Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The correct order of C-O bond length among CO, CO_3^(2-), CO_2 is: |
|
Answer» `CO_3^2 ltCO_2 LTCO` |
|
| 2. |
The correct order of C-X bond polarity is |
|
Answer» `CH_(3)Br gtCH_(3)ClgtCH_(3)I` `CH_(3)Cl gt CH_(3)Br gt CH_(3)I` |
|
| 3. |
The correct order of C–X bond polarity is: |
|
Answer» `CH_(3) Br gt CH_(3) CL gt CH_(3) l` |
|
| 4. |
The correct order of B.P. of R-X |
|
Answer» `R-I GT R-Br gt R-Cl` |
|
| 5. |
The correct order of b.p. of hydride is ...... |
|
Answer» `PH_3ltAsH_3ltNH_3ltSbH_3` |
|
| 7. |
The correct order of bond dissociation enthalpy of halogens is........... |
|
Answer» `F_(2) GT Cl_(2) gt Br_(2) gt I_(2)` <BR>`I_(2) gt Br_(2) gt Cl_(2) gt F_(2)` |
|
| 8. |
The correct order of bond anlge is : |
|
Answer» `NO_(2)^(-)gtNO_(2)^(+)gtNO_(2))` |
|
| 9. |
The correct order of boiling points of isomeric amines is |
|
Answer» TERTIARY gtsecondary gtprimary |
|
| 10. |
The correct order of bond angle (smallest first) in H_(2)S, NH_(3), BF_(3)andSiH_(4)is : |
|
Answer» `H_(2)S LT NH_(3)lt SiH_(4) lt BF_(3)` |
|
| 11. |
The correct order of boiling points of alkyl halides is |
|
Answer» `CH_3FgtCH_3ClgtCH_3BrgtCH_3I` |
|
| 12. |
The correct order of boiling points of the following isomeric amines is C_4H_9NH_2,(C_2H_5)_2NH,C_2H_5N(CH_3)_2 |
|
Answer» `C_2H_5N(CH_3)_2 GT (C_2H_5)NH gt C_4H_9NH_2` |
|
| 13. |
The correct order of boiling points for primary (1^@), secondary (2^@) and tertiary (3^@) alcohols is |
|
Answer» `1^@GT2^@GT3^@` |
|
| 14. |
The correct order of boiling point is: |
|
Answer» `T_(2)ltD_(2)gtH_(2)` (b)n-pentane`gt`neo-pentane[BP`prop`VWF`prop`CONTACT area `prop(1)/("BLEACHING")]` (c)`XegtArgtHe`[BP`prop`mol.wt.] (d) 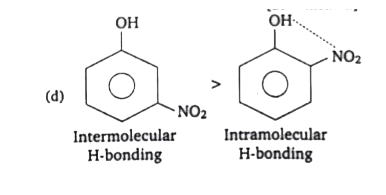
|
|
| 15. |
The correct order of boiling point is |
|
Answer» `T_(2) lt D_(2) lt H_(2)` |
|
| 16. |
The correct order of boiling point is : |
|
Answer» `I GT II gt III` |
|
| 17. |
The correct order of boiling point for the given elements is |
|
Answer» `HegtNegtXegtArgtKr` |
|
| 18. |
The correct order of boiling point is : |
|
Answer» `NH_(3) LT HF lt H_(2)O lt H_(2)O_(2)` |
|
| 19. |
The correct order of boiling point for primary (1^(@)) , secondary (2^(@)) and tertiary (3^(@)) alcohols is |
|
Answer» `1^(@) GT 2^(@) gt 3^(@)` Boiling POINT of ALCOHOLS decreases as the number of branches increases. |
|
| 20. |
Order of boiling point among primary , secondary and tertiary alcohols is : |
|
Answer» `1^(@) GT 2^(@) gt 3^(@)` |
|
| 21. |
The correct order of basictiies of the following compounds is : . 3. (CH_(3))_(2)NH. 4. CH_(3)-overset(O)overset(||)(C)-NH_(2) |
|
