Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 2. |
The compound that can work both as an oxidizing and reducing agent is : |
|
Answer» `KMnO_4` |
|
| 3. |
The compound that answers iodoform test is _______ |
| Answer» Answer :D | |
| 4. |
The compound that acts as a solvent for Grignard reagent is: |
|
Answer» ETHYL alcohol |
|
| 5. |
The compound soluble in acetic acid is |
|
Answer» CALCIUM oxide |
|
| 7. |
The compound (s) the exhibit(s) geometrical isomerism is (are ) : |
|
Answer» `[PT(en)Cl_(2)]` |
|
| 8. |
The compound (s) soluble in water ? |
|
Answer» `CaF_(2)` |
|
| 9. |
The compound [Pt(NH_(3))_(2)Cl_(2)] can exhibit |
|
Answer» LINKAGE ISOMERISM |
|
| 10. |
The compound possessing most strongly ionic nature is : |
|
Answer» ` Srcl_2 ` |
|
| 11. |
Thecompound on oxidation gives ketone, the original compound is |
|
Answer» `1^@` alcohol |
|
| 12. |
The compound of Xenon with the lowest bond angle |
|
Answer» `XeF_2` |
|
| 13. |
The compound on dehydrogenation gives a ketone . The original compound is |
|
Answer» TERTIARY ALCOHOL 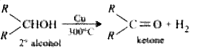
|
|
| 14. |
The compound of Xenon with highest bond angle and zero dipolement |
|
Answer» `XeF_2` |
|
| 15. |
The compound of sulphur that can be used as refrigerant is: |
|
Answer» `S_2Cl_2` |
|
| 16. |
The compound of sulphur that can be used as refrigerant is |
|
Answer» `SO_(2)` |
|
| 17. |
The compound of nitrogen which is supporter ofcombustion and is called laughing gas is. |
|
Answer» `N_2 O` |
|
| 18. |
The compound of Solvay process is |
|
Answer» `Na_2SO_5` |
|
| 19. |
The compound of molecular formula C_(5)H_(10)O(A) reacts with Tollen's reagent to give silver mirror but does not undergo aldol condensation. The compound (A) is : |
|
Answer» 3-pentanone |
|
| 20. |
The compound obtained on chlorination of toluene in the presence of sunlight is hydrolysed with an aqueous solution of NaOH to give - |
|
Answer» <P>BENZYL alcohol |
|
| 21. |
The compound obtained when acetaldehyde reacts with dilute aqueous sodium hydroxide exhibits |
|
Answer» GEOMETRICAL isomerism  It has a stereogenic centre, so it will exhibit optical isomerism. |
|
| 22. |
The compound obtained when acetaldehyde reacts with dilute aqueous sodium hudroxide exhibits |
|
Answer» GEOMETRICAL ISOMERISM 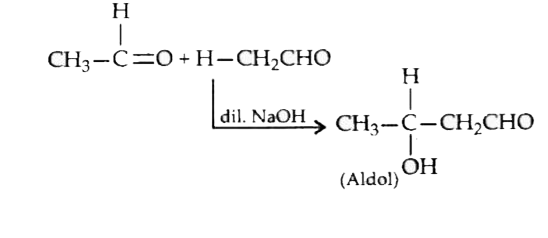 It has a stereogenic CENTRE, so it will EXHIBIT optical isomerism. |
|
| 23. |
The compoundobtainedby reactionfollowingcompoundwith 50% H_(2)SO_(4) CH_(3)- CH_(2) - overset(CH_(3))overset(|)(C) = underset(ONa)underset(|)overset(O^(-))overset(|)(C) |
|
Answer» `CH_(3) - CH_(2) - CO-CH_(2) - CH_(3)` 
|
|
| 24. |
The compound obtained by the reduction of propionaldehyde by Zn/Hgand conc. HCl is |
|
Answer» Propanol |
|
| 25. |
The compound obtained by reducing CH_3 CN with Na//C_2H_5 OH is |
