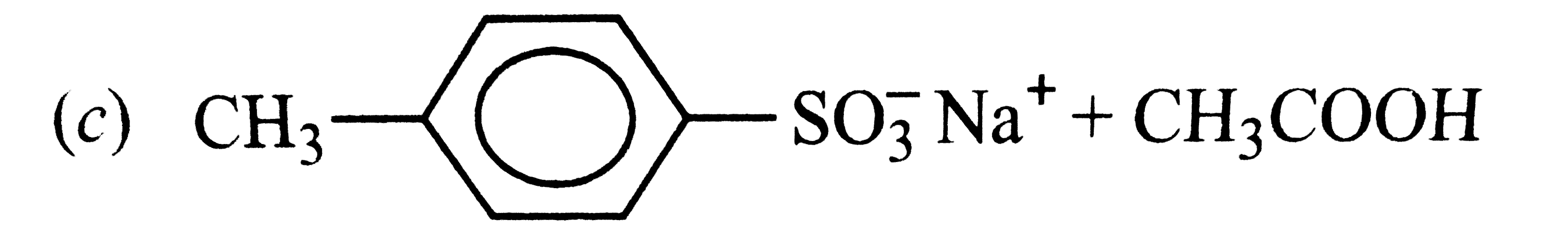Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The activation energy for a reaction at the temperature TK was found to be 2.303 RT J mol^(-1) . The ratio of the rate constant to Arrhenius factor is |
|
Answer» `10^(-1)` Putting the values of `E_(a)` and dividing by 2.303 on both sides , we get , log k = log A - 1 , log k - log A = -1 log `((k)/(A)) = -1 implies ` TAKING antilog `((k)/(A)) = 10^(-1)` . |
|
| 2. |
The activation energy for a reaction is 9.0 K cal/mol . The increase in the rate constant when its temperature is increased from 298 K to 308 K is |
|
Answer» 0.63 log `(k_(2))/(k_(1)) = (9.0 XX 10^(3))/(2.303 xx 2) [(308 - 298)/(308xx 298)]` `(k_(2))/(k_(1)) = 1.63 , K_(2) = 1.63 K_(1) , (1.63 k_(1) - k_(1))/(k_(1))xx 100 = 63.0` % |
|
| 3. |
The activation energy for a reaction is 9 kcal/mol. The increase in late (XI 'slant when its temperature is increased from 298K to 308 K is(R = 2 cal K^(-1)mot^(-1), 10^(0.213) = 1.63) |
|
Answer» 0.75 |
|
| 4. |
The activation energy for a reaction is 9.0 Kcal//mol. The increase in the rate constant when its temperature is increased from 298 K to 308 K is: |
| Answer» ANSWER :D | |
| 5. |
The activation energy and enthalpy of chemisorption of oxygen on a metal surface is 37.3kJ mol^(-1) and -72.1kJ mol^(-1) At a certain pressure, the rateconstant for chemisorption is 1.2xx10^(-3) at 3198 K . What will be the value of the rate constant at 308K ? |
|
Answer» `7.6 xx 10^(-4)s^(-1)` `ln.(K_(2))/(K_(1))=(E_(a))/(R)((1)/(T_(1))-(1)/(T_(2)))`or `lnk_(2)+(E_(a))/(R)((1)/(T_(1))-(1)/(T_(2)))` Now,`T_(1)=318K,E_(a)=37.3kJmol^(-1)` and `T_(2)=308K` `logk_(2)=log(1.2xx10^(-3))+(37300)/(8.31xx2.303)((1)/(318)-(1)/(308))=-2.9208-(37300xx10)/(8.31xx2.303xx318xx308)` `=-2.9208-0.1989` `=k_(2)=7.6xx10^(-4)s^(-1)` |
|
| 6. |
The activation energyfor a reaction at the temperature T K was found to be 2.303 RT J mol^(-1) . The ratio of the rate constant to Arrhenius factor is : |
|
Answer» `10^(-1)` `k = Ae^(-E_(a)//RT) ` or log k = log `A-(Ea)/(2.303RT)` `E_(a)= 2.303 RT` `:. ` log k = log A `-(2.303RT)/(2.303RT)` log k = log A-1 or log k - log A=-1 log `(k)/(A) =-1` `(k)/(A) = 10^(-1)` |
|
| 7. |
The activation energy for a first-order reaction is 104.5 kJ/mole and the factor A in the Arrhenius equation is 5 xx 10^15 s^(-1). Find the temperature at which the half-life period of the reaction is 1 minute. |
| Answer» SOLUTION :`75^@C` | |
| 8. |
