Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The acidic, basic or amphoteric nature of Mn_(2)O_(7), V_(2)O_(5) and CrO are respectively |
|
Answer» Acidic, acidic and basic |
|
| 2. |
The acid which reduces Tollen's reagent is |
|
Answer» ACETIC acid |
|
| 3. |
The acidic, basic or amphoric nature of Mn_(2)O_(7), V_(2)O_(5) and CrO are respectively. |
|
Answer» acidic , acidic and basic |
|
| 4. |
The acid which is obtained from plant |
|
Answer» propionic ACID |
|
| 5. |
The acid which forms two series of salts is |
|
Answer» `H_(3)PO_(4)` |
|
| 6. |
The acid which forms two series of salts is : |
| Answer» Answer :B | |
| 8. |
The acid which do not contain carboxylic acid group is |
|
Answer» GLUTARIC acid 
|
|
| 9. |
The acid which does not form an anhydride when treated with P_2O_5 is |
|
Answer» FORMIC ACID |
|
| 10. |
The acid which cannot be kept in glass bottles is |
|
Answer» HF |
|
| 11. |
The 'acid value' of an oil or fat is measured in terms of weight of |
|
Answer» `NH_4 OH` |
|
| 12. |
The acid used in soft drinks is: |
|
Answer» `H_3PO_4` |
|
| 13. |
The acid used in lead storage battery is:- |
|
Answer» `H_(2)SO_(4)` |
|
| 14. |
The acid used in lead storage battery is: |
|
Answer» NITRIC acid |
|
| 15. |
The acid that results when a base accepts a proton is called |
|
Answer» CONJUGATE BASE of the ACID |
|
| 16. |
The acid used for etching the glass is : |
|
Answer» SULPHURIC ACID |
|
| 17. |
The acid that forms primary, secondary and tertiary phosphates is |
|
Answer» `H_(3)PO_(2)` |
|
| 18. |
The acidstrength of saturated aliphaticcarboxylicacids dependsmainlyupon the inductive effectof substitutent and its position with respect to the COOH group. Theacidstrenghtof aromaticcarboxylicacids, one the other handdepends upon boththe inductiveand theresonanceaffect of thesubsistuents . Theirrends are , however, similarto thoseof aliphaticacidsin the followingtwo differneces. (a)Theacidsweakingeffectof electrondonatingsubstituentsand acid strenghteffectof the electron- withdrawingsubstituents is more pronoucedat p - positionthat at m - posititon. (b) Due to ortho effect o-substited benzoicacidsare usallystrongerthan benzoic andacidregardlessof thenatureof substituent whetherelectrondonatingdonatingor electronwithdrawing. Amongthe followingthe strongestacid is : |
|
Answer» `CH_(3)COOH` |
|
| 19. |
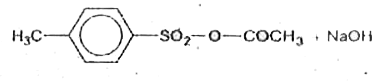
The acidstrength of saturated aliphaticcarboxylicacids dependsmainlyupon the inductive effectof substitutent and its position with respect to the COOH group. Theacidstrenghtof aromaticcarboxylicacids, one the other handdepends upon boththe inductiveand theresonanceaffect of thesubsistuents . Theirrends are , however, similarto thoseof aliphaticacidsin the followingtwo differneces. (a)Theacidsweakingeffectof electrondonatingsubstituentsand acid strenghteffectof the electron- withdrawingsubstituents is more pronoucedat p - positionthat at m - posititon. (b) Due to ortho effect o-substited benzoicacidsare usallystrongerthan benzoic andacidregardlessof thenatureof substituent whetherelectrondonatingdonatingor electronwithdrawing. Whichof the followingis obtainedwhen 4-methyl - benzene sulphonic acid is hydrolysed with excessof sodium acetate . |
|
Answer»
|
|
| 20. |
