Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 2. |
The number of vacant orbitals of element with atomic number 14 is: |
|
Answer» 2 |
|
| 3. |
The number of FIF angles which are less than 90^@ in IF_5 :- |
|
Answer» 6 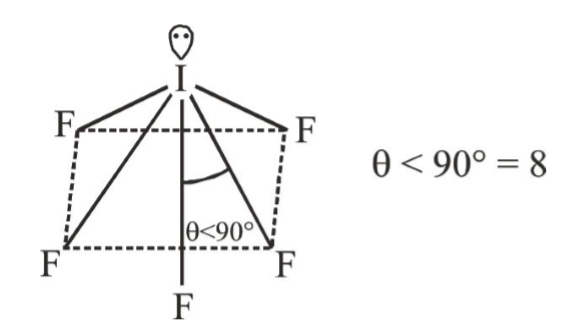
|
|
| 4. |
The number of Faradays needed to reduce 4 gram equivalents ofCu^(++)to Cu metal will be |
|
Answer» 1 4gm=4faraday. |
|
| 5. |
The number of vacant hybrid orbitals which participate in the formation of 3-centre 2 electrons bonds i.e., banana bonds in diborane structure is : |
Answer» 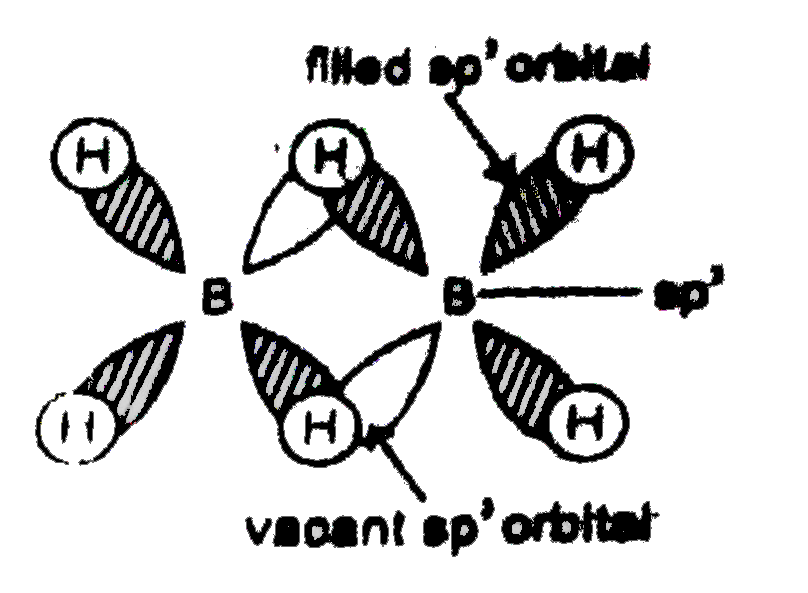 No of vacant `sp^3` hybrid ORBITALS participating in the formation of banana bonds is 2. |
|
| 6. |
The number of Faradays of electricity required to decompose 100 ml water (density = 0.99 gm/ml) is |
|
Answer» 2 |
|
| 7. |
The number of Faradays needed to reduce 4 gm. Equivalents of Cu^(2+) to Cu metal will be |
|
Answer» 1 |
|
| 8. |
The number of unpaired in the central metal ion of brown ring complex is |
|
Answer» |
|
| 9. |
The number of Faradays needed to reduce 4 gm equivalents of Cu^(2+) to Cu metal will be |
|
Answer» 1 |
|
| 10. |
The number of unpaired electrons present in Cr^(3+)is: |
|
Answer» 3 |
|
| 11. |
The number of Faradays needed to reduce 4 g equivalents ofCu^(2+)to Cu metal will be |
|
Answer» 1 |
|
| 12. |
The number of unpaired electrons present in complex ion [FeF_(6)]^(3-) is: |
|
Answer» 5 |
|
| 13. |
The number of faraday required to liberate 1 mole of any element indicates : |
|
Answer» WEIGHT ELEMENT |
|
| 14. |
The number of unpaired electrons present in Am are |
|
Answer» 2 |
|
| 15. |
Thenumber of Faraday required to gneerate 1g of Mg from MgCl_2 is: |
|
Answer» 1 |
|
| 17. |
The number of ether metamers represented by theformulaC_(4) H_(10)is - |
|
Answer» 4 |
|
| 18. |
The number of unpaired electrons ind^(6), low spin, octahedral complex is : |
|
Answer» 4 |
|
| 19. |
The number of ether metamers represented by molecular formula C_(4)H_(10)O is |
|
Answer» 4 (II) `CH_(3)OCH_(2)CH_(2)CH_(3)` (iv) `CH_(3)OCH(CH_(3))_(3)` |
|
| 20. |
The number of unpaired electrons in the square planar [Pt(CN)_(4)]^(2-) ion is |
|
Answer» Solution :In the given compled `[Pt(CN)_(4)]^(2-)` The electron configuration of Pt `Pt to [Xe]4f^(14)5D^(9)6s^(1)` `Pt^(2+)to[Xe]4f^(14)5d^(8)` 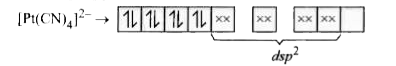 In has zero UNPAIRED electron as `CN^(-)` is a strong field ligand. |
|
| 21. |
The number of ether isomes for molecular formula C_(4)H_(10)O is ________ |
|
Answer» 2 |
|
| 22. |
The number of essential amino acids in man is |
|
Answer» 8 |
|
| 23. |
The number of unpaired electrons in the square planar complex [Pt(CN)_4]^(2-) is |
|
Answer» 2 |
|
| 24. |
The numberof essentialamino acidin man is …… . |
|
Answer» 8 |
|
| 25. |
The number of unpaired electrons in the brown ring complex is |
Answer» 
|
|
| 26. |
The number of unpaired electrons in [NiCl_(4)]^(2-), Ni(CO)_(4) and [Cu(NH_(3))_(4)]^(2+) respectively are |
|
