Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
pH of 0.5 M aqueous NaCN solution is (pK_a of HCN = 9.3, log 5 = 0.7) |
|
Answer» 10.3 |
|
| 2. |
pH of 0.1 N NaOH(aq) solution |
|
Answer» 2.7 |
|
| 3. |
pH of 0.1 M NH_(3) aqueous solution is(K_(b) = 1.8 xx 10^(-5)) |
|
Answer» 11.13 `K_(b) = C alpha^(2) , (1.8 xx 10^(-5))/(.1) = alpha^(2), alpha = 1.34 xx 10^(-3)` `[OH^(-)] = alpha. C = 1.34 xx 10^(-3) xx .1` `POH = LOG 10(1)/(1.34 xx 10^(-4)) , pOH = 2.87` `pH = pOH = 14 , pH + 2.87 = 14` `pH = 14 - 2.87, pH = 11.13`. |
|
| 5. |
pH of 0.01 M (NH_4)_2 SO_4 and 0.02 M NH_4OH buffer (PK_a of NH_(4)^(+)= 9.26) is |
|
Answer» `4.74 + log2` ` PH =pK_a+log(["Base"])/(["salt"])` `=9.26 + log(0.02 )/( 2 xx 0.001 )= 9.26+Log1` |
|
| 7. |
ph -NO_2overset((I ))toPh NH_2underset( NaNO_2 //HCI ) overset( 0 ^@C )IIunderset( H_2 O)overset(Delta )III I ,II ,III are resoectively , |
|
Answer» `Sn//HCI , PhN_(2) ^(+)Cl^(-), C_6H_5OH` |
|
| 8. |
pH of 0.0002 M formic acid [K_(a) = 2xx 10^(-4)] approximatly is |
|
Answer» 1.35 |
|
| 9. |
Ph-NH_(2)underset(0^(@)C)overset(HNO_(2))to A underset(BF_(3))overset(HF)toB overset(Delta)to C, C is |
|
Answer» `Ph-overset(+)(N)-=NBF_(4)^(-)` |
|
| 10. |
Ph - NH_(2) overset(NaNO_(2)//HCl) underset(0-5^(@) C) to overset(Ph-NH_(2))underset(dil.HCl) to (X) Find the sum of number of nitrogen atoms present in (X) and the total number of stereoisomers of (X) formed. |
Answer»  No. of N-atoms in X is 3: No. of stereo isomers = 2 (syn/anti). |
|
| 11. |
Ph-N=C=Ounderset((ii)H_2O)overset((i)CH_3MgBr)toPoverset(LiAH_4)toQ |
|
Answer» <P>P is `Ph-NH-UNDERSET(O)underset(||)C-CH_3` and Q is `Ph-NH-CH_2-CH_3` 
|
|
| 12. |
pH in stomacth is approximately |
|
Answer» 7 |
|
| 13. |
pH for the solution of salt undergoing anionic hydrolysis (say CH_3COONa) is given by: |
|
Answer» PH= `1/2 [PK_w + PK_a + LOGC]` |
