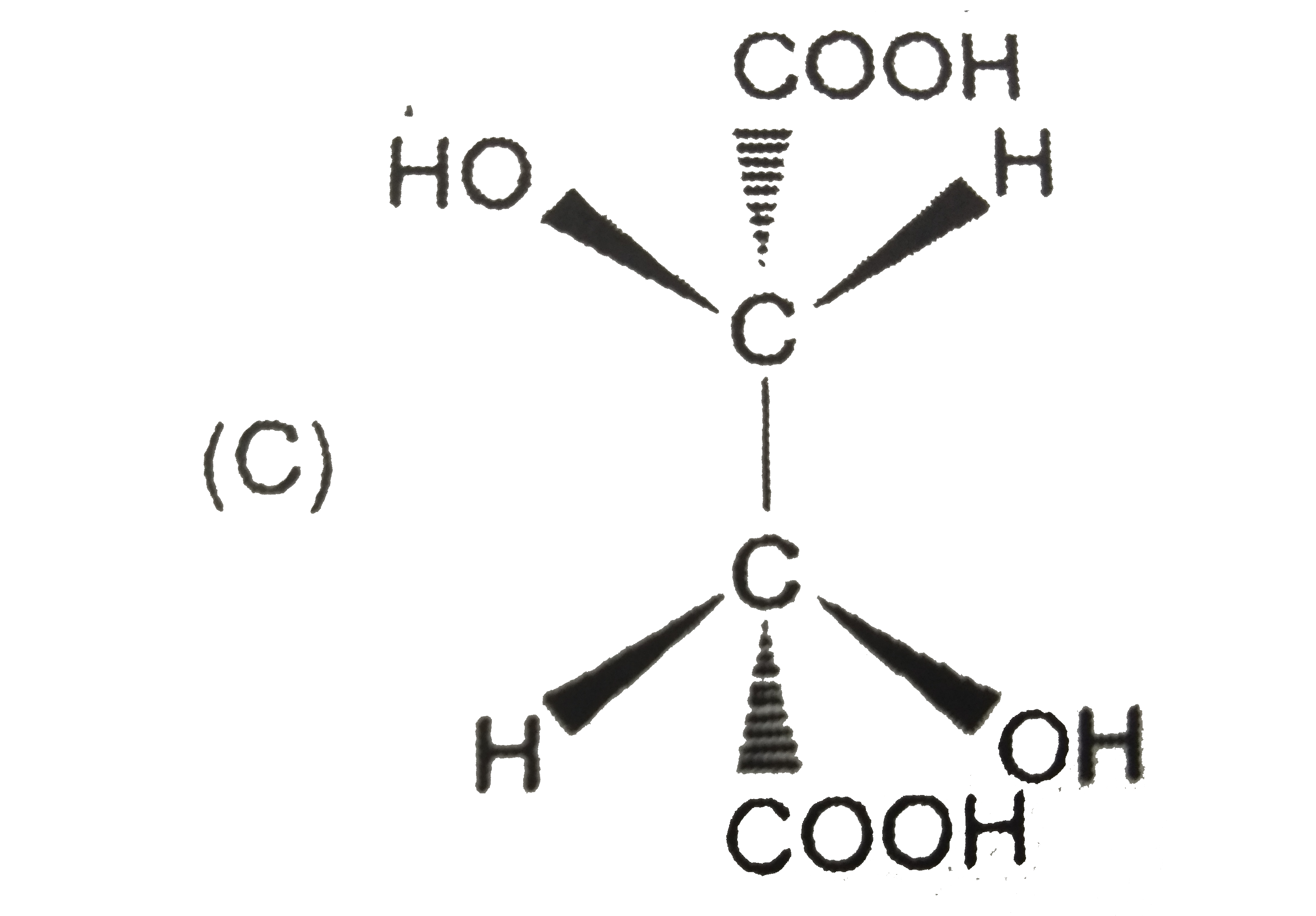Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
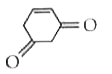
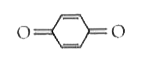
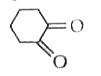
Tautomerism is not exhibit by: |
|
Answer»
|
|
| 2. |
Tautomerism is exhibited by - |
|
Answer»
|
|
| 4. |
Tautomerism is exhibited by : |
|
Answer»
|
|
| 6. |
Tautomerism is/are exhibited by |
|
Answer» `(Me_(3)"CCO")_(3)` CH 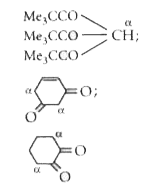
|
|
| 7. |
Tautomer of ethyl methyl ketone is |
|
Answer» but -2- EN -2-ol |
|
| 9. |
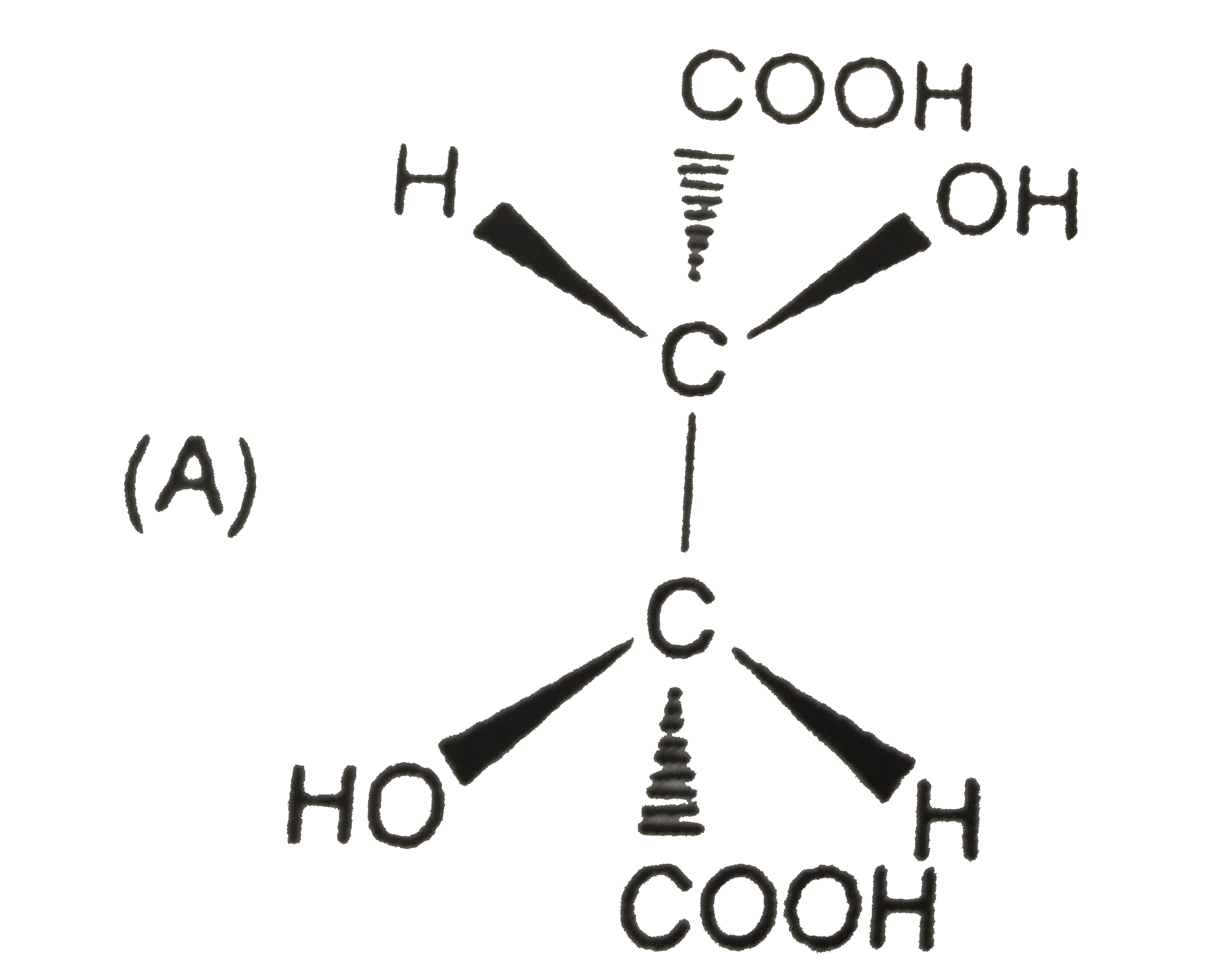
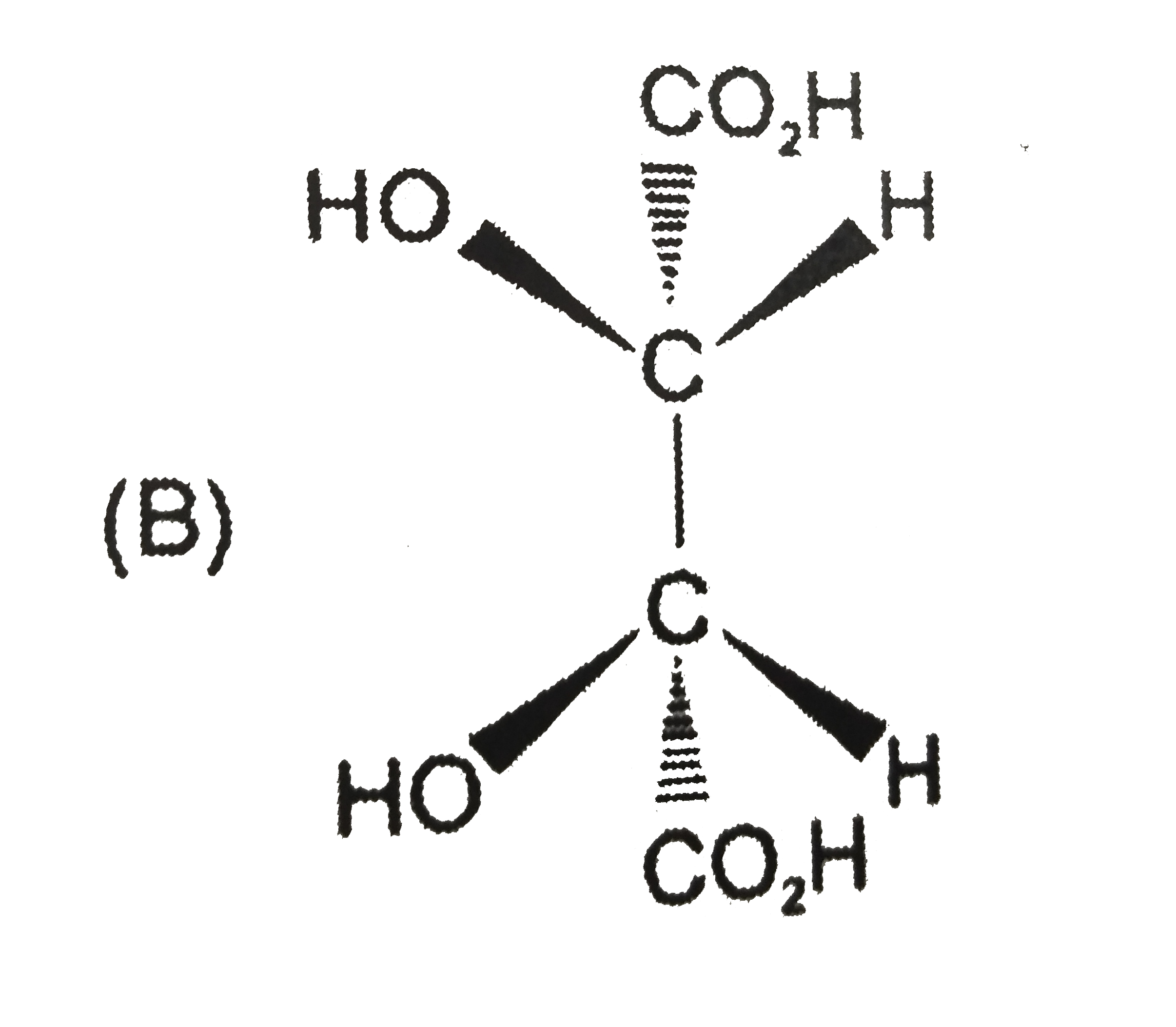
Tartaric acid [HO_(2)CCH(OH)CH(OH)CO_(2)H] was an important compound in history of stereochemistry. Two naturally occuring formsof tartaric acid are optically inactive. One optically inactive form (P) has a melting point of 210-212^(@)C and can be separated into two optically acitve forms, whereas other optically inactive form (Q) cannot be resolved further. A optically inactive form P is : |
|
Answer» OPTICALLY inactive due to INTERNAL compensation. |
|
| 10. |
Tartaric acid is a weak diprotic acid with K_1 = 1 xx 10^(-3)and K_2 = 4.6 xx 10^(-5) .(a) Letting the symbol H_2Arepresent tartaric acid, write the chemical equationsthat represent K_1 and K_2. Write the chemical equation that represents K_1 xx K_2.(b) Qualitatively describe the relative concentrations of H_2A , HA^(-) , A^(2-)and H_3O^+in a solution that is about 0.5 M in tartaric acid.(c) Calculate the pH of a 0.025 M tartaric acid solution and the equilibrium concentration of H_2A(d) What is the A^(2-) concentration? |
Answer» SOLUTION :
|
|
| 11. |