Answer» 2,2,1 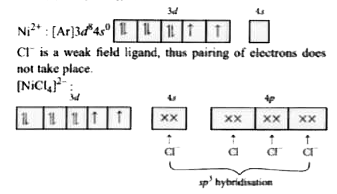 Unpaired electrons =2 In `Ni(CO)_(4),Ni` is in zero oxidation state. `Ni:(AR]3d^(8)4s^(2)`  Co is STRONG field ligand and causes pairing of electron of 4s to 3d. `Ni(CO)_(4)` 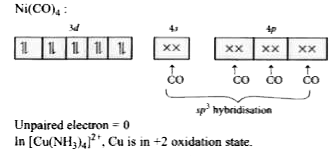 Unpaired electron =0 In `[Cu(NH_(3))_(4)]^(2+)`, Cu is in +2 oxidation state. `Cu^(2+): [Ar]3d^(9)4s^(0)` `[Cu(NH_(3))_(4)]^(2+)` 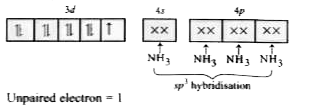 Unpaired ELECRTRON =1 |
|
| 27. |
The number of equivalent Cr-O bonds in CrO_4^(2-) is |
|
Answer» 1 |
|
| 28. |
The numberofenantiomeric paristhatcan beproduct duringmonochlorination Of 2-mehtybutane is : |
|
Answer» 2 |
|
| 29. |
The number of unpaired electrons in outer orbital [Fe(H_(2)O)_(6)]^(3+) complexis : |
|
Answer» 1 |
|
| 30. |
The number of elements that can be accommodated in the present set up of the long form of the periodic table is (upto 7^(th) period). |
| Answer» ANSWER :B | |
| 31. |
The number of elliptical orbits, including circular orbits in the M shell of an atom is: |
|
Answer» 3 |
|
| 32. |
The number of unpaired electrons in Ni^(3+). is |
|
Answer» 3 |
|
| 33. |
The number of elements in the fifth period of periodic table is |
|
Answer» 8 |
|
| 34. |
The number of elements in each of the long periods in the periodic table is |
|
Answer» 2 |
|
| 35. |
The number of unpaired electrons in Ni (atomic number = 28) are |
|
Answer» 0  `THEREFORE` No. of unpaired ELECTRONS= 2 |
|
| 36. |
The number of elements in each long period is : |
|
Answer» 18 |
|
| 37. |
The number of unpaired electrons in Ni^(2+) is : |
|
Answer» Zero |
|
| 38. |
The number of elements in each of the inner transition series is |
|
Answer» 2 |
|
| 39. |
The number of unpaired electrons in Mn^+is: |
|
Answer» 3 |
|
| 40. |
The number of electrons to balance the following equation, the value of x is - |
|
Answer» 5 |
|
| 41. |
The number of unpaired electrons in Lu ^(3+) are |
|
Answer» 0 |
|
| 42. |
The number of electrons that have a total charge of 9650 coulombs is |
|
Answer» `6.22 TIMES 10^(23)` |
|
| 43. |
The number of unpaired electrons in [Fe(CN)_(6)]^(3-) is.............and the magnetic moment value is |
|
Answer» |
|
| 44. |
The number of electrons that have a total charge of 9650 coulombs is …………………. . |
|
Answer» `6.22 xx 10^(23)` `:. 9650 C = (6.22 xx 10^(23))/(96500) = 6.022 xx 10^(22)`. |
|
| 45. |
The number of unpaired electrons in [Fe(CN)_(6)]^(3-) is : |
| Answer» Answer :B | |
| 46. |
The number of electrons that have a total charge of 965 coulombs is |
|
Answer» `6.022xx10^(23)` |
|
| 47. |
The number of electrons required to reduced4.5 xx 10^(-5)g of Al is |
|
Answer» ` 1.03xx10^(18)` 27 g of Al is reduced by `=3 xx 6.023xx10^(23)E^(-)s` ` 4.5xx10^(-5)` g of Al will be reduced by `=(3xx6.023xx10^(23) xx 4.5xx10^(-5))/(27)` `=3.01 xx 10^(18)` electrons |
|
| 48. |
The numberof electronsrequiredto balancethe followingequation, NO_(3)^(-) +4H^(+) +e^(-)to2H_2 O +NOis |
|
Answer» 5 in thisequationall theatomsarebalanced.To balancechargeAdd`3E^(-)`to L.H.Swe have `NO_3^(-)+ 4H^(+)+ 3e^(-)to 2H_2O +NO ` |
|
| 49. |
The number of unpaired electrons in d^(7), low spin, octahedral complex is |
|
Answer» |
|