
|
Answer» `Ph-CH_(2)-OH` |
|
| 7. |
Ph-overset(O)overset(||)C-O-CPh_(3)-overset(H_(3)overset(18)O^(o+))rarrProduct of this reaction is : |
|
Answer» `Ph-OVERSET(O)overset(||)C-OH+Ph_(3)C-overset(18)OH` |
|
| 8. |
Ph-overset(O)overset(||)(C)-CH_(3)+underset("(excess)")(CH_(2)=O) overset(OH^(Theta))toPh-overset(O)overset(||)(C)-C(CH_(2)OH)_(3) In the above reaction how times of aldol reaction takes place ? |
Answer» 
|
|
| 9. |
Ph-overset(O)overset(||) underset(14)(C)-OH overset(NaHCO_(3)//Delta)to(A) |
|
Answer» `CO_(2)` |
|
| 10. |
pH of which solution is not affected by dilution |
|
Answer» ` 0.01 M CH_3 COONH_4` |
|
| 11. |
pH of which solution is not affected by dilution ? |
|
Answer» 0.01 M `CH_(3) COONH_(4)` |
|
| 12. |
pH of water is 7.0 at 25^(@)C. If water is heated to 80^(@)C |
|
Answer» pH will increase |
|
| 13. |
pH of two solutions : I. 50 mL of 0.2 MHCl + 50 mL of 0.2 MHA (K_(a)=1.0xx10^(-5)) and II. 50 mL of 0.2 M HCl +50 mL of 0.2 M NaA will be respectively |
|
Answer» 0.70 and 2.85 In PRESENCE of STRONG acid HCl, the weak acid HA remains PRACTICALLY unionized. Hence `[H^+]=[H^(+)]_(HCl)=0.1M,pH=1` (II) `NaA+HCl""toNaCl+HA` `[HA]=(50xx0.2)/(100)=0.1M` No HCl is left. Hence `[H^(+)]` `=sqrt(K_(a)C)=sqrt(1.0xx10^(-5)xx0.1)=1.0xx10^(-3),pH=3` |
|
| 14. |
pH of the solution in the anode compartment of the following cell at 25^@C is x when Eced E_("cell")-E_("cell")^(@)=0.0591V. underset("1 atm")(PT (H_(2))|pH=Xx|| Ni^(2+)(M)|Ni Find the value of x. |
|
Answer» `K=([H^(+)]^(2))/([Ni^(2+)])=[H^(+)]^(2)` `E_("CELL")-E_("cell")^(@)=0.0591 XX pH` pH=1 |
|
| 15. |
pH of soluiotn obtained by mixing 50ml of 0.1M Na_(2)CO_(3) solution and 50 ml of 0.05 M HCl solution is where K_(a_(1)) and K_(a_(2)) are first and second dissociation constants of H_(2)CO_(3) respectively. |
|
Answer» `( pKa_(a_(1))+pK_(a_(2)))/(2)` |
|
| 16. |
pH of saturated solution of Ca(OH)_2 is 9. The solubility product (K_(sp)) of Ca(OH)_2 |
|
Answer» `0.5xx10^(-15)` `Ca(OH)_(2)hArr Ca^(2+)+2OH^(-)` Given that `pH=9` `pOH=14-9=5` `[pOH=-log_(10)[OH^(-)]]` `:.[OH^(-)]=10^(-pOH)` `[OH^(-)]=10^(-5)M` `K_(SP)=[Ca^(2+)][OH^(-)]^(2)` `=(10^(-5))/(2)XX(10^(-5))^(2)=0.5xx10^(-15)` |
|
| 18. |
pH of NaCl solution is |
|
Answer» 7 |
|
| 19. |
pH of human blood is 7.4. Then H^(+) concentration will be |
|
Answer» `4 XX 10^(-8)` |
|
| 20. |
pH of HCl(10^(-12)M) is |
|
Answer» 12 |
|
| 21. |
pH of completely dissociated 0.005 M H_(2)SO_(4) is |
|
Answer» 3 |
|
| 22. |
pH of buffer depends upon concentration of |
| Answer» Answer :D | |
| 23. |
pH of blood is maintained constant by mechanisms of |
|
Answer» COMMON ION effect |
