Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The number of electrons required to deposit 1 g atom of Al(at. Wt.=27) from a solution of AlCI_3 are : |
|
Answer» 1N |
|
| 2. |
The number of electrons present in the valency shell of P in PCl_3 is: |
|
Answer» 12 |
|
| 3. |
The number of unpaired electrons in Cu^(+) (Z=29) is |
|
Answer» 1 |
|
| 4. |
The number of electrons present in the valence shell of group 13: |
|
Answer» One |
|
| 5. |
The number of unpaired electrons in Cr^(3+) ion is |
|
Answer» 3 |
|
| 6. |
The number of electrons passing per second through a cross section of Cu wire carrying 10 ampere is |
|
Answer» `6 XX 10^(19)` 1 F = 96500 C = `6 xx 10^(23)` electron `therefore` 10 C = n ELECTRONS `therefore n = (6 xx 10^(23) xx 10)/(96500) = 6.2 xx 10^(19)` |
|
| 7. |
The number of electrons passing per second through a cross-section of Cu wire carrying 10 ampere is: |
|
Answer» `6XX10^(19)` |
|
| 8. |
The number of unpaired electrons in chromium (Z=24) is : |
|
Answer» 4 |
|
| 9. |
The number of electrons passing per second through a cross-section of copper wire carrying 10^(-6) ampere: |
|
Answer» `6.2xx10^(23)` |
|
| 10. |
The number of unpaired electrons in central metal of cobalt ferrocyanide, Co_2[Fe(CN)_6] is |
|
Answer» 0 |
|
| 11. |
The number of electrons passing per second through a cross section of copper wire carrying 10^(-6) aperes of current per second is found to be |
|
Answer» `1.6xx10^(-19)` NUMBER of electrons passed per second `=(10^(-6))/(1.602xx10^(-19))=6.24xx10^(12)` |
|
| 12. |
The number of electrons passing per second through a cross-section of copper wire carrying 10^6ampere: |
|
Answer» `6.2xx10^(23)` |
|
| 13. |
The number of unpaired electrons in Ce ^(3+) ion is 0 |
|
Answer» 0 |
|
| 14. |
The number of electrons lost or gained during the change, Fe + H_2O rarrFe_3O_4 +H_2 |
|
Answer» 2 |
|
| 15. |
The number of electrons involved in the reduction of nitrate ion to hydrazine is |
|
Answer» 8 tobalanceN atomsmultiply`NO_(3)^(- )` by2 WEHAVE 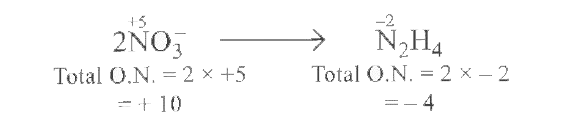 To balanceO.Nadd`14E^(-)`to L.H.Swe have `2NO_(3)^(- )+ 14 e^(- )toN_2 H_4` ` orNO_(3)^(-)+ 7 e^(-)to1//2N_2H_4 ` thereforenumberof electronsinvolvedin thereductionof ` NO_(3)^(-)` IONSIS 7 . |
|
| 17. |
The number of electrons involved in the reaction when one faraday of electricity is pased through an electrolyte is |
|
Answer» `12 xx 10^(46)` |
|
| 18. |
The number of unpaired electrons in a d^(7) tetrahedral configuration is : |
|