Tartaric acid [HO_(2)CCH(OH)CH(OH)CO_(2)H] was an important compound in history of stereochemistry. Two naturally occuring formsof tartaric acid are optically inactive. One optically inactive form (P) has a melting point of 210-212^(@)C and can be separated into two optically acitve forms, whereas other optically inactive form (Q) cannot be resolved further. Optically inactive form Q is |
|
Answer»
|
|
| 12. |
Tartaric acid is not used in : |
|
Answer» DYEING CLOTHES |
|
| 13. |
Tars'are produced due to oxidationwhile ______ ofaniline |
|
Answer» Bromination |
|
| 14. |
Tarnishing of silver is an example of |
|
Answer» rusting |
|
| 15. |
Tantalum and nobonium have sinmilar ionic radii because |
|
Answer» of diagonal relationship |
|
| 16. |
Tanning of leather is |
|
Answer» colouring of LEATHER by CHEMICALS |
|
| 17. |
Tanning makes leather get hardened - Explain why? |
| Answer» Solution :Skin and hides are protein containing POSITIVELY charged particles which are coagulated by adding TANNIN which is negatively charged to GIVE HARDENED LEATHER for further application. | |
| 18. |
Tamarind contains : |
|
Answer» (+) TARTARIC ACID |
|
| 19. |
Taking two examples of heterogeneously calalytic reaction, explain how a heterogeneous catalyst helps in the reaction. |
|
Answer» Solution :When the reactants and products are in different physical state from the catalyst, it is a case of heterogeneous catalysis. `V_2O_5` and Pt are heterogeneous CATALYSTS in Contact PROCESS and Ostwald Process, respectively. (i) Contact Process : `2SO_2(G) + O_2(g) overset(V_2O_5)iff 2SO_3(g)` (ii) Ostwald.s Process : `4NH_3(g) + 5O_2 (g) underset(1120K) overset(Pt)to 4NO(g) + 6H_2O (g)` Reacting gases get adsorbed on heterogeneous solid catalyst, their concentration on the surface increases and probability of collisions increases which result in increase in number of effective collisions, RESULTING in increase in rate of reaction. Products FORMED get desorbed from the catalyst and more reacting gases get adsorbed and react leading the formation of more products. |
