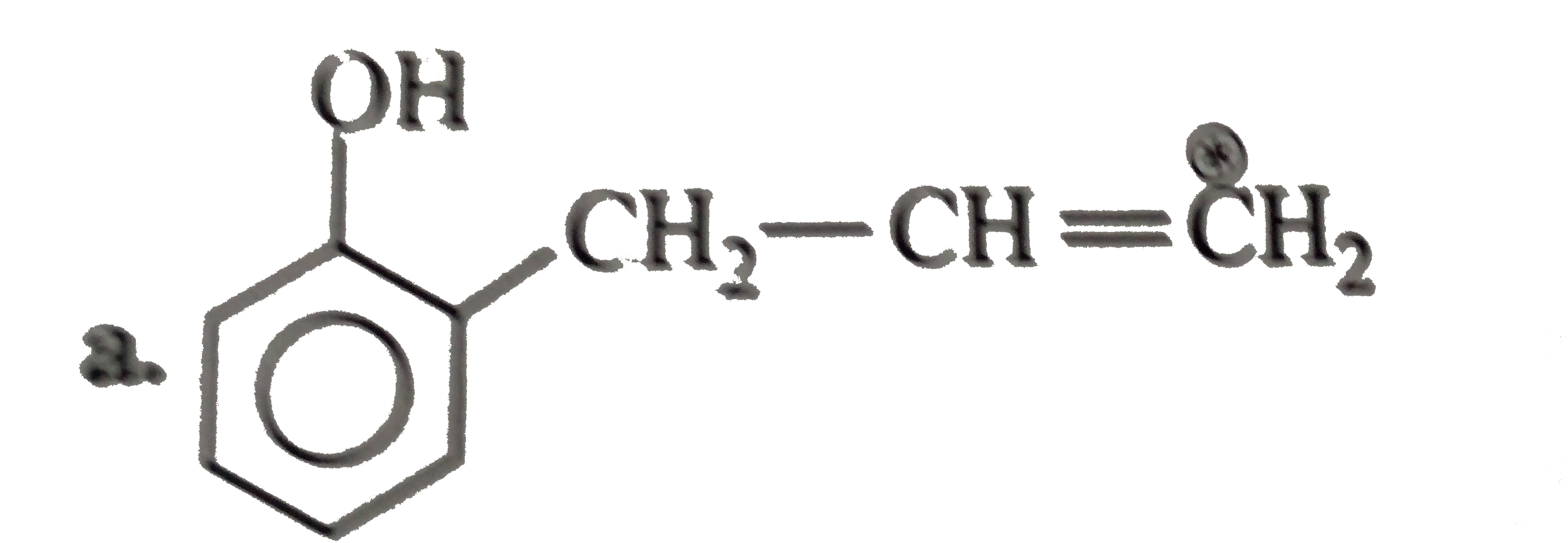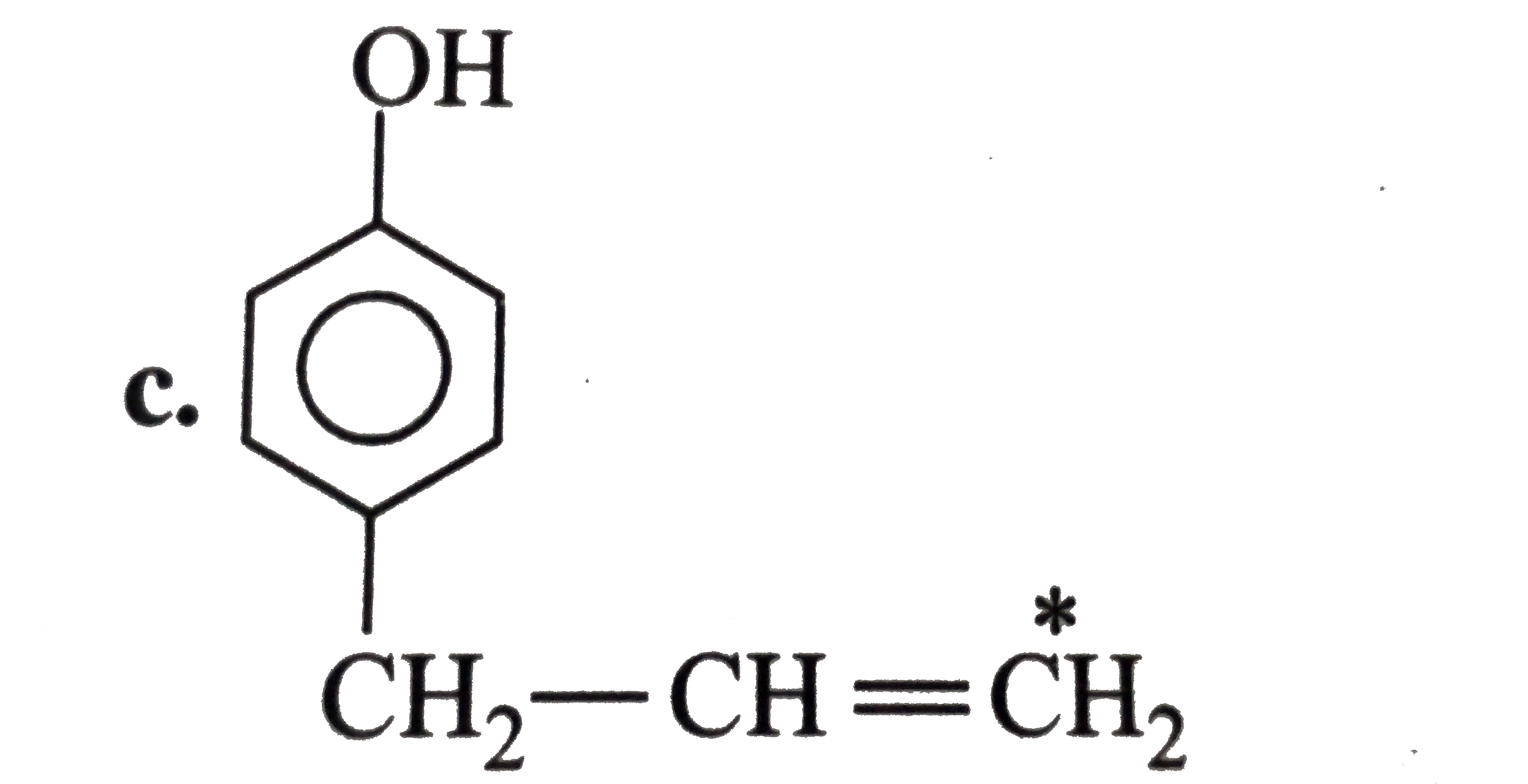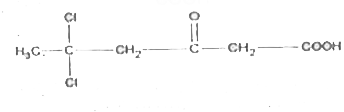Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
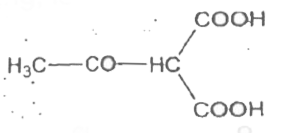
The compound in which carbon uses sp^(3) hydrid orbitals for bond formation is : |
|
Answer» `HCOOH` |
|
| 2. |
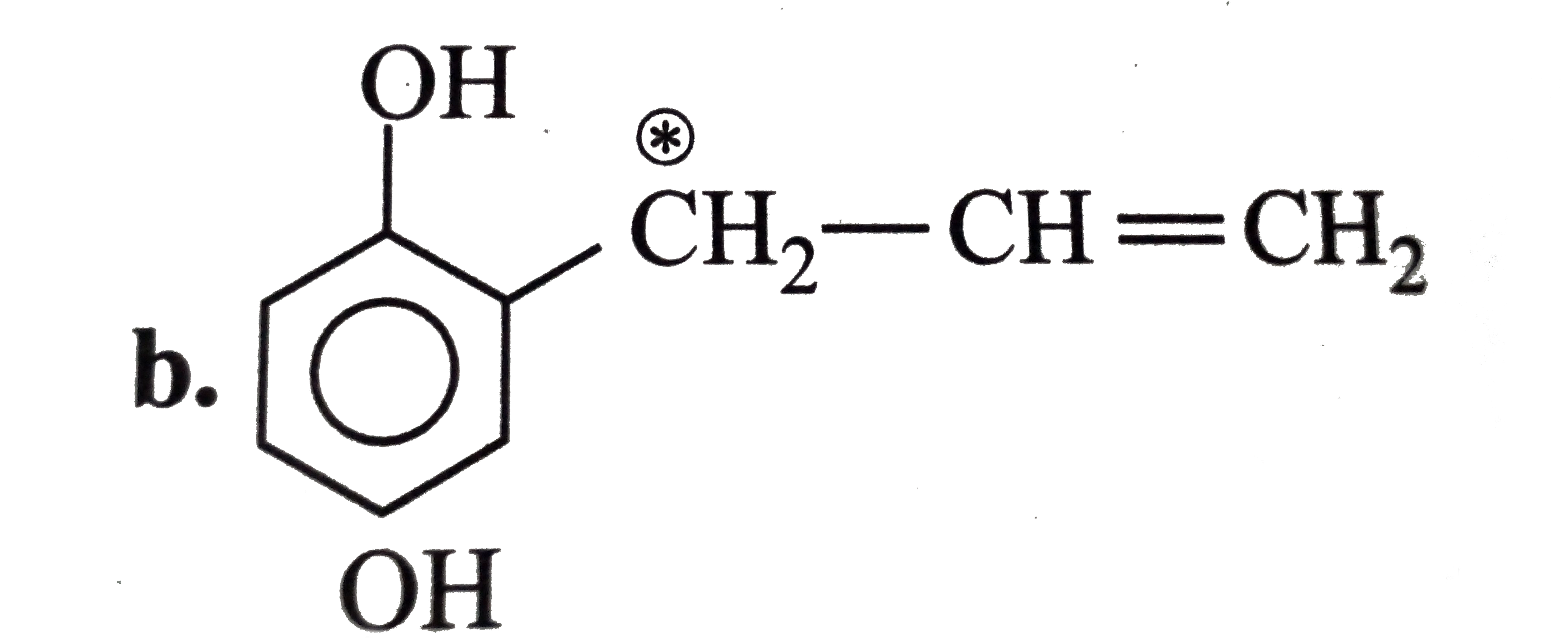
The compound in which carbon atom uses only its sp^ 3 - hybrid orbitals for bond formation is : |
|
Answer» HCOOH |
|
| 3. |
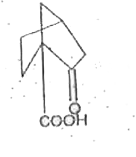
The compound in which C* uses sp^(3) hybrid orbitals for bond formation is |
|
Answer» `HC*OOH` |
|
| 4. |
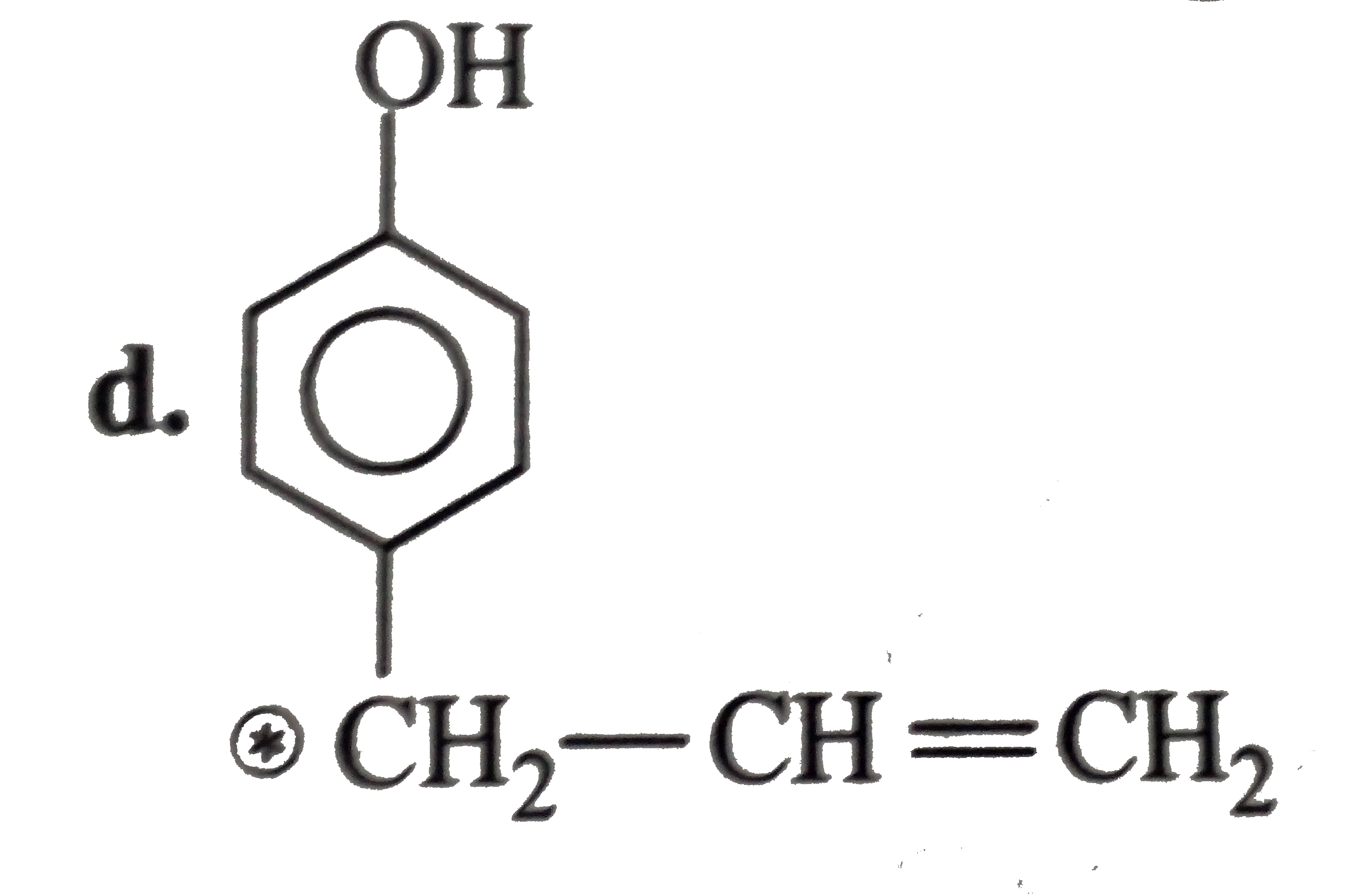
The compound in which C uses its sp^3-hybrid orbitals for bond formation is: |
|
Answer» `H oversetxxC OOH` |
|
| 5. |
