Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The process of getting freshwater from sea water is known as: |
|
Answer» Osmosis |
|
| 2. |
The process of froth floatation and chromatography are based on : |
|
Answer» Emulaification |
|
| 3. |
The process of formation of RNA from DNA is known as : |
|
Answer» Translation |
|
| 4. |
The process of extraction of sodium on a commercial scale by the electrolysis of fused sodium chloride is called : |
|
Answer» |
|
| 5. |
The process of extracting the metal from its ore is called : |
|
Answer» REFINING |
|
| 6. |
The process of evaporation of a liquid is accompanied by: |
|
Answer» INCREASE in enthalpy |
|
| 7. |
The process of eudiometry is used to calculate the volume of the various reacting gases present together in a closed vessel, it is done by sparking the content and noting the volume change or pressure change depending upon the conditions mentioned. Various reagents like alkaline pyrogallol, turpentine oil, caustic potash, conc. H_(2)SO_(4) are used to absorb theselective gases like O_(2), O_(3), CO_(2) and H_(2)O (v) respectively. A 2.0 mole mixture of H_(2)(g),O_(2)(g) and He(g) are placed together in a closed container at pressure equal to 50 atm. An electric spark is passed and pressure is noted as 12.5 atm after cooling the content to original temp. (room temp). Oxygen gas is introduced for pressure to change to 25 atm, keeping volume and temp constant. Again electrical spark is passed and pressure drops to 10 atm under original temp. conditions. The mole fraction of H_(2)(g) present in the initial mixture is: |
|
Answer» 0.05 `therefore x+y+z=20` Pressure exerted by 2 moles of gaseous MIXTURE is 50 atm. After the first electric spark decrease in pressure =50-12.5=37.5 atm `therefore` decrease in no. of moles of gaseous mixture is 1.5 But from the given information limiting reagent in first step is `O_(2)` `therefore" from "H_(2)(g)+(1)/(2)O_(2)(g) RARR H_(2)O(l)` `therefore H_(2) and O_(2)` REACTED are in the ratio of 2:1 `therefore` 0.5 mole of `O_(2)` should be reacted with 1.0 mole of `H_(2)` `therefore` No. of mole of `O_(2)` in the mixture (y) =0.5 When again `O_(2)` is passed, pressure increased from 12.5 to 25.0 atm `therefore` Change in pressure =12.5 atm `therefore` No. of moles of `O_(2)` added `=(12.5xx2)/(50)=0.5` mole Now `H_(2)` will be completely reacted No. of moles `H_(2)` actually left after first electric spark =x-1 `H_(2)+(1)/(2)O_(2) rarr H_(2)O` `(x-1)((x-1)/(2))` Now change in pressure in 25-10 =15 atm `therefore` change in no. of moles is `(15xx2)/(50)=0.6` `therefore (x-1)+((x-1)/(2))=0.6` `rArr x=1.4` `therefore` no of moles `H_(2)=1.4` `therefore` no. of moles of `He=2.0-(1.4+0.5)=0.1` `therefore` mole fraction of `H_(2)=(1.4)/(2.0)=0.7` |
|
| 8. |
The process of eudiometry is used to calculate the volume of the various reacting gases present together in a closed vessel, it is done by sparking the content and noting the volume change or pressure change depending upon the conditions mentioned. Various reagents like alkaline pyrogallol, turpentine oil, caustic potash, conc. H_(2)SO_(4) are used to absorb theselective gases like O_(2), O_(3), CO_(2) and H_(2)O (v) respectively. A 2.0 mole mixture of H_(2)(g),O_(2)(g) and He(g) are placed together in a closed container at pressure equal to 50 atm. An electric spark is passed and pressure is noted as 12.5 atm after cooling the content to original temp. (room temp). Oxygen gas is introduced for pressure to change to 25 atm, keeping volume and temp constant. Again electrical spark is passed and pressure drops to 10 atm under original temp. conditions. The moles of O_(2)(g) introduced in the vessel after first electrical spark is : |
|