Answer» 3 |
|
| 19. |
The number of electrons involved in the electro deposition of 63.5 g. of Cu from a solution ofCuSO_4 is |
|
Answer» `6.0 XX 10^(23)` |
|
| 20. |
The number of electrons involved in reox reaction when a faraday of electricity is passed through an electrolyte in solution is:- |
|
Answer» `6xx10^(23)` |
|
| 21. |
The number of unpaired electrons expected for the complex ion [Cr(NH_(3))_(6)]^(2+) is : |
|
Answer» 2 |
|
| 22. |
The number of electrons involved in redox reactions when a faraday of electricity is passed through an electrolyte in solution is : |
|
Answer» `6XX10^(23)` |
|
| 23. |
The number of electrons in the valency shell of carbon in methyl carbonium ion is: |
|
Answer» 4 |
|
| 24. |
The number of unpaired electrons and magnetic moment value of [CoF_(6)]^(3-) are ..............and............respectively. |
|
Answer» |
|
| 25. |
The number of electrons in a mole of hydrogen molecule is : |
|
Answer» `6.023 XX 10^23` |
|
| 26. |
The number of unpaired electron in respectively are : |
|
Answer» 2,2,1 |
|
| 27. |
The number of electrons in a halogen in its outermost orbit in comparison with corresponding noble gas is: |
|
Answer» ONE ELECTRON LESS |
|
| 28. |
The number of unpaired electron in [Pt(CN)_4]^(2-) ion having square planar geometry (At.No. of Pt=78) is |
|
Answer» 0 |
|
| 29. |
The number of unpaired electron in [Ni(CO)_2]is |
|
Answer» 0 |
|
| 30. |
The number of electrons donated from substance(s) getting oxidized to the substance(s) getting reduced in the chemical equaton for the following reaction is: Cr_2O_(7)^(2-) + Fe^(2+) + C_2O_(4)^(2-) rarrCr^(3+) +Fe^(3+) + CO_2(Unbalanced) |
|
Answer» 6 |
|
| 31. |
The number of unit cells in 8 gm of an element X( atomic mass 40 ) which crystallizes in bcc pattern in ( N_(A)is the Avogadro number) |
|
Answer» `6.023xx 10^(23)` 1 molecontains6.023 `xx 10^(23)` atoms `0.2 ` molecontains0.2 `xx 6 .023 xx 10^(23) ` atoms `((1 unit cell)/( 2 atom)) xx 0.2 xx 6.023 xx 10^(23) = 6.023 xx 10^(22)` unit calls |
|
| 32. |
The number of electrons in 4d-subshell of 'Pd' is |
|
Answer» 7 |
|
| 33. |
The number of unit cells in 8 gm of an element X (atomic mass 40) which crystallizes in bcc pattern is (N_(A)" is the Avogadro number")………………. . |
|
Answer» `6.023xx10^(23)` Number of atoms in 8g of element is, Number of moles `=(8g)/("40g, MOL"^(-1))=0.2 mol` `"1 MOLE contains "6.023xx10^(23)" atoms"` `"0.2 mole contains"0.2xx6.023xx10^(23)" atoms"` `(("1 unit cell")/("2 atoms"))xx0.2xx6.023xx10^(23)""6.023xx10^(22)" unit cells"` |