Thecompoundin whichC usesits sp^(2)hybridorbitalsfor bondformationis : |
|
Answer» `HCOOH` |
|
| 6. |
The compound ibuprofen is used as |
|
Answer» antiseptic |
|
| 8. |
The compound (I) is |
|
Answer»
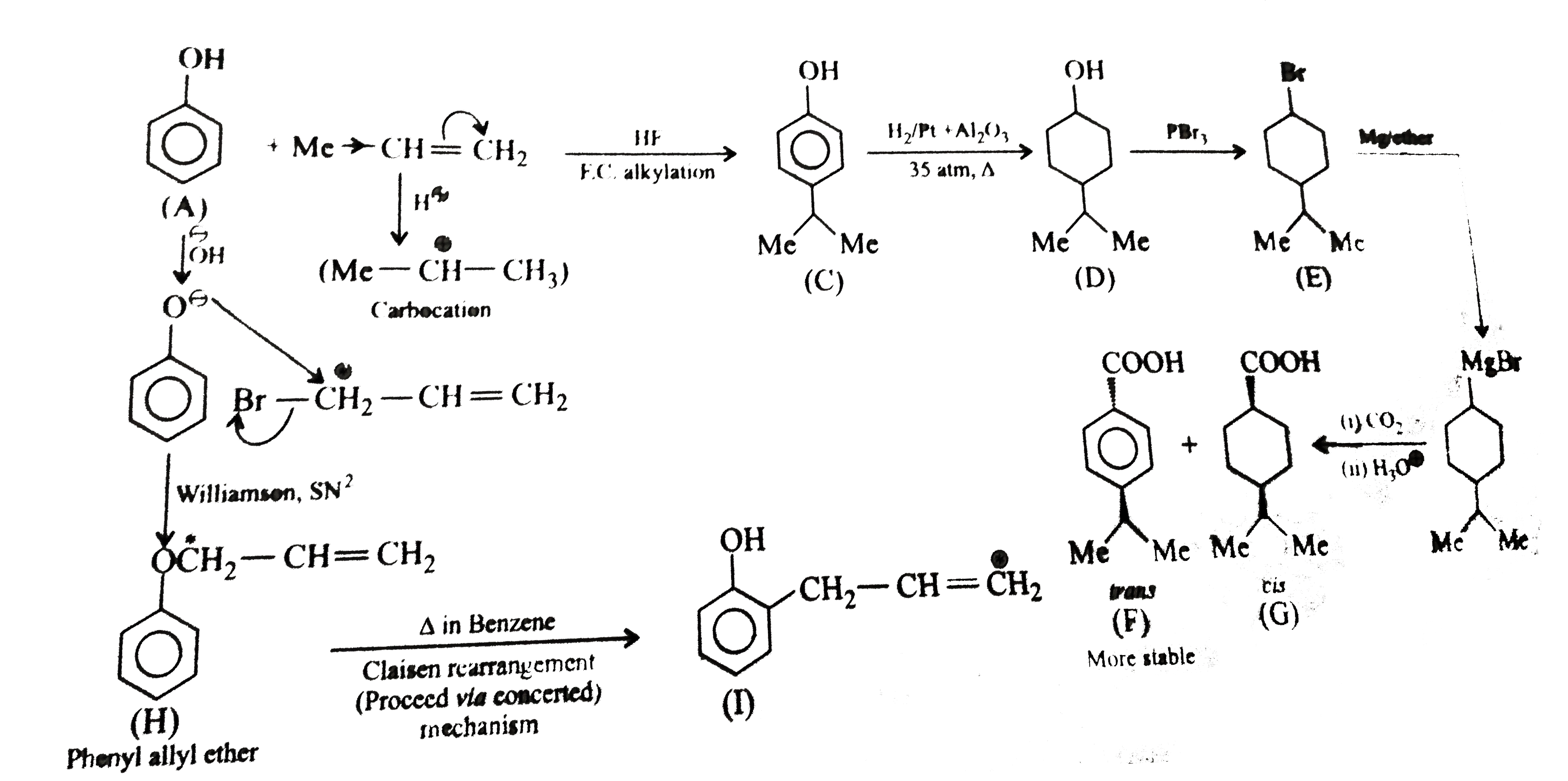
|
|
| 9. |
The compound HCl behaves as .......in the reaction, HCl + HF rarr H_(2)^(+)Cl + F^(-) |
|
Answer» Weak base |
|
| 10. |
The compound having the molecular formula C_3H_9N represents: |
|
Answer» TRIMETHYLAMINE |
|
| 11. |
The compound (i) decolourises KMnO_4(ii) forms ozonide with ozone and (iii) undergoes polymerisation.It will be: |
|
Answer» `C_6H_6` |
|
| 12. |
The compound having S - S single bond is .......... |
|
Answer» `H_(2)S_(2)O_(3)` |
|
| 13. |
The compound having tetrahedral geometry is |
|