Answer» 0.25 `therefore x+y+z=20` Pressure exerted by 2 moles of gaseous mixture is 50 atm. After the first electric spark DECREASE in pressure =50-12.5=37.5 atm `therefore` decrease in no. of moles of gaseous mixture is 1.5 But from the given information limiting reagent in first STEP is `O_(2)` `therefore" from "H_(2)(G)+(1)/(2)O_(2)(g) rarr H_(2)O(l)` `therefore H_(2) and O_(2)` reacted are in the ratio of 2:1 `therefore` 0.5 MOLE of `O_(2)` should be reacted with 1.0 mole of `H_(2)` `therefore` No. of mole of `O_(2)` in the mixture (y) =0.5 When again `O_(2)` is passed, pressure increased from 12.5 to 25.0 atm `therefore` Change in pressure =12.5 atm `therefore` No. of moles of `O_(2)` added `=(12.5xx2)/(50)=0.5` mole Now `H_(2)` will be completely reacted No. of moles `H_(2)` actually left after first electric spark =x-1 `H_(2)+(1)/(2)O_(2) rarr H_(2)O` `(x-1)((x-1)/(2))` Now change in pressure in 25-10 =15 atm `therefore` change in no. of moles is `(15xx2)/(50)=0.6` `therefore (x-1)+((x-1)/(2))=0.6` `rArr x=1.4` `therefore` no of moles `H_(2)=1.4` `therefore` no. of moles of `He=2.0-(1.4+0.5)=0.1` `therefore` mole fraction of `H_(2)=(1.4)/(2.0)=0.7` |
|
| 9. |
The process of eudiometry is used to calculate the volume of the various reacting gases present together in a closed vessel, it is done by sparking the content and noting the volume change or pressure change depending upon the conditions mentioned. Various reagents like alkaline pyrogallol, turpentine oil, caustic potash, conc. H_(2)SO_(4) are used to absorb theselective gases like O_(2), O_(3), CO_(2) and H_(2)O (v) respectively. A 2.0 mole mixture of H_(2)(g),O_(2)(g) and He(g) are placed together in a closed container at pressure equal to 50 atm. An electric spark is passed and pressure is noted as 12.5 atm after cooling the content to original temp. (room temp). Oxygen gas is introduced for pressure to change to 25 atm, keeping volume and temp constant. Again electrical spark is passed and pressure drops to 10 atm under original temp. conditions. The moles of He (g) present in the mixture samples is |
|
Answer» 0.2 `therefore x+y+z=20` Pressure EXERTED by 2 moles of gaseous MIXTURE is 50 atm. After the FIRST electric spark decrease in pressure =50-12.5=37.5 atm `therefore` decrease in no. of moles of gaseous mixture is 1.5 But from the given information limiting reagent in first step is `O_(2)` `therefore" from "H_(2)(g)+(1)/(2)O_(2)(g) rarr H_(2)O(l)` `therefore H_(2) and O_(2)` reacted are in the ratio of 2:1 `therefore` 0.5 MOLE of `O_(2)` should be reacted with 1.0 mole of `H_(2)` `therefore` No. of mole of `O_(2)` in the mixture (y) =0.5 When again `O_(2)` is passed, pressure increased from 12.5 to 25.0 atm `therefore` Change in pressure =12.5 atm `therefore` No. of moles of `O_(2)` added `=(12.5xx2)/(50)=0.5` mole Now `H_(2)` will be completely reacted No. of moles `H_(2)` actually left after first electric spark =x-1 `H_(2)+(1)/(2)O_(2) rarr H_(2)O` `(x-1)((x-1)/(2))` Now change in pressure in 25-10 =15 atm `therefore` change in no. of moles is `(15xx2)/(50)=0.6` `therefore (x-1)+((x-1)/(2))=0.6` `rArr x=1.4` `therefore` no of moles `H_(2)=1.4` `therefore` no. of moles of `He=2.0-(1.4+0.5)=0.1` `therefore` mole fraction of `H_(2)=(1.4)/(2.0)=0.7` |
|
| 10. |
The process of employed for the concentration of sulphide ore is |
|
Answer» Froth-floatation |
|
| 11. |
The process of covering iron sheet with a layer of zinc is called: |
|
Answer» Galvanizing |
|
| 12. |
The process of converting precipitate into colloidal solution on adding an electrolyte is called |
|
Answer» Peptisation 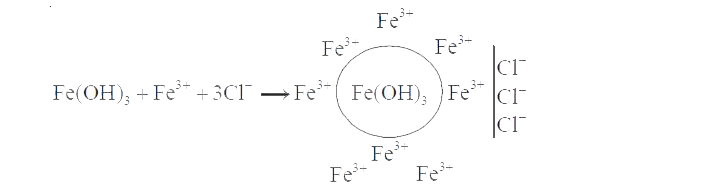 i.e., ions are adsorbed on particles to give colloidal SIZE. |
|
| 13. |
The process of converting hydrated alumina to anhydrous alumina is called |
|
Answer» Calcination |
|
| 14. |
The process of converting hydrated alumina into anhydrous alumina is called : |
|
Answer» Roasting |
|
| 15. |
The process of converting alkyl halides into alcohols involves _________reaction. |
|
Answer» addition |
|
| 16. |
The process of converting alkyl halides into alcohols involves_____ |
|
Answer» ADDITION reaction |
|
| 17. |
The process of converting alkyl halide into alcohols involves ......... |
|
Answer» ADDITION reaction |
|
| 18. |