The activation energies for the two reactions are Ea and Ea^(') with E_(a)gtE_(a)^('). If the temperature of the reaction system is increased from T_(1) to T_(2). Predict which alternative is correct. (k^(')) are the rate constants at highest temperature. |
|
Answer» `(k_(1)^('))/(k+(1)) = (k_(2)^('))/(k_(2))` `k=Ae^((-Ea)/(RT))` This means that more is the activation energy, lesser is the RATE constant. Since `E_(a) gt E_(a)^(')` `THEREFORE k_(1) lt k_(2)` and `k_(1)^(')` and `k_(2)^(')` |
|
| 9. |
The activation energies of two reactions are E_1" and "E_2 (E_1 gt E_2). If the temperature of the system is increased from T_1" to "T_2, the rate constant changes from k_1" to "k'_1 in the first reaction and k_2" to "k'_2 in the second reaction. Predict which of the following expressions is/are incorrect? |
|
Answer» `(k'_1)/(k_1)=(k'_2)/(k_2)` `[thereforeT_2 gtT_1]` `log(k_2.)(k_2)=(E_2)/(2.303R)((T_2-T_1)/(T_1T_2))`…(ii) As, `E_1 gt E_2` From (i) and (ii), we GET `log(k_1.)/(k_1) gt log (k_2.)/(k_2) implies (k_1.)/(k_1) gt (k_2.)/(k_2)` |
|
| 10. |
The activation energies of two reactions are E_(1) and E_(2) (E_(1) gt E_(2)) . If the temperature of the system is increased from T_(1) to T_(2) , the rate constant of the reaction changes from k_(1) to k_(1) in the first reaction and k_(2) to k_(2) in second reaction predict which of the following expression is correct ? |
|
Answer» `(k_(1))/(k_(1))= (k_(2))/(k_(2))` log`(k_(2))/(k_(2))=(E_(2))/(2.303R)[(T_(2)-T_(1))/(T_(1)-T_(2))]` SINCE `E_(1) gt E_(2)` `("log"(k_(1))/(k_(1)))/("log"k_(2)/(k_(2)))gt1` or ` (k_(1))/(k_(1))gt (k_(2))/(k_(2))` |
|
| 11. |
The activation energies of the forward and backward reactions in the case of a chemical reaction are 37.5 and 51.3 kJ/mole respectively The reaction is |
|
Answer» ENDOTHERMIC |
|
| 12. |
The activation energies of two reactions are E_a andE_awithE_agt E_a if the temperature of the reacting systems is increased from T_1toT_2, predict which alternative is correct k are rate constants at higher temperature. Assume A being same for both the reactions: |
|
Answer» `k_1/k_2=k_2/k_2` |
|
| 13. |
The action of nitrous acid on an aliphatic primary amine gives |
|
Answer» ALCOHOL |
|
| 14. |
The action of sodium on alkyl halide to form an alkane is called |
|
Answer» GRIGNARD reaction |
|
| 15. |
The action that does not form an amine complex with excess of ammonia is: |
| Answer» Answer :A | |
| 16. |
The action of nitrous acid on ehtyl amine gives |
|
Answer» Nitroethane |
|
| 17. |
The action of nitrous acid on a primary amine gives : |