Answer» `[NI(CN)_(4)]^(2-)`  Hybridization of `[NiCI_(4)]^(2-)= sp^(3)` SHAPE of `[NicI_(4)]^(2-) = Tetrahedral` Hence, (A) is the correct answer |
|
| 14. |
The compound having one isopropyl group is: |
|
Answer» 2,2,3,3,-tetramethylpentane |
|
| 16. |
The compound having molecular formula C_4H_10O can show: |
|
Answer» Metamerism |
|
| 17. |
The compound having least solubility in water is : |
|
Answer» METHANOL |
|
| 18. |
The compound having general formula M^(2+)Fe_(2)O_(4)^(2–) are called as : |
|
Answer» Ferrite |
|
| 19. |
The compound having both sp^2 and Sp^3hybridised carbon atom is: |
|
Answer» Propene |
|
| 20. |
The compound having both sp and sp^2- hybridised carbon atom is : |
|
Answer» Propene |
|
| 21. |
The compoundhaving maximum boilingpoint is : |
|
Answer» n-Pentane |
|
| 22. |
The compound formed when silver powder is heated with chloroform: |
|
Answer» `CH_4` |
|
| 23. |
The compound formed when malonic ester reacts with urea is : |
|
Answer» Cinnamic ACID |
|
| 24. |
The compound formed when propyl magnesium bromide is treated with carbon dioxide is |
|
Answer» `C_(3)H_(7)COOH` |
|
| 25. |
The compound formed when ethyl bromide is heated with dry silver oxide is |
|
Answer» Dimethyl ether If we take moist `Ag_(2)O` then alcohol is FORMED `Ag_(2)O+H_(2)OTO 2AgOH` `C_(2)H_(5)Br+ AGOH to C_(2)H_(5)OH + AgBr` |
|
| 26. |
The compound formed when calcium acetate and calcium formate is dry distilled |
|
Answer» Acetone 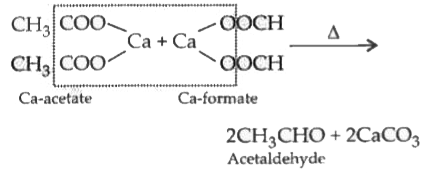
|
|
| 27. |
The compound formed on heating chlorobenzene with chloral in the presence of concentrated sulphuric acid is: |
|
Answer» freon |
|
| 28. |
