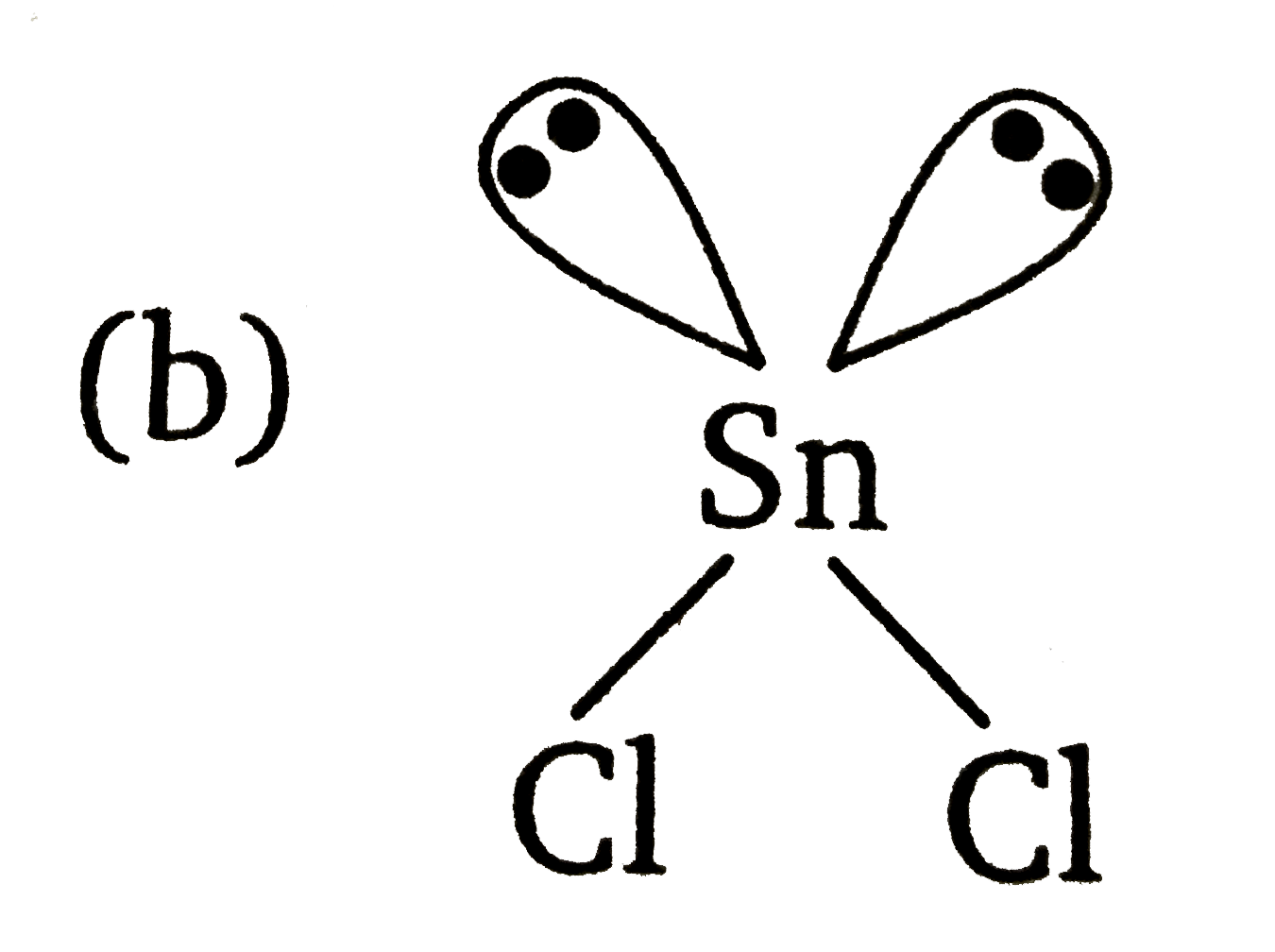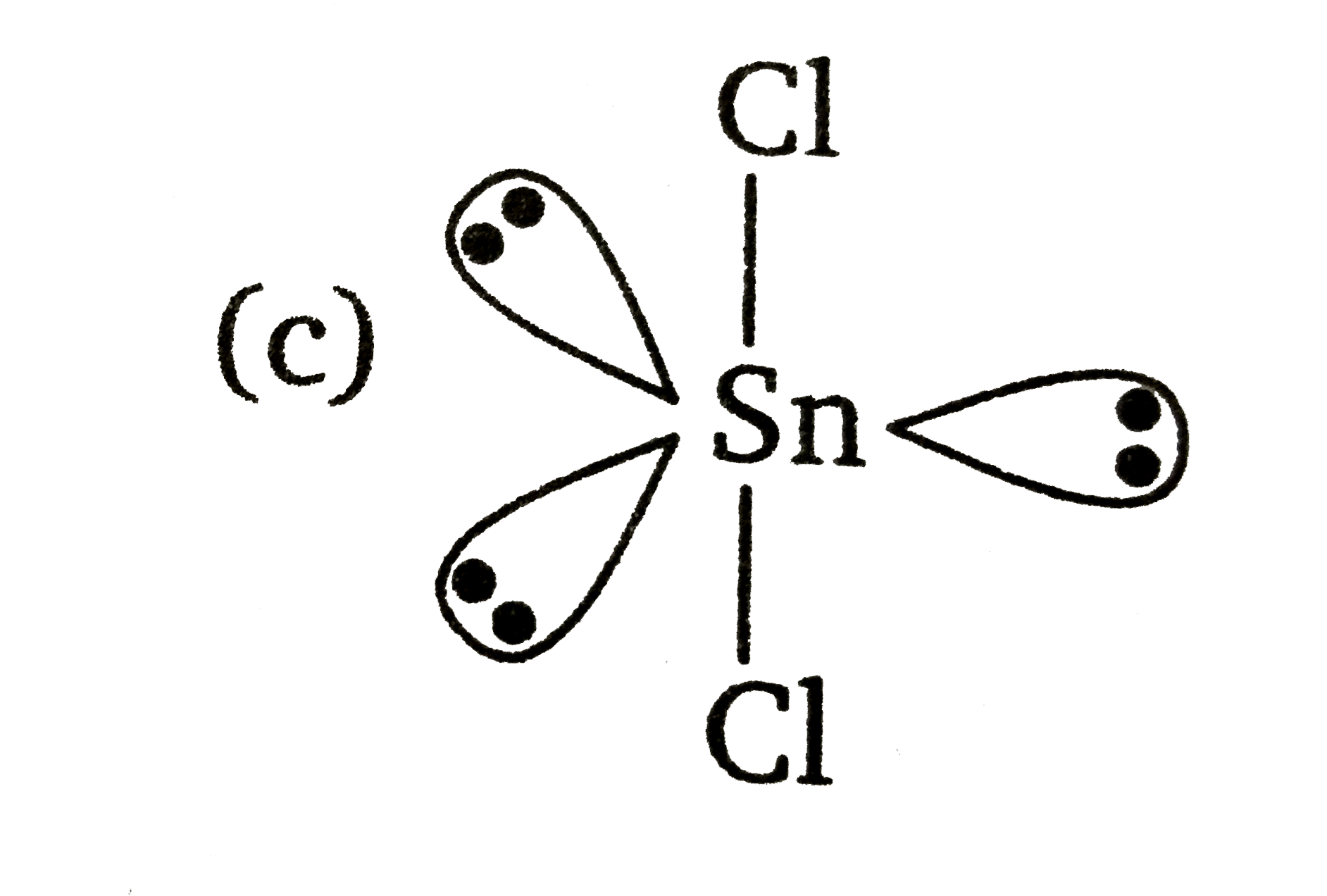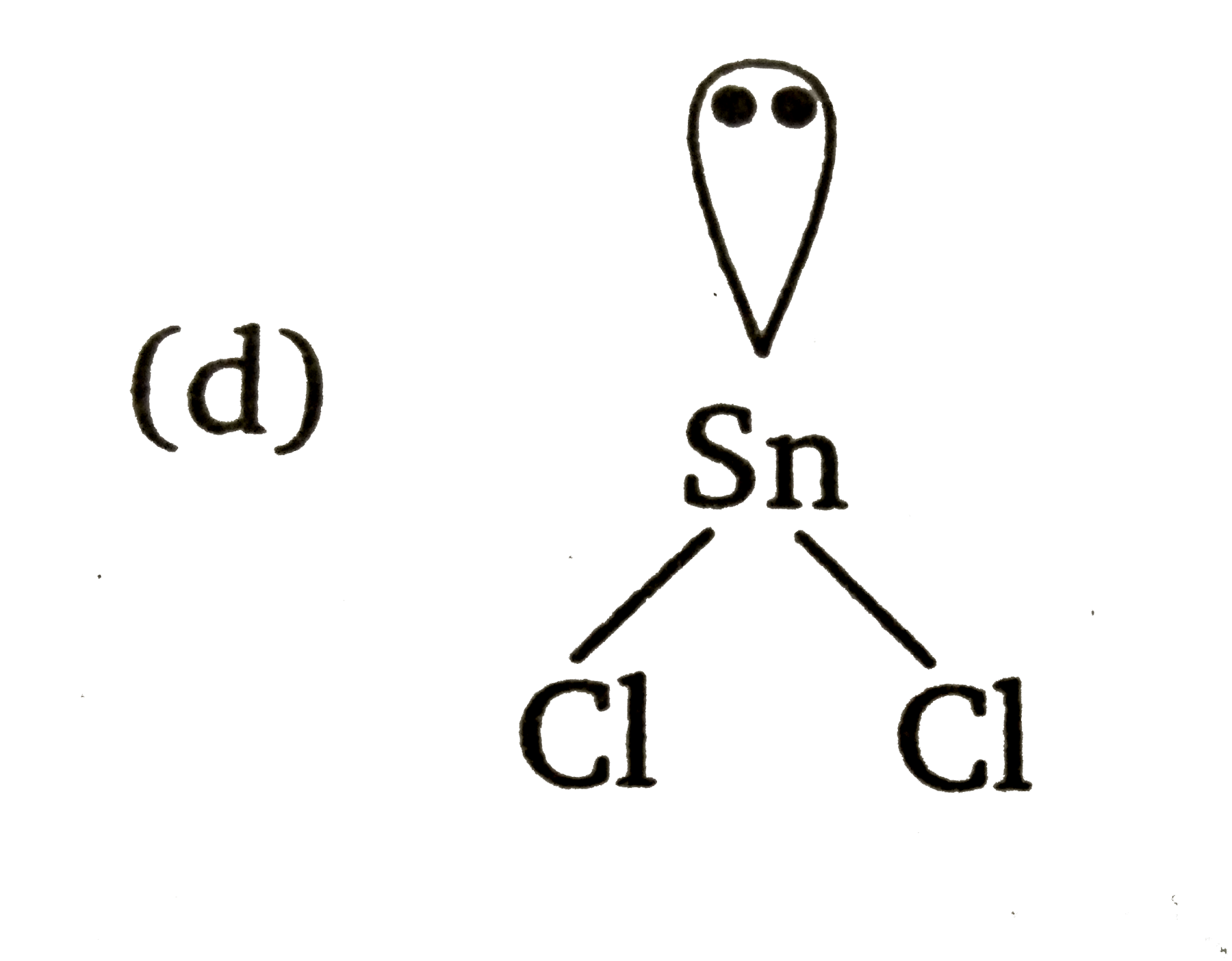Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 2. |
Theauto- reductionprocess is notusedin themetallurgyof |
|
Answer» Hg |
|
| 3. |
The electrophile involved in the sulphonation of benzene is : |
|
Answer» `SO_(3)` |
|
| 4. |
The attacking species in the aromatic sulphonation is: |
|
Answer» `SO_3H^+` |
|
| 5. |
The attacking reagent in electrophilic sulphonation of benzene is |
|
Answer» `SO_2` |
|
| 6. |
The attacking reagent in electrophilic sulphonation of benzene is : |
|
Answer» `SO_(4)^(2-)` |
|
| 7. |
The atoms the face centre is being shared by ……………………………… unit cells. |
|
Answer» 4 |
|
| 8. |
The atomicity of yellow phosphorous is ____ |
|
Answer» |
|
| 9. |
The atomicity of white Phosphorous is ‘x' and the P-hat(P)-P bond angle in the molecule is 'y'. What are 'x' and 'y' ? |
|
Answer» `X = 4 , y = 90^@` |
|
| 10. |
The atomicity of sulphur in orthorhombic sulphur is- |
|
Answer» 8 |
|
| 11. |
The atomic weights of two elements A and B are 20 and 40 respectively. Which of the following statements are correct for these two elements? |
|
Answer» x g of A contains y ATOMS which is EQUAL to atoms present in x g of B. `underset(1 mol)(20)A underset(1 mol)underset(40)B` Suppose 10 g of A = 1/2 mol of A = 1/2 mol of B = 20 g of B = 2x g of B Hence, XG of A and 2x g of B will contain same number of atoms. x L of A = yg Suppose, 11.2 mL of A = 1/2 mol of A = 10 g of A `=x/2` L of B at STP |