|
| 14. |
Ph-CH=CH-underset(O)underset(C)-H overset((i) CH_(3)CH_(2)MgBr)to overset((ii) H_(3)O^(o+))to(X) overset(Cu,Delta)to(Y) |
|
Answer» X is 1,4-ADDITION product , Y is `Ph-UNDERSET(O)underset(||)(C)-CH=CH-CH_(2)-CH_(3)` |
|
| 15. |
Ph-CH=CH-CH_(2)-COOHunderset((2)Br_(2)"CC"l_(4).Delta)overset((1)AgOH)to Product can be : |
|
Answer» `Ph-CH=CH-CH_(2)-BR` |
|
| 16. |
Ph-CH_2-oversetoverset(CH_3)(|)CH-CH_3underset(("monochlorination"))overset(Cl//hv)to Which statements is/are correct about photochemical chlorination of the above compound ? |
|
Answer» The major product will be chiral carbon atom having OPTICALLY inactive compound |
|
| 17. |
Ph-CH_(2)-overset(CH_(2))overset(|)underset((+))CH-OHoverset(TaCl)rarrXoverset(KSH)rarrY: |
|
Answer» `X " is "Ph-underset((+))CH_(2)-overset(CH_(3))overset(|)CH-OTs` |
|
| 18. |
Ph-CH_(2)-O-overset(O)overset("||")C-CH_(3)+overset(Θ18)(OH)rarrX+Y: |
|
Answer» `X " is"Ph-CH_(2)-overset(18)(OH)` |
|
| 19. |
Ph-CH_(2)-Cloverset(Mg)underset(Ether)rarroverset(CO_(2))underset(H^(oplus)//HOH)rarroverset(SOCl_(2))rarroverset(NH_(3))rarroverset(LiAlH_(4))rarr |
|
Answer» |
|
| 20. |
Ph-CH_2-C-=CHunderset(Y)overset(X)hArrPh-C-=C-CH_3. The reagents X and Y respectively are |
|
Answer» Lindlar CATALYST , `NaNH_2` |
|
| 21. |
pH calculation upon dilute of strong acid solution is generally done by equating n_(H) in original solution & diluted solution.However . If strong acid solution is very dilute then H^(+) from water are also to be considered take log3.7=0.568 and answer the following questions. A 1 litres solution of pH=4(solution ofa strong acid ) is added to the 7//3 litres of water.What is the pH of resulting solution? |
|
Answer» `4.52` `[H^(+)]=10^(-4)` `N_(1)V_(1)=N_(2)V_(2)rArr 10^(-4)=N_(2)xx[1+(7)/(3)]` `10^(-4)=N_(2)xx(10)/(3)rArr N_(2)=3xx10^(-5)"" gt10^(-6)` so `[H^(+)]` of WATER is not consider `[H^(+)]=3xx10^(-5)`"" so""`pH=5-log(3)`""`=5-0.48=4.52` |