|
| 34. |
The number of electrons delivered at the cathode during electrolysis by a current of 1A in 60 seconds is (charge of electron = 1.6 xx 10^(-19) C) |
|
Answer» `6.22 xx 10^(23)` `= 1A = 60S` 96500 C CHARGE = `6.022 xx 10^(23) ` ELECTRONS 60 C charge = `(6.022 xx 10^(23))/(96500) xx 960 = 3.744 xx 10^(20)` electrons . |
|
| 35. |
The number of unidentate ligands in the comples ion is called |
|
Answer» oxidation number e.g. `[Co(NH_(3))_(6)]^(3+)` - coordination number = 6. |
|
| 36. |
The number of tripeptides formed by three different amino acids is …………........... |
| Answer» SOLUTION :Six | |
| 37. |
The number of types of bonds between two carbon atoms in calcium carbide is : |
|
Answer» One sigma, one pi `Ca^(2+)[C-= C]^(2-)` It CONTAINS one sigma and 2 pi bonds. |
|
| 38. |
The number of unpaired electrons in P-atom is |
|
Answer» 1 `1s^(2)2S^(2)2P^(6)3S^(2)3p_(x)^(1)3p_(y)^(1)3p_(z)^(1)` No. of unpaired electrons =3 |
|
| 39. |
The number of electrons delivered at the cathode during electrolysis by a current of 1 ampere in 60 seconds is (charge on the electron=1.60xx10^(-19)C) |
|
Answer» `6XX10^(23)` `=1xx60=60` coulombs But charge flowing=number of electrons flowing`xx`charge on each ELECTRON `therefore60=nxx1.60xx10^(-19)` or `n=(60)/(1.60xx10^(-19))=37.5xx10^(19)=3.75xx10^(20)` |
|
| 41. |
Thenumber of electrons delivered at the cathode during electrolysis by a current of 1A in 60 seconds is (charge of electron =1.6 times 10^(-19) C) |
|
Answer» `6.22 TIMES 10^(23)` |
|
| 42. |
The number of tripeptides formed by thee different amino acids are |
|
Answer» Three |
|
| 43. |
the number of electrons accommodated in an orbital with principal quantum number 3 is |
|
Answer» 2 |
|
| 44. |
The number of tripeptides formed by three different amino acids having three different amino acids is |
| Answer» ANSWER :D | |
| 45. |
The number of electrons change during the conversion of nitrobenzene to aniline is |
|
Answer» |
|
| 46. |
The number of electron in 3.1 mg NO_(3)^(-) is (N_(A) = 6 xx 10^(23) ) |
|
Answer» 32 |
|
| 47. |
The number of electron and proton in the third alkaline earth metal ion will be |
|
Answer» `e/20,p/20` |
|
| 48. |
The number of transition metals present in Rinmann's greens compound. |
|
Answer» `:.` TWO transitions metals are present `Co&Ti :.2` |
|
| 49. |
The number of double bonds in gammexane is |
|
Answer» 0 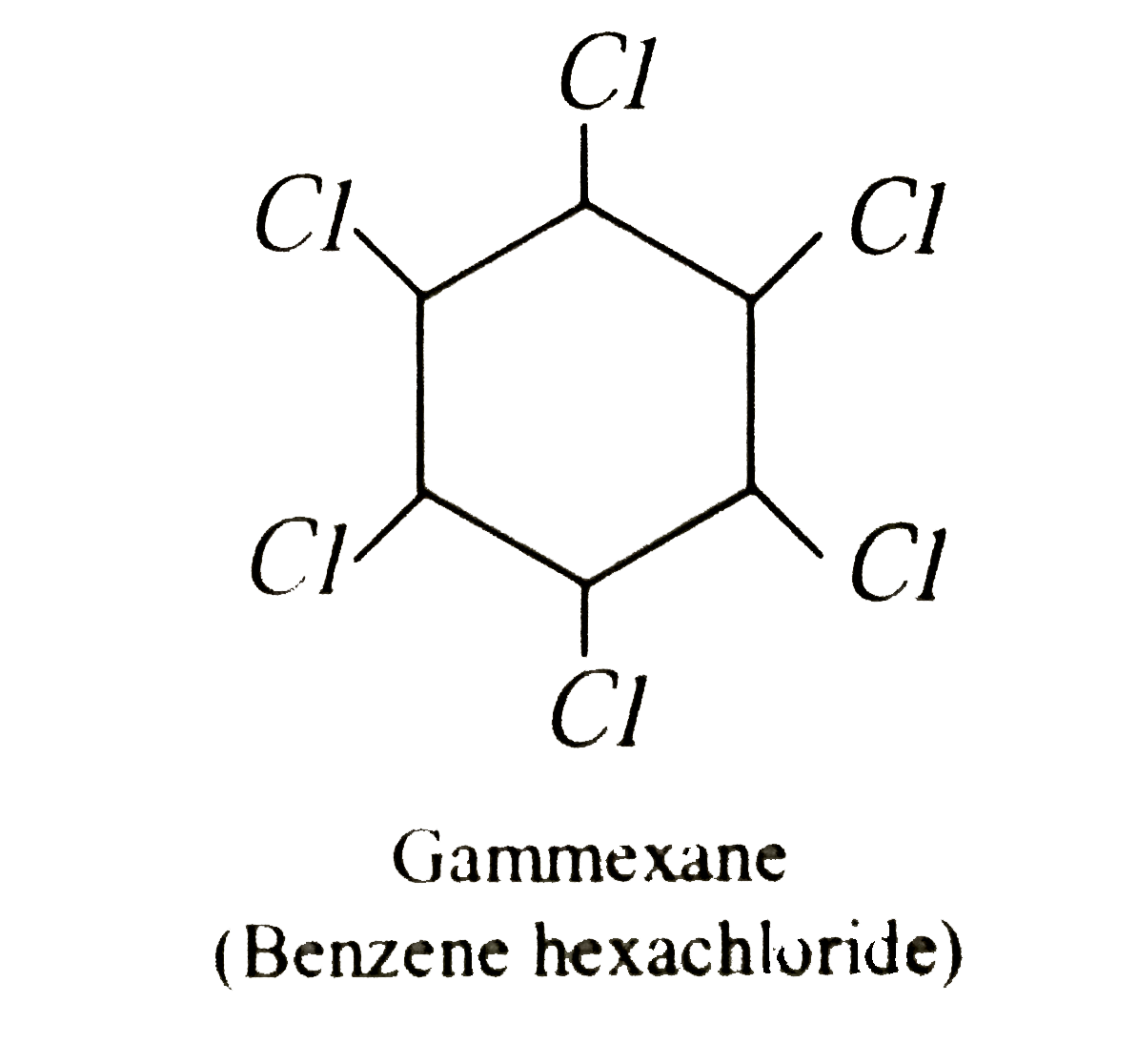 It has no DOUBLE BOND. |
|