The process of converign one enatiomer of an optically active compound inot racemic mixture is called: |
|
Answer» Resolution |
|
| 19. |
The process of conversion of colloidal solution into precipitate is called ............. |
| Answer» SOLUTION :COAGULATION | |
| 20. |
The process of conversion of colloidal solution into precipitate is known as ..... |
|
Answer» PEPTISATION |
|
| 21. |
The process ofconcentrating silver are is based on its solubility in : |
|
Answer» HCL |
|
| 23. |
The process of combining metals present in their native ores with mercury to form an alloy is called |
|
Answer» ALLOYING |
|
| 24. |
The process of calcination and roasting are carried out in: |
|
Answer» BLAST furnace |
|
| 25. |
The process of AgCN+KCNtoK[Ag(CN)_(2)] involves the oxidation of Ag. |
|
Answer» |
|
| 26. |
The process of bringing the metal or its ore into solution by the action of a suitable chemical reagnetfollowing by extraction of the metal either by electrolysis or by suitable precipitating agentis called |
|
Answer» Electrometallurgy |
|
| 27. |
The process of a protein, losing its higher order structure without losing the primary structure is called…………….. |
| Answer» SOLUTION :DENATURATION | |
| 28. |
The process involving reduction of metal oxide with coke or carbon monoxide is called……….. |
| Answer» SOLUTION :SMELTING | |
| 29. |
The process is spontaneous at the given temperature, if |
|
Answer» `Delta H" is " +ve and Delta S " is " -ve` When, `Delta H` is -ve, but `T Delta S` is `+ve rarr Delta G` will be (-ve) [Highly spontaneous]. |
|
| 30. |
The process involving transfer of five electrons is: |
|
Answer» `CrO_4^(2-) rarrCr^(3+) |
|
| 31. |
The process involving heating of rubber with sulphur is called |
|
Answer» galvanisation |
|
| 32. |
The process involving heating of natural rubber with sulphur is known as: |
| Answer» Answer :B | |
| 33. |
The process in which solution containing more than one typeof ions (either cation or anion) and in which one ion undergoes almst complete precipitation followed by the second ion and similarly second ion undergoes complete precipitation followed by the third ion is known as preferentialprecipitation. It is the solubility of the salt,not the solubility product of the salt, by means of which one can predict the preferential precipitation of the salt or ion among the lot of ions in the solution. For example , in a solution containing Cl^(-), Br^(-) and I^(-) ions, if Ag^(+) ions are added, then out of three , the less soluble salt is precipitated first . If the addition of Ag^(+) ions is continued, eventually , a stage is reached when the next lesser soluble salt starts precipitating along with the least soluble salt and so on. If the stiochiometry of the salt is same, then the salt with the minimum K_(sp) ( solubility product ) will have minimum solubility and will precipitate first, followed by the salt of next higher solubility product and so on. If the stiochiometry of teh salt is not same, then from the solubility product data alone, we cannot predict which ions will precipitate first. For example, a solution containing Cl^(-) and CrO_(4)^(2-). In order to predict which ion will precipitate first, we have to calculate the amount of Ag^(+) ions needed to start precipitation through the solubility productdata given. When AgNO_93) is added to the solution, the minimum of the two concentrations needed to start the precipitation will be reached and thus corresponding [Ag^(+)] to start the precipitation increases. Its concentration eventually becomes equal to the value required for CrO_(4)^(2-) . At this stage, practically almost the whole of Cl^(-) ions get precipitated . Addition of more AgNO_(3) causes simultaneous precipitation of both the ions together. Suppose a student of chemistry performs the following two sets of experiments in order to known the preferential precipitation of ions as well as its precipitated percentage amount. Experiment 1. He prepare a solution of anions composed of 0.1 M each of Cl^(-),Br^(-) and I^(-) . Further he adds