|
| 12. |
The atomicitiesof oxygen and sulphur are different. What is the ratio of the atomicities of sulphur to oxygen ? |
|
Answer» |
|
| 13. |
The atomic weights are expressed in terms of atomic mass unit. Which one of the following is used as a standard ? |
|
Answer» `.^(1)H_(1)` |
|
| 14. |
The atomic weights of carbon, nitrogen and oxygen are 12,14 and 16 respectively. Among the following pairs of gases, the pair that will diffuse at the same rate is : |
|
Answer» 1. CARBON DIOXIDE and NITROUS oxide |
|
| 15. |
The atomicweight of Fe is 56 The weightof Fe deposited fromFeCI_(3)solutionby passing0.6 faraday of electricity is |
|
Answer» 5.6 g `therefore` 3 MOLESOF electrondeposite = 1 moleof Fe `therefore`0.6 molesof electron deposite `=1/3xx0.6` moleof Fe `because` 1 moleof Fe =56 g (atomicweightof fe =56) `therefore`0.2 moleof Fe `=(56)/(1) xx0.2 =11.2 g` |
|
| 16. |
The atomic weight of noble gases is obtained by using the relationship |
|
Answer» ATOMIC WEIGHT=equivalent weightXvalency |
|
| 17. |
The atomic weight of Al is 27. When a current of 5 Faradays is passed through a solution of Al^(++) ions, the weight of al deposited is |
|
Answer» 27gm `E_(Al)=(27)/(3)=9` `W_(Al)=E_(Al)XX"No. of FARADAYS"=9xx5=45gm`. |
|
| 18. |
The atomic weight of Al is 27. When a current of 5 Faraday is passed through a solution of Al^(3+) ions, the wt. of Al depostited is : |
|
Answer» 27g |
|
| 19. |
The atomic weight of a metal (M) is 27 and its equivalent weight is 9, the formula of its chloride will be |