Answer» `(2) GT (1) gt (3) gt (4)` 1.` (##KSV_CHM_ORG_P2_C15_E01_152_S01.png" width="80%"> LTBRGT More basci due to the presence of two ` LP overlinee^, s` on each (N). 2.` EtNH_2`, Due to `+I` effect of (Et) group , but `+ I` effect to two Me groups is greater than `+I` effect of `(Et)` group. Hence (3) is more basic than (2). 3.`(CH_3)_2 NH`. Due to `(+I)` effect of two Me groups. 4.` (##KSV_CHM_ORG_P2_C15_E01_152_S02.png" width="80%"> (Amides are resonance STABILISED , so they are the weakest base , Amides behalve as amphoteric ). |
|
| 22. |
The correct order of boiling point among the following. |
|
Answer» `PH_(3) gt NH_(3) gt N (CH_(3))_(3) gt N (C_(2)H_(5))_(3)` |
|
| 23. |
Thecorrectorderof basicityof the followingcompoundin aqueousmediumis(1) NH_(3) (2) CH_(2)CH_(2)NH_(2) (3) (CH_(3))_(2)NH (4) (C_(2)H_(5))_(3)N |
|
Answer» `2 GT 3 gt 1 gt 4` |
|
| 24. |
Thecorrectorderof basicityof followingcompoundin aqueousmediumis(1) (C_(2)H_(5))_(2)NH (2) C_(2)H_(5) - NH_(2) (3) (CH_(3))_(2)NH (4) CH_(3)NH_(2) |
|
Answer» `1 gt 2 gt 3 gt 4` |
|
| 25. |
The correct order of basicity of the following compouds is |
|
Answer» `1 lt 2 lt 3 lt 4 ` (i) Donation capacity of nitrogen increases if electron donating GROUP(s) (+/ or +R-effect CONTAINING) is/arc attached with it. (ii) More nitrogen atoms (or donor atoms) means more BASICITY (in general). (iii) `sp^(2)`-hybridiscd nitrogen is less capable of donating lone pair than `sp^(3)` -hybridised. Now, look into the structures individually. 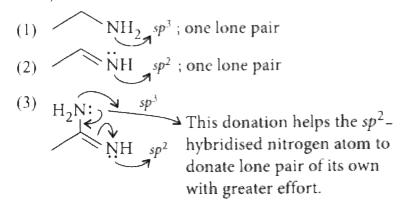 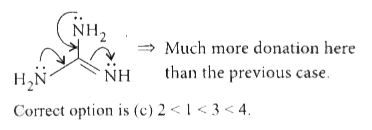
|
|
| 26. |
The correct order of basicities of the following compounds is - (I) , (II) CH_(3)CH_(2)NH_(2) : (III) (CH_(3))_(2)NH , (IV) CH_(3)-overset(O)overset(||)C-NH_(2)) |
|
Answer» `IIgtIgtIIIgtIV` |
|
| 27. |
The correct order of basicity of CH_(3)NH_(2)(I),(CH_(3))_(2)NH(III) and (CH_(3))_(3)N in chlorobenzene is- |
|
Answer» `IltIIIltII` |
|
| 28. |
The correct order of basicities of the following compounds is |
|
Answer» `2 gt 1 gt 3 gt 4` `{:(""NH),("||"),(CH_(3)-C-NH_(2) gt UNDERSET(2^(@)"-Amine")((CH_(3))_(2)NH) gt underset(1^(@)"-Amine")(CH_(3)CH_(2)NH_(2)) gt underset("AMIDE")(CONH_(2))):}` |
|
| 29. |
The correct order of basicities of PhNH_(2)(A), and Ph_(2)NH(B) and cyclohexyl-NH_(2)(C) is |
|
Answer» `AgtBgtC` `UNDERSET((A))(C_(6)H_(5)NH_(2))gtunderset((B))((C_(6)H_(5))_(2)NH)` The LONE pairs of N are not involved in delocalization in cylcohexylamine (C), hence it has HIGHEST basicity. `therefore` Order of basicity : `CgtAgtB` |
|
| 30. |
The correctorderof basicstrengthof amines inaqueousreaction? (1) C_(2)H_(5)NH_(2) (2) (CH_(3))_(2)NH (3) (C_(2)H_(5))_(2)NH (4) (C_(2)H_(5))_(3)N |
|
Answer» ` 3 GT 2 gt 1 gt 4` |
|
| 31. |
Correct order of basic strengths of given amines is : |
|