gradually solid AgNO_(3) to this solution. He assumes thatvolume of the solution does not change after the addition of solid AgNO_(3)(K_(sp)AgCl=1.0 xx 10^(-9)M^(2),K_(sp) AgBr=10^(-10)M^(2) and K_(sp) AgI=10^(-12)M^(2)) Experiment -2. He prepares a solution of cations Cd^(2+)(0.2 M) and Bi^(3+) (0.3 m) . Now he adds S^(2-) ions into the solution of cations in order to separate them by selective precipitationCd^(2+) forms yellow precipitate of CdSand Bi^(3+) forms black precipitate of Bi_(2)S_(3) with S^(2-) ions respectively. (K_(sp)Bi_(2)S_(3) = 9 xx 10^(-25) M^(5) and K_(sp)CdS =2 xx 10^(-20)M^(2)).Answer the following questions ont he basis of the above write up. Which one of the following statements is correct regarding the experiment ? |
|
Answer» First of all YELLOW ppt. will appear |
|
| 34. |
The process in which solution containing more than one typeof ions (either cation or anion) and in which one ion undergoes almst complete precipitation followed by the second ion and similarly second ion undergoes complete precipitation followed by the third ion is known as preferentialprecipitation. It is the solubility of the salt,not the solubility product of the salt, by means of which one can predict the preferential precipitation of the salt or ion among the lot of ions in the solution. For example , in a solution containing Cl^(-), Br^(-) and I^(-) ions, if Ag^(+) ions are added, then out of three , the less soluble salt is precipitated first . If the addition of Ag^(+) ions is continued, eventually , a stage is reached when the next lesser soluble salt starts precipitating along with the least soluble salt and so on. If the stiochiometry of the salt is same, then the salt with the minimum K_(sp) ( solubility product ) will have minimum solubility and will precipitate first, followed by the salt of next higher solubility product and so on. If the stiochiometry of teh salt is not same, then from the solubility product data alone, we cannot predict which ions will precipitate first. For example, a solution containing Cl^(-) and CrO_(4)^(2-). In order to predict which ion will precipitate first, we have to calculate the amount of Ag^(+) ions needed to start precipitation through the solubility productdata given. When AgNO_93) is added to the solution, the minimum of the two concentrations needed to start the precipitation will be reached and thus corresponding [Ag^(+)] to start the precipitation increases. Its concentration eventually becomes equal to the value required for CrO_(4)^(2-) . At this stage, practically almost the whole of Cl^(-) ions get precipitated . Addition of more AgNO_(3) causes simultaneous precipitation of both the ions together. Suppose a student of chemistry performs the following two sets of experiments in order to known the preferential precipitation of ions as well as its precipitated percentage amount. Experiment 1. He prepare a solution of anions composed of 0.1 M each of Cl^(-),Br^(-) and I^(-) . Further he adds gradually solid AgNO_(3) to this solution. He assumes thatvolume of the solution does not change after the addition of solid AgNO_(3)(K_(sp)AgCl=1.0 xx 10^(-9)M^(2),K_(sp) AgBr=10^(-10)M^(2) and K_(sp) AgI=10^(-12)M^(2)) Experiment -2. He prepares a solution of cations Cd^(2+)(0.2 M) and Bi^(3+) (0.3 m) . Now he adds S^(2-) ions into the solution of cations in order to separate them by selective precipitationCd^(2+) forms yellow precipitate of CdSand Bi^(3+) forms black precipitate of Bi_(2)S_(3) with S^(2-) ions respectively. (K_(sp)Bi_(2)S_(3) = 9 xx 10^(-25) M^(5) and K_(sp)CdS =2 xx 10^(-20)M^(2)).Answer the following questions ont he basis of the above write up. In experiment -2what % of the metal ions get precipitated at which S^(2-) get saturated with another ion ? |
| Answer» ANSWER :A | |
| 35. |