|
Answer» `MCl_3` |
|
| 20. |
The atomic sizesof Fe,Co and Ni are nearlysame, Explain with reason. Or Atomic size of 3dseries elements from chromium to copper is almost the same. Give reason. |
| Answer» Solution :As we MOVE from LEFT to right alonga TRANSITIONSERIES, the nuclear change increases which tends to decrease the size but the addition of ELECTRONS in thed-subshellincreases the screening effect which counter balances the effect of increased nuclear CHARGE. | |
| 21. |
The atomic sizes are not significantly different for the series of elements |
|
Answer» BI, Na, K, Pb |
|
| 22. |
The atomic reactor when used to generate electricity is termed breeder reactor. |
|
Answer» |
|
| 23. |
The atomic radius of strontium ( Sr ) is 215 pm and it crystallizes in FCC . Edge length of the cube is |
|
Answer» `430` PM |
|
| 24. |
The atomic radius of palladium is 1.375 A. The unit cell of palladium is a face-centred cube. Calculate the density of palladium. |
|
Answer» |
|
| 25. |
The atomic radius of Nb is closest to : |
| Answer» Solution :Due to lanthanoid CONTRACTION. | |
| 27. |
The atomic radius of an ion which crystallizes in fcc structure is 9/7 Å. Calculate the lattice constant. |
|
Answer» For FCC STRUCTURE, `r = a//(2sqrt2)` or lattice constant, = `9/7 xx 10^(-10) m xx 2 xx 1.4142 = 3.6 xx 10^(-10)` m. |
|
| 28. |
The atomic radius of Ag is closest to: |
|
Answer» Ni |
|
| 29. |
The atomic radius of a face centred cubic cell is: |