|
Answer» NITRO alkane |
|
| 18. |
The action of HCI on silver nitrate produces: |
|
Answer» A PRECIPITATE of AgCl |
|
| 19. |
The action of boiling water on actinoids gives |
|
Answer» OXIDES and hydroxides |
|
| 20. |
The action of enzyme is inhibited during the fermentation once the concentration of alcohol exceeds ……. |
| Answer» Solution :14 percent | |
| 21. |
The action of Bleaching powder on ethyl alcohol gives |
|
Answer» `C CCl_(4)` `C_(2)H_(5)OH+(O) to CH_(3)CHO+H_(2)O` `CH_(3)CHO+3Cl_(2) to C Cl_(3) CHO+3HCl` `2C Cl_(3)CHO+CA(OH)_(2)) to Ca(HCOO)_(2)+2CHCl_(3)` |
|
| 22. |
The action of halogen acids on an ether, has the following order of reactivity: |
|
Answer» `HCl GT HBr gt HI` |
|
| 24. |
The actinoids exihibits more number of oxidation states in general than the lanthanoids.This is because |
|
Answer» the 5f ORBITALS extend farther from the nucleus than the 4f orbitals |
|
| 25. |
The action of aniline with benzoyl chloride in the presence of aqueous NaOH gives : |
|
Answer» BENZAMIDE |
|
| 26. |
The actinoids exhibit more number of oxidation states in general than the lanthanoids. This is because |
|
Answer» the 5f orbitals extend further from the NUCLEUS than the 4f orbitals So actinoieds EXHIBIT more number of oxidation states in gerneral than the lanthnoids. |
|
| 27. |
The actinoid elements which show the highest oxidation state of +7 are |
| Answer» Solution :Np, Pu Am | |
| 28. |
Theactinoidelements whichshow thehighestoxidationstate of +7are |
| Answer» Answer :A | |
| 29. |
The actinide element which can exhibit +7 oxidation state in its compounds |
|
Answer» THORIUM |
|
| 30. |
The actinide contraction differs from lanthanide contraction? Explain. |
| Answer» Solution :Lanthanide CONTRACTION refers to the gradual DECREASE in the size of the lanthanoids and their trivalent ions, `( Z = 58 ` to `71)` whereas actinoid contraction refers to the gradual decrease in the size of the actinoids or their ions `( M^(3+)` or `M^(4+)) ( Z = 90 ` to `103 )`. The differ in the FACT that int he actinoid series, the contraction is greater from element to element DUE to poor shielding by 5f electrons thanby 4f electrons. | |
| 31. |
The acidity of phenol is due to the ____of its anion. |
| Answer» SOLUTION :RESONANCE STABILIZATION | |
| 32. |
The acidic strength of the given compounds follows the order I. CH_(3)-CH=CH-overset(O)overset(||)C-OH II. CH_(3)-overset(..)underset(..)O-CH=CH-overset(O)overset(||)C-OH III. CH_(3)-CH_(2)-overset(O)overset(||)C-OH |