|
| 22. |
Ph-C-=C-Ph overset(Na//NH_3)toAoverset(HC Cl//H^+)toB |
|
Answer»
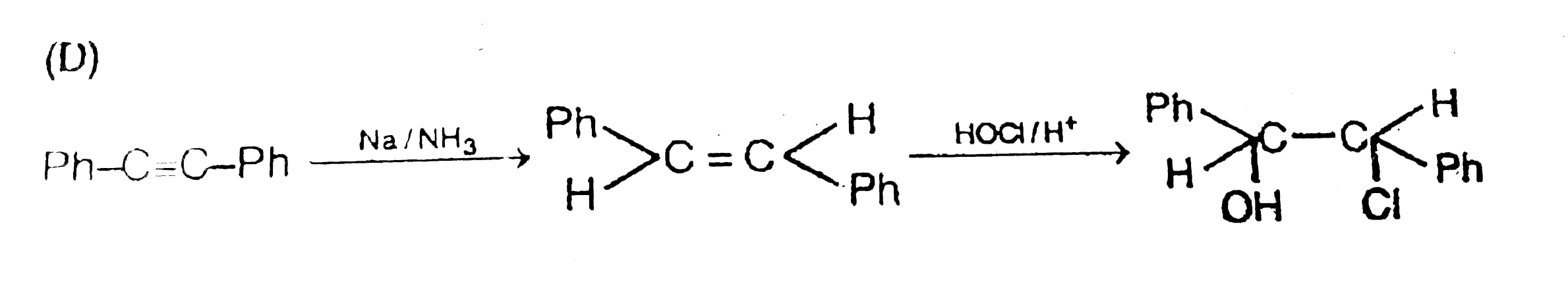
|
|
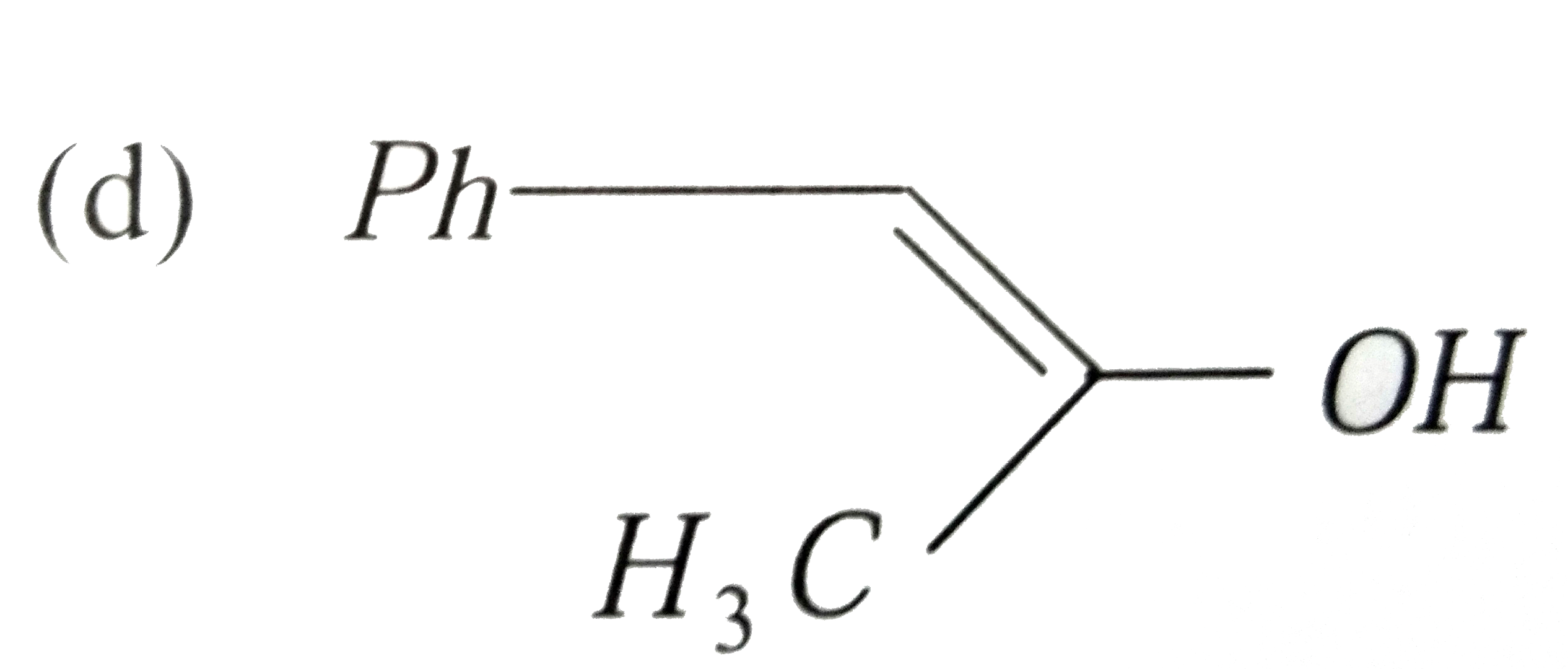
| 23. |
Ph-C-=C-CH_(3) overset(Hg^(2+)//H^(+))toA A is |
|
Answer»

|
|
| 24. |
Ph-C-=C-CH_3overset(Hg^(2+)//H^+)to A . A is |
|
Answer»
|
|
| 25. |
Ph-C -= C-CH_(3) overset(Hg^(2+)//H^(+))rarr A,A is |
|
Answer»

|
|
| 26. |
PF_(5) is known but NF_(5) is not. Explain. |
| Answer» SOLUTION :Nitrogen cannot extend its valency from 3 to 5 DUE to absence of d-orbitals while PHOSPHORUS SHOWS penta covalency as.d-orbitals are present in it. | |
| 27. |
PF_(3) reacts with XeF_(4) to give PF_(5) underset((g))(2PF_(3))tounderset((s))(XeF_(4))tounderset((g))(2PF_(5))+underset((g))(Xe) If 100.0gm of PF_(3) and 50.0 gm XeF_(4) react, then which of the following statement is true? |
|
Answer» `XeF_(4)` is the limiting reagent |
|
| 28. |
PF_3 molecule is: |
|
Answer» SQUARE PLANAR |
|
| 30. |
Petroleum refining involves: |
|
Answer» VACUUM DISTILLATION |
|
| 31. |
Petroleum is mainly a mixture of |
|
Answer» Alkanes |
|
| 32. |
Petroleumisobtainedformwatergasname of thereactioninvolved is : |
| Answer» Answer :A | |
| 33. |
Petroleum refining and naturalgas plant are the main sources of |
|
Answer» `H_(2) S` |
|
| 34. |
Petroleumis amixtureof : |
|
Answer» ALKANES |
|
| 35. |
Pertrolis amixturehydrocabonsfromC_(6)"to "C_(5) thequalityofpetrolis determindedin termsofoctanenumber,Thehigher the octanenumber betteris thequalityof fuel . Thecorrectorderof octane numberis : |
|
Answer» cycloalknes LT ALKANES ltalkanes lt AROMATIC hydrocarbons |
|
| 36. |
Petroleum ether can be used as |
|
Answer» Solvent for fat, OIL, VARNISH and RUBBER |
|
| 37. |
Petroleum consists mainly of |
|
Answer» ALIPHATIC hydrocarbons |