|
| 24. |
pH of aqucous solution of 0.1M, NH_(4),CI is found to be 5. The equilibrium constant for the neutralization of NH_(4),OH by HCl is 10^(y). The value of 'y' is |
|
Answer» |
|
| 26. |
pH of a solution produced when an aqueous solution of pH 6 is mixed with an equal volume of an aqueous solution of pH 3 is about |
|
Answer» 3.3 |
|
| 27. |
pH of a solution of 10 ml. 1 N sodium acetate and 50 ml 2N acetic acid (K_(a) = 1.8 xx 10^(-5)), is approximately |
|
Answer» 4 `pH = - log (1.8 xx 10^(-5)) + log.([10])/([100])` `= -log 1.8 + 5 + log 10^(-1)` `= 0.2553 + 5 - 1 = 3.7447` or = 4. |
|
| 28. |
pH of a solution of 0.1 M [CH_3COONH_4(aq)] is [given: K_a(CH_3COOH) = K_b(NH_4OH) = 1.8 x 10^-5)] |
|
Answer» 1 |
|
| 29. |
pH of a solution is 9.5. The solution is |
|
Answer» Neutral |
|
| 30. |
pH of a solution is 5.5 at 25^(@)C. Calculate its [OH^(-)] |
|
Answer» SOLUTION :`pH+POH=14.0` `THEREFORE pOH=14.0-pH` `=14.0 -5.5 =8.50` `pOH=8.5""therefore "ANTILOG" [-pOH]` `therefore [OH^-]= "antilog" [-8.5]=3.2xx10^-9 M`. |
|
| 31. |
pH of a solution is 5. Thus. the concentration of hydroxyl ion in the solution is |
|
Answer» `9 MOL L^-1` |
|
| 32. |
pH of a solution is 5. Its hydroxyl ion concentration is……. |
|
Answer» 5 `pOH=14-pH=14-5=9` `[OH^-]=10^(-pOH)=10^-9` |
|
| 33. |
pH of a solution having 0.00001 gm ions of ions of hydrogen per litre is |
|
Answer» SOLUTION :`[H^(+)] = 1 XX 10^(-5) M` `pH = - log 10^(-5)` pH = 5 log 10 = 5. |
|
| 34. |
pH of a saturated solution of Ba(OH)_(2) is 12. The value of solubility product (K_(sp)) of Ba(OH)_(2) is |
|
Answer» `3.3 xx 10^(-7)` `[OH^(-)] = 10^(-2)` `2s = 10^(-2)` `s = (10^(-2))/(2) K_(sp) = 4s^(3) = 4 xx ((10^(-2))/(2))^(3) = 5 xx 10^(-7)` |
|
| 35. |
pH of a solution can be expressed as |
|
Answer» `-log_(e)(H^(+))` |
|
| 36. |
pH of a saturated solution of Ca(OH)_2 is 9. The solubility product (K_(sp)) of Ca(OH)_2…….. |
|
Answer» `0.5 times 10^-15` Given that pH=9 `POH=14-9=5` `[pOH=-log_10[OH^-]]` `therefore[OH^-]=10^(-pOH)` `[OH^-]=10^-5 M` `K_(SP)=[Ca^(2+)][OH^-]^2` `=10^-5/2 times (10^-5)^2=0.5 times 10^-15` |
|
| 37. |
PH value of a saturated solution Ba(OH)_2 is 12. Calculate solubility product K_sp for Ba (OH)_2(3+2 |
|
Answer» `3.3 xx 10^(-7)` |
|
| 38. |
pH of a saturated solution of Al(OH)_3, is 9. The value of solubility product (K_sp) of Al(OH)_3 is |
|
Answer» `1/2*1^-20` |
|
| 39. |
pH of a neutral solution is equal to 7. Prove it. |
|