|
| 20. |
Talc contains which of the following |
|
Answer» Zinc, calcium and OXYGEN |
|
| 21. |
Taking N_(2) and O_(2) as main compounts of air (79% N_(2),21%O_(2) by volume ) what is the molecular mass of air? How has it been arrived at ? |
|
Answer» Solution :For a mixture of GASES, the average molecular mass is taken. `"Average molecular mass "=Sigmax_(i)M_(i)=x_(N_(2))M_(N_(2))+x_(O_(2))M_(O_(2))` where `x_(N_(2)) and x_(O_(2))` are mole fractions of `N_(2)` MOLES, therefore, their MOLAR ratio is same as ratio of their volumes. `therefore x_(N_(2))=0.79,x_(O_(2))=0.21.` Also `M_(N_(2))=28u, M_(O_(2))=32U` `therefore"Average molecular mass "=0.79xx28+0.21xx32u=22.12+6.72u=28.84u` |
|
| 22. |
Taking the example of Al_(2)(SO_(4))_(3), derive the relation between molar conductivity and equivalent conductivity. |
|
Answer» SOLUTION :Molar conductivity, `wedge_(m)=(kappaxx1000)/(c_(m)("Molarity"))`, Equivalent conductivity, `wedge_(eq)=(kappaxx1000)/(c_(eq)("Normality"))` Eq. wt. of `Al_(2)(SO_(4))_(3)=(Mol." " wt.)/(6)` (Total +ve VALENCY of AL in `Al_(2)(SO_(4))_(3)=6`) `therefore`Normality`=6xx`Molarity Hence, `(wedge_(m))/(wedge_(eq))=("Normality")/("Molarity")=6` or `wedge_(m)=6wedge_(eq)` Alternatively, `wedge_(eq)=(wedge_(m))/(v_(+)z_(+))=(wedge_(m))/(2xx3)=(wedge_(m))/(6)` |
|
| 23. |
Taking CFSE value of octahedral complex (Delta_(0)) as 10 Dq, the e_(g) orbitals are …… Dq above the bari centre |
|
Answer» |
|
| 24. |
Tailing of mercury can be removed by : |
|
Answer» `H_2O_2` |
|
| 25. |
Taj mahal is thretened by pollution from |
|
Answer» Oxygen |
|
| 26. |
Taj Mahal is threatened due to the effect of |
|
Answer» Chlorine |
|
| 27. |
Taj Mahal is threatened by pollution from :- |
|
Answer» Chlorine |
|
| 28. |