The process in which solution containing more than one typeof ions (either cation or anion) and in which one ion undergoes almst complete precipitation followed by the second ion and similarly second ion undergoes complete precipitation followed by the third ion is known as preferentialprecipitation. It is the solubility of the salt,not the solubility product of the salt, by means of which one can predict the preferential precipitation of the salt or ion among the lot of ions in the solution. For example , in a solution containing Cl^(-), Br^(-) and I^(-) ions, if Ag^(+) ions are added, then out of three , the less soluble salt is precipitated first . If the addition of Ag^(+) ions is continued, eventually , a stage is reached when the next lesser soluble salt starts precipitating along with the least soluble salt and so on. If the stiochiometry of the salt is same, then the salt with the minimum K_(sp) ( solubility product ) will have minimum solubility and will precipitate first, followed by the salt of next higher solubility product and so on. If the stiochiometry of teh salt is not same, then from the solubility product data alone, we cannot predict which ions will precipitate first. For example, a solution containing Cl^(-) and CrO_(4)^(2-). In order to predict which ion will precipitate first, we have to calculate the amount of Ag^(+) ions needed to start precipitation through the solubility productdata given. When AgNO_93) is added to the solution, the minimum of the two concentrations needed to start the precipitation will be reached and thus corresponding [Ag^(+)] to start the precipitation increases. Its concentration eventually becomes equal to the value required for CrO_(4)^(2-) . At this stage, practically almost the whole of Cl^(-) ions get precipitated . Addition of more AgNO_(3) causes simultaneous precipitation of both the ions together. Suppose a student of chemistry performs the following two sets of experiments in order to known the preferential precipitation of ions as well as its precipitated percentage amount. Experiment 1. He prepare a solution of anions composed of 0.1 M each of Cl^(-),Br^(-) and I^(-) . Further he adds gradually solid AgNO_(3) to this solution. He assumes thatvolume of the solution does not change after the addition of solid AgNO_(3)(K_(sp)AgCl=1.0 xx 10^(-9)M^(2),K_(sp) AgBr=10^(-10)M^(2) and K_(sp) AgI=10^(-12)M^(2)) Experiment -2. He prepares a solution of cations Cd^(2+)(0.2 M) and Bi^(3+) (0.3 m) . Now he adds S^(2-) ions into the solution of cations in order to separate them by selective precipitationCd^(2+) forms yellow precipitate of CdSand Bi^(3+) forms black precipitate of Bi_(2)S_(3) with S^(2-) ions respectively. (K_(sp)Bi_(2)S_(3) = 9 xx 10^(-25) M^(5) and K_(sp)CdS =2 xx 10^(-20)M^(2)).Answer the following questions ont he basis of the above write up. In experiment 02, what is the maximum concentration of S^(2-)ion at which one of the two metal ions gets maximum precipitation ? |
| Answer» Answer :A | |
| 36. |
The process in which the concentrated ore is strongly heated in the absence of air is called as……… |
|
Answer» Roasting |
|
| 37. |
The process in which solution containing more than one typeof ions (either cation or anion) and in which one ion undergoes almst complete precipitation followed by the second ion and similarly second ion undergoes complete precipitation followed by the third ion is known as preferentialprecipitation. It is the solubility of the salt,not the solubility product of the salt, by means of which one can predict the preferential precipitation of the salt or ion among the lot of ions in the solution. For example , in a solution containing Cl^(-), Br^(-) and I^(-) ions, if Ag^(+) ions are added, then out of three , the less soluble salt is precipitated first . If the addition of Ag^(+) ions is continued, eventually , a stage is reached when the next lesser soluble salt starts precipitating along with the least soluble salt and so on. If the stiochiometry of the salt is same, then the salt with the minimum K_(sp) ( solubility product ) will have minimum solubility and will precipitate first, followed by the salt of next higher solubility product and so on. If the stiochiometry of teh salt is not same, then