|
Answer» `IIgtIIIgtI` |
|
| 33. |
The acidic strength of saturated aliphatic carboxylic acids depends mainly upon the inductive effect of the substituent and its position w.r.t. the -COOH group. Whereas electron-donating substituents tend to decrease, electron-withdrawing substituents tend to increase the acid strength. the acid weakening effect of electron donating substituents and acid-strengthening effect of the electron-withdrawing substituents is more pronounced at p-position than that at m-position. Due to ortho effect, o-substituted benzoic acids are usually stronger than benzoic acid regardless of the nature of substituent whetehr electron-donating or electron-withdrawing Q. The pK_(a) of acetylsalicylic acid (aspirin) is 3.5 The pH of gastric juice in human stomach is about 2-3 and pH in the small intestine is about 8. Aspirin will be |
|
Answer» Unionized in the SMALL intestine and in the stomach |
|
| 34. |
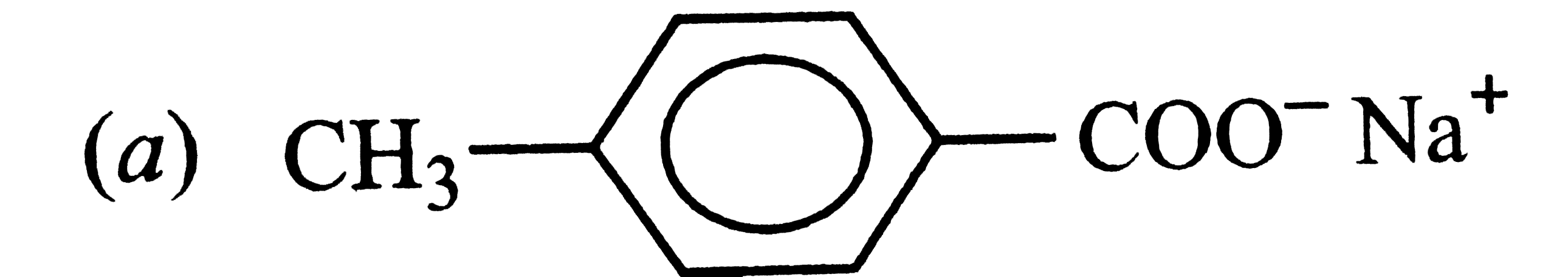
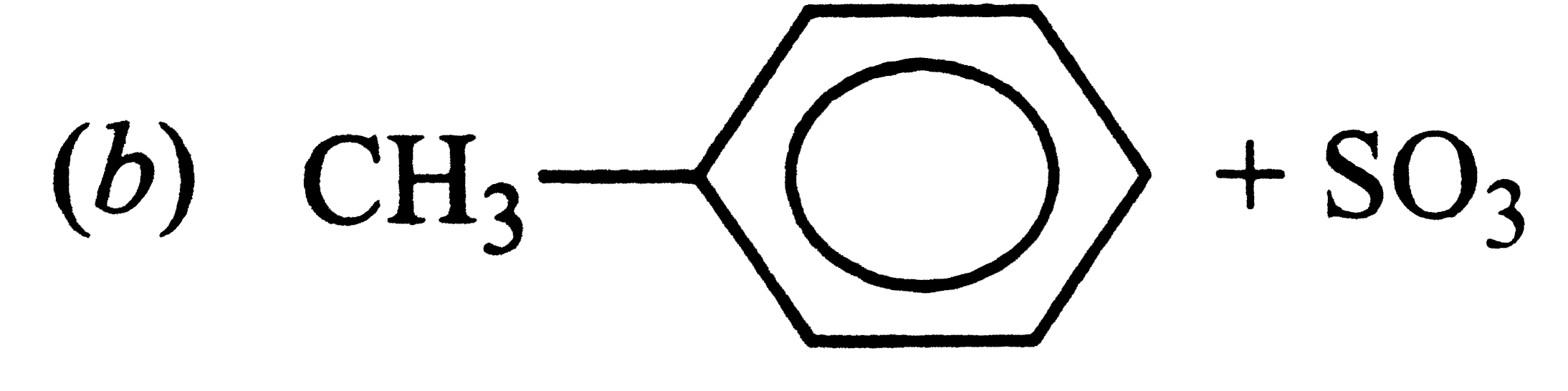
The acidic strength of saturated aliphatic carboxylic acids depends mainly upon the inductive effect of the substituent and its position w.r.t., the -COOH group. Whereas electron donating substituents tend to decrease, electron withdrawing substituents tend to increase the acid sttrength. the acidic strength of aromatic carboxylic acids, on the other hand, depends upon both the inductive and the resonance effect of the substituents Q. Which of the followig is obtained when 4-methylbenzenesulphonic acid is hydrolysed with excess of sodium acetate? |
|
Answer»
`4-Me-C_(6)H_(4)-SO_(3)H+CH_(3)COONa to 4-Me-C_(6)H_(4)SO_(3)^(-)Na^(+)+CH_(3)COOH` |
|
| 35. |
The acidic strength of saturated aliphatic carboxylic acids depends mainly upon the inductive effect of the substituent and its position w.r.t., the -COOH group. Whereas electron donating substituents tend to decrease, electron withdrawing substituents tend to increase the acid sttrength. the acidic strength of aromatic carboxylic acids, on the other hand, depends upon both the inductive and the resonance effect of the substituents Q. Among the following, the strongest acid is |
|
Answer» `CH_(3)COOH` |
|
| 36. |
The acidic strength of oxyacids follow the order |
|
Answer» `HCIO_3gtHCIO_2gtHOCl` |
|
| 37. |
The acidic strength decreases in the order : HCl gt H_(2)S gt PH_(3). Explain. Or The acidic strength of compounds increases in the order : PH_(3) lt H_(2)S lt HCl. |
|