Taj Mahal is threatened by pollution form: |
|
Answer» chrlorine |
|
| 30. |
T_(50) of first - order reaction is 10 min . Starting with 10 mol L^(-1) , rate after 20 min is |
|
Answer» `0.0693` mol `L^(-1) "min"^(-1)` K = 0.693/10 = 0.0693 `"min"^(-1)`. CONCENTRATION at starting = 10 M Concentration after 20 min (two half-lives) = 2.5 M `((dx)/(dt)) = k[A] = 0.0693 xx 2.5` mol `L^(-1) "min"^(-1)`. |
|
| 31. |
t_(1//4) can be taken as the time taken for the concentration of a reactant to drop to 3//4 of its initial value. If the rate constant for a first order reaction is k, the t_(1//4) can be written as |
|
Answer» `0.10//k` |
|
| 32. |
t_(1//4) can be taken as the time taken for the concentration of a reactant of drop to (3)/(4) of its initial value. If the rate constant for a first order reaction is K, then t_(1//14) can be written as |
|
Answer» `0.10//K` `=(2*303xx 0*125)/(K) = (0*29)/(K)` |
|
| 33. |
t_(1//2) of the reaction increases with increases in initial concentration of the reaction means the order of reaction will be ...... |
|
Answer» FIRST ORDER |
|
| 34. |
(t_(1))/(4) can be taken as the time taken for the concentration of a reactant to drop to (3)/(4) of its initial value. If the rate constant for a first order reaction is k, the (t_(1))/(4) can be writen as |
|
Answer» `(075)/(K)` |
|
| 35. |
T_(1//2) " of "C^(14) isotope is 5770 years. Time after which 72% of isotope left is |
|
Answer» 2740 YEARS `:. t = (2.303)/(K)"LOG"(100)/(72) = (2.303 xx 5770)/(0.693) "log" (100)/(72)` `= 19175.05 xx (log 100 - log 72)` `19175.05 xx 0.143 = 2742.03` years |
|
| 36. |
t-Butyl chloride underset("cut")overset(u)toAoverset("n-Pentyl bromide")toB B is |
|
Answer» 2-methyl heptane |
|
| 38. |