from the solubility product data alone, we cannot predict which ions will precipitate first. For example, a solution containing Cl^(-) and CrO_(4)^(2-). In order to predict which ion will precipitate first, we have to calculate the amount of Ag^(+) ions needed to start precipitation through the solubility productdata given. When AgNO_93) is added to the solution, the minimum of the two concentrations needed to start the precipitation will be reached and thus corresponding [Ag^(+)] to start the precipitation increases. Its concentration eventually becomes equal to the value required for CrO_(4)^(2-) . At this stage, practically almost the whole of Cl^(-) ions get precipitated . Addition of more AgNO_(3) causes simultaneous precipitation of both the ions together. Suppose a student of chemistry performs the following two sets of experiments in order to known the preferential precipitation of ions as well as its precipitated percentage amount. Experiment 1. He prepare a solution of anions composed of 0.1 M each of Cl^(-),Br^(-) and I^(-) . Further he adds gradually solid AgNO_(3) to this solution. He assumes thatvolume of the solution does not change after the addition of solid AgNO_(3)(K_(sp)AgCl=1.0 xx 10^(-9)M^(2),K_(sp) AgBr=10^(-10)M^(2) and K_(sp) AgI=10^(-12)M^(2)) Experiment -2. He prepares a solution of cations Cd^(2+)(0.2 M) and Bi^(3+) (0.3 m) . Now he adds S^(2-) ions into the solution of cations in order to separate them by selective precipitationCd^(2+) forms yellow precipitate of CdSand Bi^(3+) forms black precipitate of Bi_(2)S_(3) with S^(2-) ions respectively. (K_(sp)Bi_(2)S_(3) = 9 xx 10^(-25) M^(5) and K_(sp)CdS =2 xx 10^(-20)M^(2)).Answer the following questions ont he basis of the above write up. What %of the (ion) will get precipitate when the second ion start precipitating in experiment -1 ? |
| Answer» ANSWER :D | |
| 38. |
The process in which solution containing more than one typeof ions (either cation or anion) and in which one ion undergoes almst complete precipitation followed by the second ion and similarly second ion undergoes complete precipitation followed by the third ion is known as preferentialprecipitation. It is the solubility of the salt,not the solubility product of the salt, by means of which one can predict the preferential precipitation of the salt or ion among the lot of ions in the solution. For example , in a solution containing Cl^(-), Br^(-) and I^(-) ions, if Ag^(+) ions are added, then out of three , the less soluble salt is precipitated first . If the addition of Ag^(+) ions is continued, eventually , a stage is reached when the next lesser soluble salt starts precipitating along with the least soluble salt and so on. If the stiochiometry of the salt is same, then the salt with the minimum K_(sp) ( solubility product ) will have minimum solubility and will precipitate first, followed by the salt of next higher solubility product and so on. If the stiochiometry of teh salt is not same, then from the solubility product data alone, we cannot predict which ions will precipitate first. For example, a solution containing Cl^(-) and CrO_(4)^(2-). In order to predict which ion will precipitate first, we have to calculate the amount of Ag^(+) ions needed to start precipitation through the solubility productdata given. When AgNO_93) is added to the solution, the minimum of the two concentrations needed to start the precipitation will be reached and thus corresponding [Ag^(+)] to start the precipitation increases. Its concentration eventually becomes equal to the value required for CrO_(4)^(2-) . At this stage, practically almost the whole of Cl^(-) ions get precipitated . Addition of more AgNO_(3) causes simultaneous precipitation of both the ions together. Suppose a student of chemistry performs the following two sets of experiments in order to known the preferential precipitation of ions as well as its precipitated percentage amount. Experiment 1. He prepare a solution of anions composed of 0.1 M each of Cl^(-),Br^(-) and