Answer» Solution :(i) In the neutral solution the CONCENTRATIONS of `[H_3O^+]` as well as `[OH-]` are EQUAL to `1 TIMES 10^-7M` at `25^@C` (ii) The pH of a neutral solution can be calculated by substituting this `[H_3O^+]` concentration in the expression. `pH=-log_10[H_3O^+]` `=-log_10[1 times 10^-7]` `=-(-7)log""10/10=+7(1)=7` (III) pH=7 for a neutral solution. |
|
| 40. |
pH of a lemon water bottle is 3 and that of orange juice is 4. Calculate the pH of final solution obtained by mixing above solutions in volume ratio of 1 : 2 respectively. (Assuming that no change in volume on mixing and no chemical reaction is taking place) [Given : log_(10)2 =0.30] |
|
Answer» `[H^(+)] = (10^(-3) + 2 xx 10^(-4))/(3)= 4 xx 10^(-4)` `PH= 4- LOG 4= 4- 6= 3.40` |
|
| 41. |
pH of a human blood is 7.4. Then H^+ concentration will be…… |
|
Answer» `4 times 10^-8` `7.4=-log[H^+]` `7.4= log1-log[H^+]` `log[H^+]=log 1-7.4` `log[H^+]=8.6` `THEREFORE [H^+]=ANTILOG of 8.6` `=4 times 10^-8` |
|
| 42. |
pH of a buffer solution decreases by 0.02 units when 0.12 g of acetic acid is added to 250 mL of a buffer solution of acetic and potassium acetate at 27^(@)C. The buffer capacity of the solution is |
|
Answer» 0.1 |
|
| 43. |
pH of a buffer solution decreases by 0.02 units when 0.12g of acetic acid is added to 250 mL of a buffer solution of acetic acid and potassium acetate at 27^(@)C. The buffer capacity of the solution is |
|
Answer» `0.1` |
|
| 44. |
pH of 10^(-8) M HNO_(3) is (nearly) |
|
Answer» 7 `:. [H^(+)] = 10^(-8) m` but pH = 8 is not POSSIBLE because it is acid. Now `[H^(+)] = 10^(-7) M` are already present in solution and SINCE `10^(-8) lt 10^(-7)` and thus, it should not be NEGLECTED. `:. [H^(+)] = 10^(-8) + 10^(-7) = 10^(-7)(1.1) M` `= 1.1 xx 10^(-7)M` `:. pH = 6.9586`. |
|
| 45. |
pH of 1M HA (weak acid) is 2. Hence van't Hoff factor is - |
|
Answer» 1.2 |
|
| 46. |
pH of a 0.1 M monobasic acid is found to be 2 .Hence , its osmotic pressure at a given temperature T K is : |
|
Answer» 0.1 RT |
|
| 47. |
pH of a 0.001 M NaOH solution will be |
|
Answer» 9 |
|
| 49. |
pH of 0.1M solution of NaA (sodium salt of weak acid HA) is 8.92.Calculate pK_(a) of HA.If a drop HPh(pK_("in")=9.52) be added to the above solution.predict whether the pink colour will visible or not the under the medical fact that our eyes can see the pink colour if the mole % of ionised form of indicated is 25% or more. |
|
Answer» Solution :`pH=(1)/(2)(pK_(a)+pK_(w)+logc)` `8.92=7+(1)/(2)pK_(a)+(1)/(2)(-1)` `(1)/(2)pK_(a)=8.92-6.5=2.42` `pK_(a)` of `HA=4.84` `:.pK_(a)=pK_("in")+LOG""([LN^(-)])/([HLN])` `=8.92=9.52+log""([ln^(-)])/([Hln])` `=8.92=9.52+log"([ln^(-)])/([Hln])` `([ln^(-)])/([Hln])=(ionised)/(unionised)=(1)/(4)` Since ionised from `%=(1)/(1+4)xx100=20%` PINK colour will not be visible. |
|