|
Answer» METHYL ALCOHOL |
|
| 26. |
The compound obtained by heating salicylic acid with phenol in the presence of phosphorus oxychloride is |
|
Answer» Salol 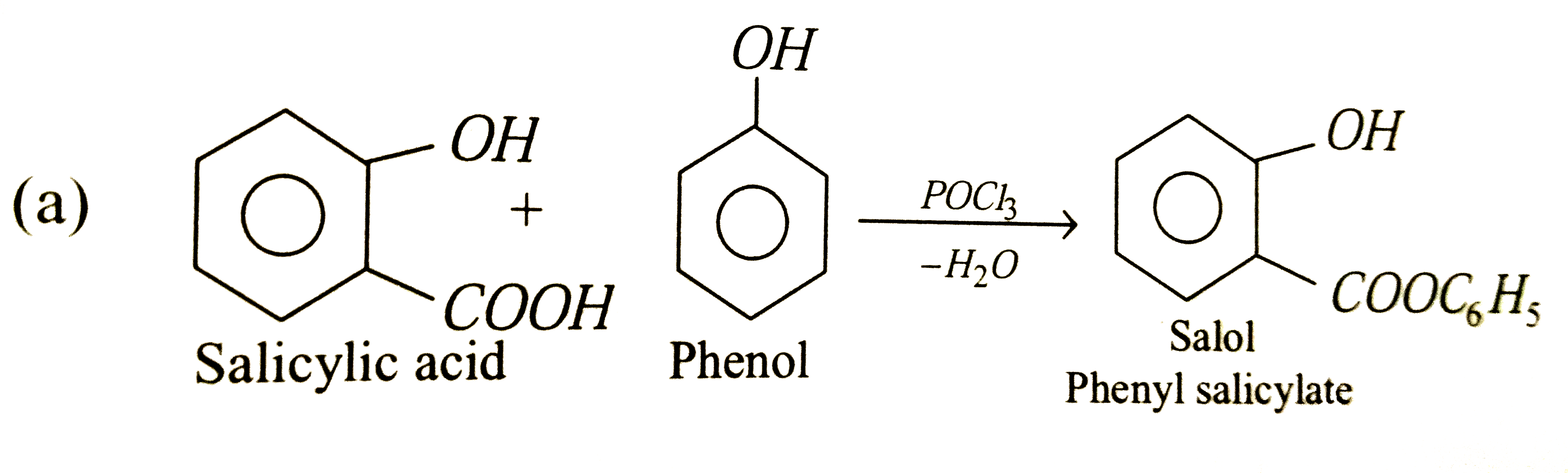
|
|
| 27. |
The compound obtained by heating a mixture of a primary amine and chloroform with ethanolic potassium hydroxide is |
|
Answer» An alkyl isocyanide |
|
| 28. |
The compound obtained by heating a mixture of a primary amine and chloroform with ethanolic potassium hydroxide (KOH) is |
|
Answer» an ALKYL isocyanide |
|
| 29. |
The compound obtainedby catalytichydrogenationof nibrobenationof nitrobenzene is |
|
Answer» ANILINE |
|
| 30. |
Thecompound Na_(2)IrCl_(6) reacts with triphenylphosphine in diethyleneglycol in an atmosphere of CO to give [IrCl(CO)(PPh_(3))_(2)]. (Atomic number of Ir=77) Which of the following statements is/are correct ? |
|
Answer» The IUPAC NAME of the complex is carbonylchloridobis (triphenylphosphine) iridium(I). |
|
| 31. |
The compound Na_(2)[Fe(CN)_(5)NO) is called (A) |
|
Answer» SODIUM pentacyanonitrosoniumferrate (II) |
|
| 32. |
The compound Na_(2)[Fe(CN_(5)NO] is called |
|
Answer» SODIUM nitroprusside |
|
| 33. |
The compound most reactive towards S_N1 reaction is |
|
Answer» `MeCOCH_2Cl`  Though (ii) contains +ve charge on OXYGEN. SINCE octet around each atom is complete the STRUCTURE II is more STABLE than I . |
|
| 34. |
The compound most likely to decolourize a solution of potassium permanganate is |
|
Answer» `CH_3CH_3` |
|
| 35. |
The compound K_(3)[Co(ONO)_(6)] is called |
|
Answer» potassium cobaltinitrite |
|
| 36. |
The compound K_(4)[Fe(CN)_(6)] is called |
|
Answer» POTASSIUM ferrocyanide |
|
| 37. |
The compound K_(3)[Co(NO_(2))_(6)] is called |
|
Answer» FISCHER's SALT |
|
| 38. |
The compound isomeric with acetone is: |
| Answer» Answer :A | |
| 39. |
The compound is not isomeric with diethyl ether |
|
Answer» butan-1-ol |
|
| 40. |
The compound is used as |
|
Answer» antiseptic |
|
| 41. |
The compound is used as |
|
Answer» ANTISEPTIC |
|
| 42. |
The compound insoluble in water is: |
|
Answer» `CaF_2` |
|
| 43. |
The compound insoluble in acetic aid is |
|
Answer» calcium oxide |
|
| 44. |
The compound insoluble in acetic acid is: |
|
Answer» CALCIUM oxide |
|
| 45. |
The compound insoluble in acetic acid is |
|
Answer» Calcium oxide |
|
| 46. |
The compound insoluble in acetic acid |
|
Answer» Calcium oxide `CAO, CaCO_(3) and Ca(OH)_(2)` are soluble in acetic acid. |
|
| 47. |
The compound insolubel in acetic acid is |
|
Answer» CALCIUM OXIDE |
|
| 48. |
The compound in which there is no lone pair on xenon atom is |
|
Answer» `XeF_6` |
|
| 49. |
The compound in which the number of d pi - p pi bonds are equal to those present in ClO_(4)^(-) |
|
Answer» `XeF_(4)` |
|