I^(-) . Further he adds gradually solid AgNO_(3) to this solution. He assumes thatvolume of the solution does not change after the addition of solid AgNO_(3)(K_(sp)AgCl=1.0 xx 10^(-9)M^(2),K_(sp) AgBr=10^(-10)M^(2) and K_(sp) AgI=10^(-12)M^(2)) Experiment -2. He prepares a solution of cations Cd^(2+)(0.2 M) and Bi^(3+) (0.3 m) . Now he adds S^(2-) ions into the solution of cations in order to separate them by selective precipitationCd^(2+) forms yellow precipitate of CdSand Bi^(3+) forms black precipitate of Bi_(2)S_(3) with S^(2-) ions respectively. (K_(sp)Bi_(2)S_(3) = 9 xx 10^(-25) M^(5) and K_(sp)CdS =2 xx 10^(-20)M^(2)).Answer the following questions ont he basis of the above write up. What percent of the anions Br^(-) and I^(-) get precipitated respectively when the third ion starts precipitating in experiment -1 ? |
|
Answer» `90%,99.9%` |
|
| 39. |
The process in which solution containing more than one typeof ions (either cation or anion) and in which one ion undergoes almst complete precipitation followed by the second ion and similarly second ion undergoes complete precipitation followed by the third ion is known as preferentialprecipitation. It is the solubility of the salt,not the solubility product of the salt, by means of which one can predict the preferential precipitation of the salt or ion among the lot of ions in the solution. For example , in a solution containing Cl^(-), Br^(-) and I^(-) ions, if Ag^(+) ions are added, then out of three , the less soluble salt is precipitated first . If the addition of Ag^(+) ions is continued, eventually , a stage is reached when the next lesser soluble salt starts precipitating along with the least soluble salt and so on. If the stiochiometry of the salt is same, then the salt with the minimum K_(sp) ( solubility product ) will have minimum solubility and will precipitate first, followed by the salt of next higher solubility product and so on. If the stiochiometry of teh salt is not same, then from the solubility product data alone, we cannot predict which ions will precipitate first. For example, a solution containing Cl^(-) and CrO_(4)^(2-). In order to predict which ion will precipitate first, we have to calculate the amount of Ag^(+) ions needed to start precipitation through the solubility productdata given. When AgNO_93) is added to the solution, the minimum of the two concentrations needed to start the precipitation will be reached and thus corresponding [Ag^(+)] to start the precipitation increases. Its concentration eventually becomes equal to the value required for CrO_(4)^(2-) . At this stage, practically almost the whole of Cl^(-) ions get precipitated . Addition of more AgNO_(3) causes simultaneous precipitation of both the ions together. Suppose a student of chemistry performs the following two sets of experiments in order to known the preferential precipitation of ions as well as its precipitated percentage amount. Experiment 1. He prepare a solution of anions composed of 0.1 M each of Cl^(-),Br^(-) and I^(-) . Further he adds gradually solid AgNO_(3) to this solution. He assumes thatvolume of the solution does not change after the addition of solid AgNO_(3)(K_(sp)AgCl=1.0 xx 10^(-9)M^(2),K_(sp) AgBr=10^(-10)M^(2) and K_(sp) AgI=10^(-12)M^(2)) Experiment -2. He prepares a solution of cations Cd^(2+)(0.2 M) and Bi^(3+) (0.3 m) . Now he adds S^(2-) ions into the solution of cations in order to separate them by selective precipitationCd^(2+) forms yellow precipitate of CdSand Bi^(3+) forms black precipitate of Bi_(2)S_(3) with S^(2-) ions respectively. (K_(sp)Bi_(2)S_(3) = 9 xx 10^(-25) M^(5) and K_(sp)CdS =2 xx 10^(-20)M^(2)).Answer the following questions ont he basis of the above write up. Which of the following will precipitate first in experiment -1 ? |
| Answer» Answer :A | |
| 40. |