The acidstrength of saturated aliphaticcarboxylicacids dependsmainlyupon the inductive effectof substitutent and its position with respect to the COOH group. Theacidstrenghtof aromaticcarboxylicacids, one the other handdepends upon boththe inductiveand theresonanceaffect of thesubsistuents . Theirrends are , however, similarto thoseof aliphaticacidsin the followingtwo differneces. (a)Theacidsweakingeffectof electrondonatingsubstituentsand acid strenghteffectof the electron- withdrawingsubstituents is more pronoucedat p - positionthat at m - posititon. (b) Due to ortho effect o-substited benzoicacidsare usallystrongerthan benzoic andacidregardlessof thenatureof substituent whetherelectrondonatingdonatingor electronwithdrawing. Whichof the followingacidshasthe smallest, dissociationconstant ? |
|
Answer» `CH_(3)CHFCOOH` |
|
| 21. |
The acid that does not contain carboxylic group is : |
|
Answer» ACETIC acid |
|
| 22. |
The acid strength of active methylene group in (I) CH_(3)COCH_(2)COOC_(2)H_(5)(II) CH_(3)COCH_(2)COCH_(3) (III) C_(2)H_(5)OO"CC"H_(2)COOC_(2)H_(5), decreases as : |
|
Answer» `IgtIIIgtII` |
|
| 23. |
The acid strength of active methylene group in : (i) CH_(3)COCH_(2)COOC_(2)H_(5) (ii)CH_(3)COCH_(2)COCH_(3)(iii) C_(2)H_(5)OOCCH_(2)COOC_(2)H_(5) decreases as : |
|
Answer» (i) `GT` iii `gt` II |
|
| 24. |
Write the names of the following: (i) CH_(3)CH_(2) - C -= CH (ii) CH_(3) CH_(2) OH (iii) CH_(3)COCH_(3). |
|
Answer» `igt iii gt ii` which increases the ACIDITY. Esters (iii) are LESS ACIDIC than ketones (i) because the enolate ion produced from an ester is less stable than that produced from a ketone. Hence, `CH_(3)-overset(O)overset("||")C-underset("(ii)")(CH_(2))-overset(O)overset("||")C-CH_(3) gt CH_(3)-overset(O)overset("||")C-underset("(i)")(CH_(2))-overset(O)overset("||")C-OC_(2)H_(5)gtC_(2)H_(5)O-overset(O)overset("||")C-underset("(iii)")(CH_(2))-overset(O)overset("||")C-O-C_(2)H_(5)` |
|
| 25. |
The acid strength of active methylene group in A) CH_3COCH_3COOC_2H_5 B) CH_3COCH_2COCH_3 C) C_2H_5 OO C CH_2COOC_2H_5 decreases as |
|
Answer» ` a gt c gt B` `H_3C -overset(O)overset(||)C-CH_2-overset(O)overset(||)C-CH_3 gt H_3C-overset(O)overset(||)C-CH_2-overset(O)overset(||)C-OC_2H_5 gt H_5C_2-O-overset(O)overset(||)C-CH_2-overset(O)overset(||)C-OC_2H_5` Because ester grop has O-R group which decreases electron WITHDRAWING nature of carbonyl group. |
|
| 26. |
The acid showing salt like structure in aqueous solution is: |
|
Answer» ACETIC ACID |
|
| 27. |
The acid rain does not contian |
|
Answer» SULPHURIC acid |
|
| 28. |
The acid obtained when P_4O_6 reacts with water is |
|
Answer» `H_(3)PO_(2)` |
|
| 29. |
The acid obtained by the distillation of ants is called as |
|
Answer» FORMALIN |
|
| 30. |
The acid present in tomatoes is |
|
Answer» LACTIC acid |
|
| 31. |
The acid ionization constantfor Zn^(2+)+H_(2)O hArr (OH^(+)) + H^(+) is 1.0 xx 10^(-9).Calculatethe pH of 0.0010M solution of ZnCl_(2) . Also calculate basic dissociation constant of Zn(OH)^(+) |
|
Answer» Solution : `Zn^(2+) +H_(2)O hArrZn(OH)^(+) + H^(+) ` ` :.[H^(+)] = C.h =Csqrt((K_(H))/(C)) = sqrt((K_(H))/(C))` ` = sqrt((K_(W))/(K_(b).C))"" [("where "K_(b) " is basic dissociation CONSTANT "),("of " Zn(OH) "i.e ., " Zn(OH)^(+) hArr Zn^(2+) +OH^(-))]` We know`Zn^(2+)`and `Zn(OH)^(+)`are conjugate acid and base. ` :. K_(a) xx K_(b) = 10^(-14)` or `K_(b) = (10^(14))/(10^(-9)) = 10^(-5)` Now, `[H^(+)] = sqrt((10^(-14))/(10^(-5)xx0.001)) = sqrt(10^(-12)) = 10^(-6)` pH = 6 |