Answer» Solution :The strength of an acid depends upon the stability of the anion (i.e., conjugate base) it gives after release of a proton. Now the disssociation of `PH_(3), H_(2)S` and HCl in aqueous solution occurs as FOLLOWS : `{:(PH_(3)(g)+aq rarr H^(+) (aq) + H_(2)P^(-) (aq), K_(a) = 1.6 xx 10^(-29)),(H_(2)S(g) + aq rarr H^(+) (aq) + HS^(-)(aq), K_(a)=1.6 xx 10^(-7)),(HCl(g)+aq rarr H^(+) (aq) + Cl^(-)(aq), K_(a) = 1 xx 10^(10)):}` Since the ELECTRONEGATIVITY decreases in the order : `Cl gt S gt P`, therefore, stability of the conjugate bases decreases in the same order, i.e., `Cl^(-) gt HS^(-) gt H_(2)P^(-)`, In other words, acidity of the corresponding acids decreases in the same order, i.e., `HCl gt H_(2)S gt PH_(3)`. |
|
| 38. |
The acidic organic compound, which does not have carboxylic function group is … |
| Answer» Solution :Picric acid | |
| 39. |
The acidic strength of HF,HCl, HBr, HI depends on |
|
Answer» ELECTRO negativity difference |
|
| 40. |
The acidic nature of carboxylic acids is due to : |
|
Answer» High degree of ionisation of the ACID |
|
| 41. |
The acidic nature of carboxylic acid is due to |
|
Answer» HIGH DEGREE of ionisation of acid |
|
| 42. |
The acidic dehydration method is not souitble for converting a tertiary alcohol into ether. Justify. |
| Answer» SOLUTION :TERTIARY alcohols are dehydrated to alkenes and ETHERS are formed in negligible AMOUNTS. | |
| 43. |
The acidic hydroysis of ether (X)shown below is fastest when : |
|
Answer» one PHENYL group is REPLACED by a methyl group |
|
| 44. |
The acidic nature of carboxylic acid is due to : |
|
Answer» Higher DEGREE of ionisation of theacid |
|
| 45. |
The acidiccharacterof phenol is due to __________ . |
|
Answer» greaterresonance STABILIZATION of PHENOXIDEION overphenol. |
|
| 46. |
The acidic character of phenol is due to |
|
Answer» GREATER RESONANCE stabilization of phenoxide ion over phenol |
|
| 47. |
The acidic character of glycine is due to ..................group and the basic character is due to ..........group. |
| Answer» SOLUTION :`-OVERSET(N)H_(3),-COO^(-)` | |
| 48. |
The acidic character of fatty acids…… with an increase in molecular weight: |
|
Answer» INCREASES |
|
| 49. |
The acidic aqueous solution of ferrous ion forms a brown complex in the presence of NO_(3)^(-), by the following two steps : [Fe(H_(2)O)_(6)]^(2+) + NO_(3)^(-)+H^(+) rarr .... + [Fe(H_(2)O)_(6)]^(3+) [Fe(H_(2)O)_(6)]^(2+) + .... rarr ..... + H_(2)O Complete and balance the equation. |
| Answer» | |