Answer» `CH_3 NH_2 GT (CH_3)_2NH gt (CH_3)_3N gt NH_3` |
|
| 32. |
The correct order of basic strength of hydrides of group-15 is ............ |
|
Answer» `NH_(3) GT PH_(3) gt AsH_(3) gt SbH_(3) gt BiH_(3)` |
|
| 33. |
The correct order of basic strength of CH_(3)NH_(2)(I),(CH_(3))_(2)NH(II),(CH_(3))_(3)N(III),C_(6)H_(5)CH_(2)(IV) in gaseous phase is |
|
Answer» `IV lt III lt II lt I` |
|
| 34. |
The correct order of basic strength is |
|
Answer» `H_(2)O lt OH^(-) lt CH_(3)OH lt CH_(3)O^(-)` `underset("(Increasing order of basic strength)")overset(H_(2)O lt CH_(3)OH lt OH^(-) lt CH_(3)O^(-))rarr` |
|
| 35. |
The correct order of basic strength amongst the following compound is |
|
Answer» `I gt II gt III gt IV` |
|
| 36. |
The correct order of adsorption of the gases studies will be |
|
Answer» `NH_(3)gt SO_(2) gt CO_(2)gt HCl` |
|
| 37. |
The correct order of acidity of the following compounds is |
|
Answer» III GT IV gt I gt II |
|
| 38. |
The correct order of acidity is |
|
Answer» `C_6H_5COOH GT HCOOH gt CH_3COOH` Acidic NATURE : `HCOOH gt C_6H_5COOH gt CH_3COOH` `pK_a : 3.75"" 4.2 "" 4.76` |
|
| 39. |
The correct order of acidity in given compounds (i) FCH_(2)COOH (ii) CICH_(2)COOH (iii) NO_(2) CH_(2)COOH (iv) CH_(3)COOH. |
|
Answer» `IGT ii GT iii gt iv` |
|
| 40. |
The correct order of acidity for the following compounds is |
|
Answer» `IgtIIgtIIIgtIV` |
|
| 41. |
The correct order of acidic strength of the following is (I) Oxalic acid HOOC-COOH (II) Malonic acid HOOC-CH_(2)-COOH (III) Succinic acid HOOC-(CH_(2))_(2)-COOH (IV) Glutaric acid HOOC-(CH_(2))_(3)-COOH |
|
Answer» `I GT II gt III gt IV` |
|
| 42. |
The correct order of acidity for the following compounds: |
|
Answer» `I gt II gt III gt IV ` |
|
| 43. |
The correct order of acidic strength of oxo acids of phosphorus is ............. |
|
Answer» `H_(3)PO_(4) GT H_(3)PO_(3) gt H_(3)PO_(2)` |
|
| 44. |
The correct order of acidic strength of carboxylic acids is |
|
Answer» FORMIC acidltbenzoic acidltacetic ACID |
|
| 45. |
The correct order of acidic strength of acids : H_3PO_4, H_4P_2O_7 and H_5P_3O_10 is .......... |
|
Answer» `H_(3)PO_(4) gt H_(4)P_(2)O_(7) gt H_(5)P_(3)O_(10)` 
|
|
| 46. |
Which of the the order of acidic strength is not correct? |
|
Answer» `HI gt HBr gt HCL` `HClO lt HClO_(2)ltHClO_(3)ltHClO_(4)` |
|
| 47. |
The correct order of acidic strength is: |
|
Answer» `CI_(2)O_(7) GT SO_(2) gtP_(4)O_(10)` |
|
| 48. |
The correct order of acidic strength is : |
|
Answer» `IIIgtIVgtIgtII` |
|
| 49. |
The correct order of acidic strength. |
|
Answer» `Cl_(2)O_(7) gt SO_(2) gt P_(4)O_(10)` |
|
| 50. |
The correct order of acidic stength is : |
|
Answer» <P>`Cl_2O_7gt SO_3gt P_4O_(10)` `HClO_(4) gt H_(2)SO_(3)gt H_(3)PO_(4)`. |
|