|
Answer» `a/2` |
|
| 30. |
The atomic radius of a body centred cubic cell is: |
|
Answer» `a/2` |
|
| 31. |
The atomic radius of 5d elements and 4d elements are nearly same due to….. |
| Answer» SOLUTION : LANTHANOID CONTRACTION | |
| 32. |
The atomic radius in a face-centred cubic cell is: |
|
Answer» `a/2` |
|
| 33. |
The atomic radii of Nb and Ta are comparable due to __________. |
| Answer» SOLUTION :LANTHANOID CONTRACTION | |
| 35. |
The atomic radii of __________ and __________ series of transition elements are comparable. |
| Answer» SOLUTION :SECOND, THIRD | |
| 36. |
The atomic radii in periodic table among elements from right to left |
|
Answer» DECREASES |
|
| 37. |
The atomic radii from Cr to Cu is almost identical because of |
|
Answer» Increasing NUCLEAR CHARGE from Cr to Cu |
|
| 38. |
The atomic orbitals are progressively filled in order of increasing energy. This principle is called: |
|
Answer» HUND's rule |
|
| 39. |
The atomic orbital not allowed in quantum theory is |
|
Answer» 2p |
|
| 40. |
The atomic numbers of the metallic and non-metallic elements which are liquid at room temperature respectively are: |
|
Answer» 55,87 |
|
| 41. |
The atomic numbers of other elements which lie in same group as the tenth element in the periodic table are |
|
Answer» 18, 32, 54, 86 |
|
| 42. |
The atomic numbers of chromium and iron are 24 and 26 respectively. Which one of the following complexes exhibits paramagnetic character due to electronic spin? |
|
Answer» `[Fe(CO)_5]` |