|
| 32. |
The acid in which O - O bonding is present, is |
|
Answer» `H_2S_2O_3` `HO-underset(O)underset(|)overset(O)overset(||)(S)-O-O-underset(O)underset(||)overset(O)overset(||)S-OH` This is known as Marshall.s acid. It contains O - O BOND. |
|
| 33. |
The acidhydrolysis whichof the following willgivesacetic acid(1) CH_(3) -CN (2)CH_(3) - CH_(2) - NO_(2) (3)CH_(3) - underset(CH_(3))underset(|)(CH) -NO_(2) (4)C_(2)H_(5)-NO_(2) |
|
Answer» only 1,3 ` CH_(3) - CH_(2)- NO_(2) overset(H_(3) O^(+)) toCH_(3) - CH_(2) - COOH + NH_(2) - OH` |
|
| 34. |
The acid in gastricjuice is |
|
Answer» ACETIC ACID |
|
| 35. |
The acid hydrolysis of ester is/are |
|
Answer» FIRST ORDER reaction |
|
| 36. |
The acid hydrolysis of ester is: |
|
Answer» FIRST ORDER REACTION |
|
| 38. |
The acid having the highest pK_(a) value among the following is |
|
Answer» HCOOH |
|
| 39. |
The acid hydrolysis of ester is |
|
Answer» I order reaction Rate= [Ester] `[H_(2)O]` In this rate depends on concentration of ester but INDEPENDENT on `H_(2)O` . Hence it is pesudo unimolecular reaction . In this two molecular involved hence it is bimolecular . |
|
| 41. |
Among the following acids which has the lowest pK_a value? |
|
Answer» `CH_3COOH` |
|
| 42. |
The acid formed when propyl magnesium bromide is treated with CO_2 is : |
|
Answer» `C_3H_7COOH` |
|
| 43. |
The acid formed when propyl magnesium bromide is treated with carbondioxide and followed by hydrolysis is |
|
Answer» `C_3H_7COOH` |
|
| 44. |
The acid dissociation constant for Al(H_2 O)_(6)^(3+) is 1.4 xx10^(-5) . It suggests |
|
Answer» `H_2 O` molecules in the hydrated cation are much STRONGER proton donors than free SOLVENT water molecules pHofsolutionwillalwaysbelessthan7. |
|
| 45. |
The acid D obtained through the following sequence of reactions is: C_(2)H_(5)Br overset("Alc. KOH")to A underset(C Cl_(4))overset(Br_(2))to B underset(("excess"))overset(KCN)toC overset(H_(3)O^(+))toD |
|
Answer» succinic ACID |
|
| 46. |
The acid dissociation constants of the following acids are given as under : From this data, the following observations can be made . Mark the correct statements for above mentioned compounds. (i)The above variation in acidities of the above acids are due to inductive effect only. (ii)The above variation are both to inductive and resonance effects (iii)Inductive effect varies sharply with distance (iv)-I effect of chlorine is not much . |
|
Answer» `i&ii` |
|
| 47. |
The acid formed when iodine reacts with conc. HNO_3 is : |
|
Answer» Hydroiodic ACID |
|
| 48. |
The acid catalysed hydrolysis of an organic compound A at 30^(@)C has a time for half change of 100 min, when careied out in a buffer solution at pH=5, and 10 min. when carried out at pH=4. Both times of half change are independent of the initial concentration of A. If the rate constant K is given by (-d[A])/(dt)=K[A]^(a)[H^(+)]^(b) what are the values of a and b? |
| Answer» Answer :A | |
| 49. |
The acid catalysed hydrolysis of ethyl acetate is first order with respect to ester and rate constant for the hydrolysis reaction is 0.693 sec^(-1). Determine time (in sec) required for 93.75% hydrolysis of ester. ["In" 2= 0.693] |
|
Answer» |
|