On heating sodium metal in a current of dry ammonia, the compound formed is |
|
Answer» SODIUM imide |
|
| 29. |
The compound formed on heating chlorobenzene with chloral in the presence of conc. sulphuric acid is |
|
Answer» GAMMEXENE |
|
| 30. |
The compoundformed in the tailing of mercury byO_(3) is |
|
Answer» HGO |
|
| 31. |
The compound formed in the positive test for nitrogen with the Lassaigne solution of an organic compound is |
|
Answer» `Fe_(4)[Fe(CN)_(6)]_(3)` `3Na_(4)[Fe(CN)_(6)]+2Fe^(3+) to underset("prussian blue")(Fe_(4)[Fe(CN)_(6)]_(3)+12Na^(+))` |
|
| 32. |
The compound formed in the positive test for nitrogen with Lassaigne's solution of an organic compound is |
|
Answer» `Na_4[FE(CN)_5NOS]` |
|
| 33. |
The compound formed in the borax bead test of Cu^(2+) ion in oxidising flame is : |
|
Answer» Cu |
|
| 34. |
Thecompound formed in the borax berax test of Cu^(2+) in oxidising flame is |
|
Answer» CU |
|
| 35. |
The compound formed in carbylamine test is: |
|
Answer» `C_6H_5C=N` |
|
| 36. |
The compound formed in borax bead test of Cu^(2+)lon, in oxidizing flame is |
| Answer» Answer :C | |
| 37. |
The compound formed by the reaction of ethyl chloride with a sodium-lead alloy at 400°C is: |
|
Answer» TETRAETHYL lead |
|
| 38. |
The compound formed by reaction of ethylene glycol and periodic acid (HIO_4)is |
|
Answer» CARBONDIOXIDE |
|
| 39. |
The compound formed by reaction of 1, 1- dichloropropane with Zn can also be prepared by the following reaction |
|
Answer» 3, 4-Dibromohexane with zinc `CH_(3)CH_(2)CHCl_(2)+Zn + Cl_(2)CHCH_(2)CH_(3) to CH_(3)-CH_(2)-CH = underset("3 - Hexene")(CH - CH_(2)-CH_(3))` CHOICE (1) and (2) also give 3 - hexene `CH_(3)-CH_(2)-underset(Br)underset(|)CH-underset(Br)underset(|)CH-CH_(2)CH_(3)+Zn to CH_(3)CH_(2)-CH = CH - CH_(2)CH_(3)` `CH_(3)-CH_(2)-underset(Br)underset(|)CH-CH_(2)CH_(3) + alc.KOH to CH_(3)CH_(2) - CH = CH - CH_(2)CH_(3) + KBr + H_(2)O` |
|
| 40. |
The compound form when PtF_(6) oxidised by oxygen d) both He and Ne is |
|
Answer» `PtO_(2)` |
|
| 41. |
The compound formed as a result of oxidation of ethyl benzene by KMnO_4 is |
|
Answer» Benzophenone HENCE(C ) is correct |
|
| 42. |
The compound exhibiting maximum value of equivalent conductance in fused state is |
|
Answer» `SrCl_(2)` |
|
| 43. |
The compound exhibited maximum value of equivalent conductance in a fused state is |
|
Answer» `SrCl_(2)` |
|
| 44. |
The compound ethylisocyanide is prepared by the reaction between : |
|
Answer» `C_(2)H_(5)BR` and KCN |
|
| 45. |
The compounddoesno decarboxylateon simpleheatingis . |
| Answer» Answer :C | |
| 46. |
The compounds [Co(NO_2)(NH_3)_5]CI_2 and [Co(ONO)(NH_3)_5]CI_2 are examples of: |
|
Answer» GEOMETRICAL ISOMERS |
|
| 47. |
The compound condensation of |
|
Answer» ethyl - 2 - METHYL pentanoate 
|
|
| 48. |
The compound commonly used as electrolyte in lead storage batteries is : |
|
Answer» `CuSO_(4)` |
|
| 49. |
The compound CHCl = CHCHOHCOOH with molecular formuls C_(4)H_(5)O_(3)Cl can exhibit |
|
Answer» GEOMETRIC, OPTICAL, POSITION and FUNCTIONAL ISOMERISM 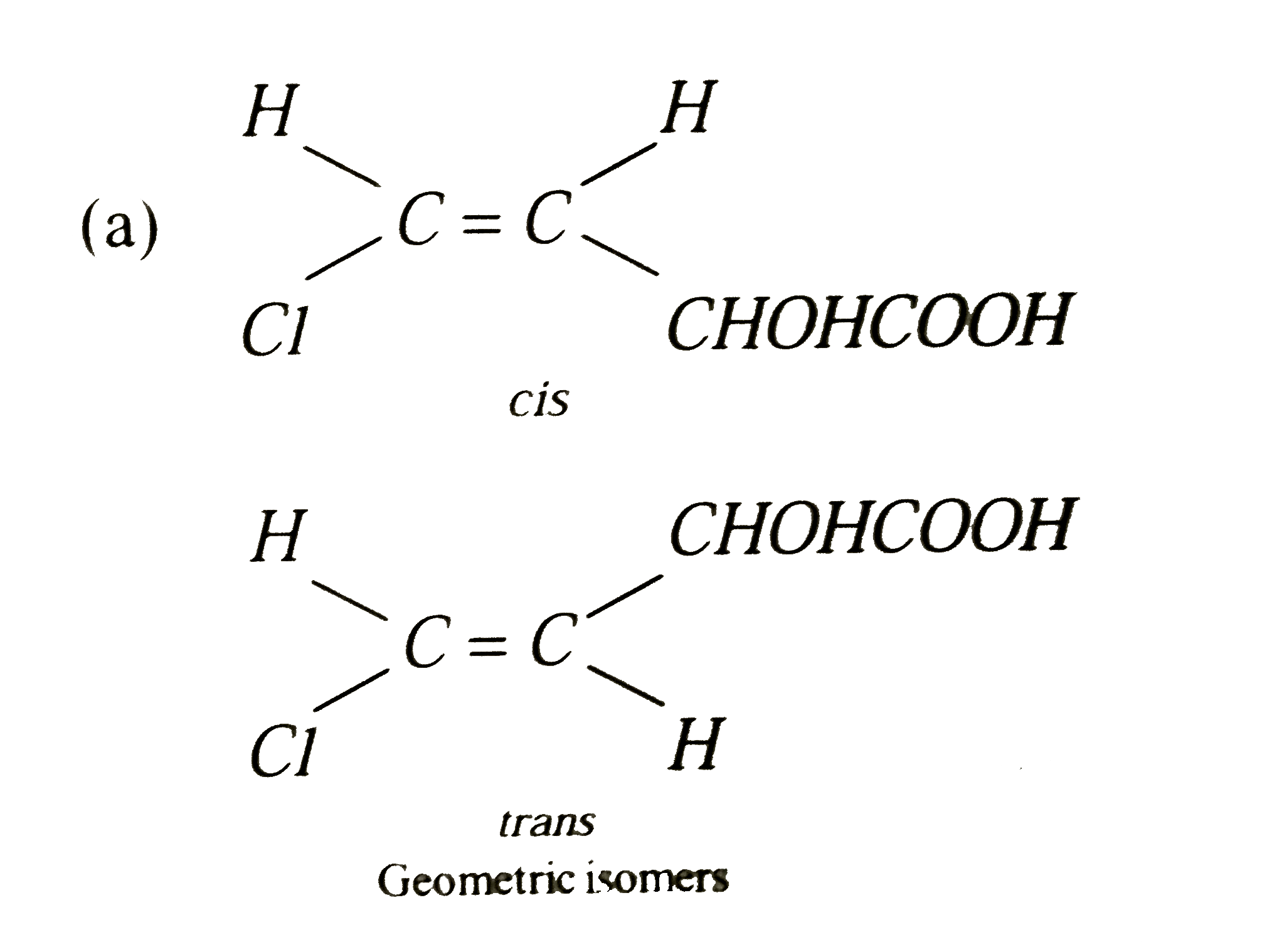 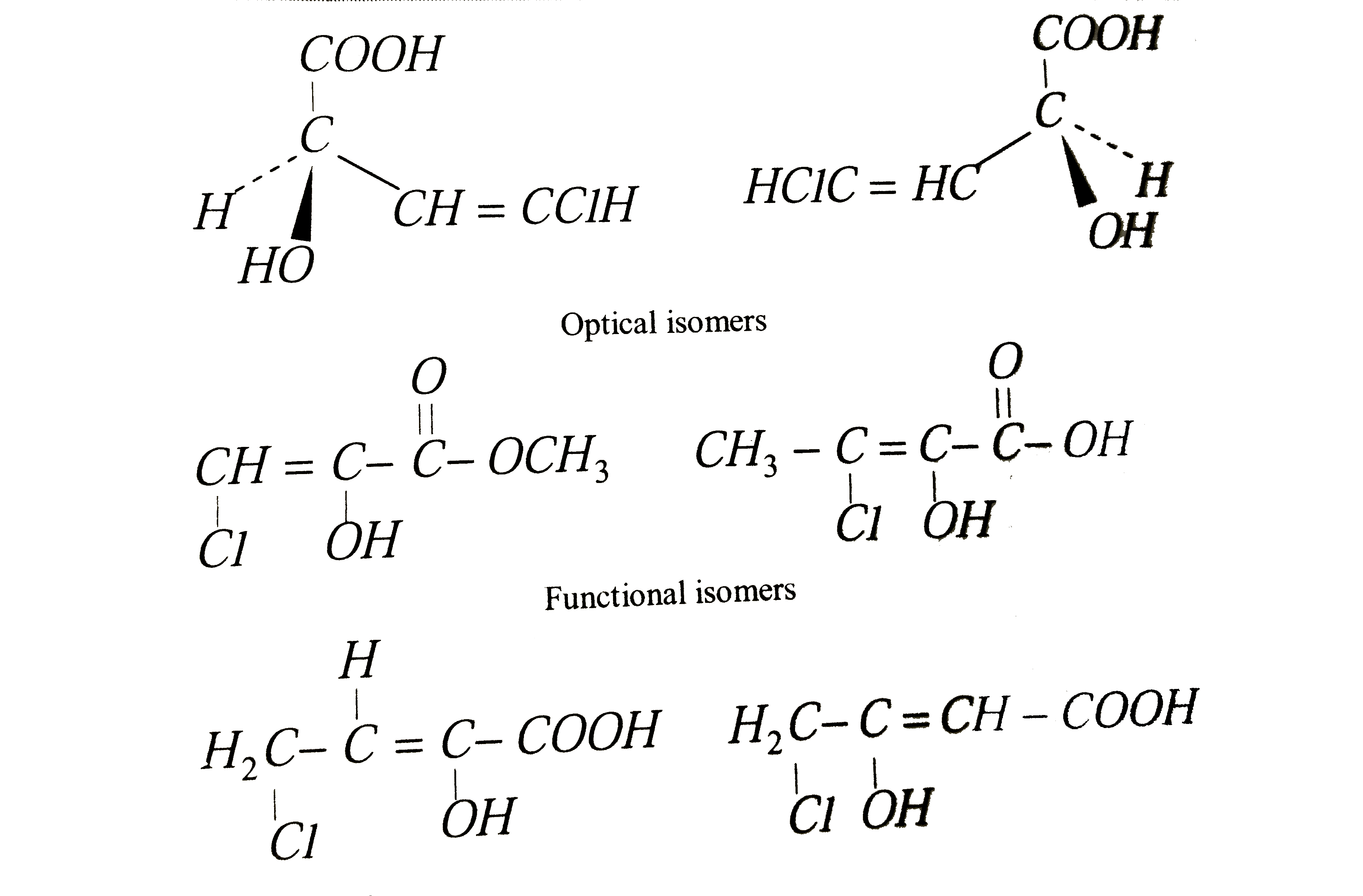
|
|
| 50. |
The compound CHCl-CHCHOHCOOH with molecular |
|
Answer» GEOMETRICAL, OPTICAL, POSITION and FUNCTIONAL ISOMERISM |
|