The process in which solution containing more than one typeof ions (either cation or anion) and in which one ion undergoes almst complete precipitation followed by the second ion and similarly second ion undergoes complete precipitation followed by the third ion is known as preferentialprecipitation. It is the solubility of the salt,not the solubility product of the salt, by means of which one can predict the preferential precipitation of the salt or ion among the lot of ions in the solution. For example , in a solution containing Cl^(-), Br^(-) and I^(-) ions, if Ag^(+) ions are added, then out of three , the less soluble salt is precipitated first . If the addition of Ag^(+) ions is continued, eventually , a stage is reached when the next lesser soluble salt starts precipitating along with the least soluble salt and so on. If the stiochiometry of the salt is same, then the salt with the minimum K_(sp) ( solubility product ) will have minimum solubility and will precipitate first, followed by the salt of next higher solubility product and so on. If the stiochiometry of teh salt is not same, then from the solubility product data alone, we cannot predict which ions will precipitate first. For example, a solution containing Cl^(-) and CrO_(4)^(2-). In order to predict which ion will precipitate first, we have to calculate the amount of Ag^(+) ions needed to start precipitation through the solubility productdata given. When AgNO_93) is added to the solution, the minimum of the two concentrations needed to start the precipitation will be reached and thus corresponding [Ag^(+)] to start the precipitation increases. Its concentration eventually becomes equal to the value required for CrO_(4)^(2-) . At this stage, practically almost the whole of Cl^(-) ions get precipitated . Addition of more AgNO_(3) causes simultaneous precipitation of both the ions together. Suppose a student of chemistry performs the following two sets of experiments in order to known the preferential precipitation of ions as well as its precipitated percentage amount. Experiment 1. He prepare a solution of anions composed of 0.1 M each of Cl^(-),Br^(-) and I^(-) . Further he adds gradually solid AgNO_(3) to this solution. He assumes thatvolume of the solution does not change after the addition of solid AgNO_(3)(K_(sp)AgCl=1.0 xx 10^(-9)M^(2),K_(sp) AgBr=10^(-10)M^(2) and K_(sp) AgI=10^(-12)M^(2)) Experiment -2. He prepares a solution of cations Cd^(2+)(0.2 M) and Bi^(3+) (0.3 m) . Now he adds S^(2-) ions into the solution of cations in order to separate them by selective precipitationCd^(2+) forms yellow precipitate of CdSand Bi^(3+) forms black precipitate of Bi_(2)S_(3) with S^(2-) ions respectively. (K_(sp)Bi_(2)S_(3) = 9 xx 10^(-25) M^(5) and K_(sp)CdS =2 xx 10^(-20)M^(2)).Answer the following questions ont he basis of the above write up. Which of the following is the correctorder of appearance of coloour of the precipitate in experiment ? |
|
Answer» YELLOW `GT` PALE yellow `gt` CURDY white |
|
| 41. |
The process in which pressure remains constant throughout a change is: |
|
Answer» Adiabatic |
|
| 42. |
The process, in which no heat enters or leaves the system, is termed as |
|
Answer» Isochoric |
|
| 43. |
The process in which metalsurface is madeinactiveis called |
|
Answer» passivation |
|
| 44. |
The process in which metal surface is made inactive is called |
|
Answer» PASSIVATION |
|
| 45. |
The process in which higher hydrocarbons are broken down into lower hydrocarbons by controlled pyrolysis, is called |
|
Answer» Hydrolysis |
|
| 46. |
The process in which chemical change occurs on passing electricity is termed as __________. |
|
Answer» neutralisation |
|
| 47. |
The process in which chemical change occurs on passing electricity is termed: |
|
Answer» Ionisation |
|
| 48. |
The process in which chemical change accompanies the passage of current is called : |
|
Answer» CONDUCTION |
|
| 49. |
The process in which lighter earthy particles are freed fromthe heavier particle by washing with water is called : |
|
Answer» LEACHING |
|
| 50. |
The process (es) by which lighter earthy particles are freed from the heavier particles using water is / are : |
|
Answer» GRAVITY SEPARATION method |
|