|
| 38. |
Peroxy links are present in H_(2)SO_(5) and H_(2)S_(2)O_(8). How manynumber of peroxy bonds are presentin each acid? |
|
Answer» |
|
| 39. |
Persons working in cement plants and lime stone quarries are prone to disease like : |
|
Answer» Cancer |
|
| 40. |
Peroxy linkage is present in: |
|
Answer» Caro.s acid |
|
| 41. |
Perptization is a process of |
|
Answer» reducing the IMPURITIES of the electrolytes |
|
| 42. |
Peroxydisulphuric acid has the following bond |
|
Answer» `O LARR O =O`  Thus, peroxydisulphuric acid contains PEROXIDE (-O-O-) bond. |
|
| 43. |
Peroxy linkage is present in |
|
Answer» `H_(2)S_(2)O_(2)` |
|
| 44. |
Peroxoacids of sulphur are: |
|
Answer» `H_(2)SO_(4)` |
|
| 45. |
Peroxodisulphuric acid has the following bond |
|
Answer» `O LARR O = O` |
|
| 46. |
Peroxide plays a vital role in producing |
|
Answer» carbocation |
|
| 48. |
Peroxide ion....... (i) Has five completely filled antibonding molecular orbitals (ii) Is diamagnetic (iii) Has bond order one (iv) Is isoelectric with neon Which one of these is correct |
|
Answer» (iv) and (iii) Peroxide ion is `O_(2)^(2-)` TOTAL electrons=18 E.c.=`sigma1s^(2)sigma^(**)1S^(2)sigma2s^(2)sigma^(**)2S^(2)sigma(2p_(X))^(2)pi (2p_(y))^(2)` `pi^(**)(2p_(x))^(2)pi^(**)(2p_(y))^2` `B.O. =(10-8)/2=1` options (ii) and (iii) are correct. |
|
| 49. |
Peroxide ion (i) has five completely filled antibonding molecular orbitals (ii) is diamagnetic (iii) has bond order one (iv) is isoelectronic with neon. Which one of these is correct ? |
|
Answer» (iv) and (iii) Total electrons `=18` `E.C.=sigma1s^(2)SIGMA^(**)1s^(2)SIGMA2S^(2)sigma^(**)2s^(2)sigma(p_(z))^(2)pi(2pi_(X))^(2)pi(2p_(y))^(2)pi^(**)(2p_(x))^(2)pi^(**)(2p_(y))^(2)` `B.O.=(10-8)/(2)=1` Options (ii) and (iii) are correct. |
|
| 50. |
Peroxide effeis observed |
|
Answer» only with HBr |
|