|
| 43. |
The atomic number of Sn is50. The shape of gaseous SnCl_(2) molecule is |
|
Answer» `Cl-Sn-Cl` 
|
|
| 44. |
The atomic number of Ni and Cu are 28 and 29 respectively. The electronic configuration 1s^(2)2s^(2)2p^(6)3s^(2)3p^(6)3d^(10) represents |
|
Answer» `Cu^(+)` `Ni-[AR]3d^(8)4s^(2), "" Ni^(2+)-[Ar]3d^(8)` `Cu-[Ar]3d^(10)4s^(1), ""Cu^(+)-[Ar]3d^(10)` `Cu^(2+)-[Ar]3d^(9)` |
|
| 45. |
The atomic number of elements lie in the range of |
|
Answer» 88 to 101 |
|
| 46. |
The atomic number of element having pseudo inert gas configuration in it's atomic state is |
|
Answer» 46 |
|
| 47. |
Which element has pseudo inert gas electronic configuration ? |
|
Answer» 46 |
|
| 48. |
The atomicnumber of cerium (Ce) is 58. The correct electronic configuration of Ce^(3+) ion is |
|
Answer» `[Xe]4F^(1)` `Z =57` ( La) is `[Xe]5d^(1)`( electron 5d in place of expected 4f) But in `Z= 58` ( Ce ) , electron enters 4f and `5d^(1)` also shifts to 4f. Hence, electronic configuration of `Ce( 58) ` is `[Xe] 4f^(2) 5d^(0) 6s^(2)`. `:. Ce^(3+) = [Xe]4f^(1)` |
|
| 49. |
The atomic number of cerium (Ce) is 58. The correct electronicconfiguration of Ce^(3+) ion is |
|
Answer» `[Xe]4f^(1)` 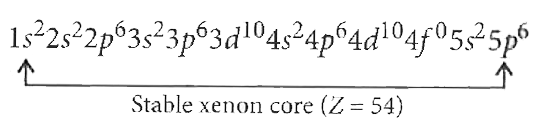 For Z = 56, it will be `[Xe]6s^(2)` In LANTHANUM (Z = 57), electrons should enter in 4f but to maintain stable xenon core it will go into 5d(DIFFERENCE is little in between 4f and 5d.) But in cerium (Ce, Z = 58), ELECTRON enters into 4f. `therefore""Ce^(3+)=[Xe]4f^(1)` |
|