(T) impartsorange colour overset(compd (U) + conc. H_2SO_4)to (V) Red gas overset(NaOH + AgNO_3)to (W) Red ppt.overset(NH_3 soln.)to(X) (W) Red ppt. overset(dil. HCl)to(Y) white ppt. (U) undersetDeltaoverset(NaOH)to (Z) gas (gives white fumes with HCl)sublimes on heating . Identify (T) to (Z) . |
|
Answer» `T=KMnO_4 , U=HCl , V=Cl_2 , W=HgI_2 , X =Hg(NH_2)NO_3 , Y=Hg_2Cl_2, Z=N_2` |
|
| 39. |
(T) imparts violet colour in the flame test overset("compound"(U)+conc.H_(2)SO_(4))to(V)_("Red gas")overset(NaOH+AgNO_(3))to(W)_("Red ppt.")overset(NH_(3)" soln")to(X) (W)_("Red ppt.")overset(dil.HCl)to(Y)_("White ppt.") (U) Sublimes on heating underset(Delta)overset(NaOH)to(Z)_(gas) Identify (T) to (Z). |
|
Answer» `T=KMnO_(4),U=HCl,V=Cl_(2),W=HgI_(2),X=Hg(NH_(2))NO_(3),Y=Hg_(2)Cl_(2),Z=N_(2)` |
|
| 40. |
t-butyl alcohol is heated with Al_2O_3 gives |
|
Answer» `CH_3CH=CHCH_3` |
|
| 41. |
(t)-2-Chloro-2-phenylethane in toluene racemises slowly in the presence of small amount of SbCl_(5). due to the formation of |
|
Answer» carbanion `CH_(3) - "CHCl" - C_(6) H_(5) + "SbCl"_(5) rarr [ CH_(3) - overset(+)(C)H - C_(6) H_(5) ] SbCl_(6)^(-)` Since the carbocation is a planar species, therefore, it can be attacked by `SbCl_(6)`either from the top or the bottom face with EQUAL case. As a result, a 50: 50 mixture of two enantiomers of 2-chloro-2-phenylethane are FORMED, i.e., (+)-2-chloro-2- phenylethane undergoes racemization due to the FORMATION of carbocation intermediate. |
|
| 42. |
Systems which "has"//"have" not been correctly characterised is |
|
Answer» cubic, a = b = c, `alpha=beta=gamma= 90^@` |
|
| 43. |
Synthetic tranquillisersare mostly barbituric acid derivatives (e.g. veronal, luminal, seconal, amytal and nembutal). Other tranquillisers which are not barbituric acid derivatives are chlordiazepoxide, equanil meprobamate, valium (diazepam), etc. Opium alkaloids such as morphine and codeine are powerful analgesics (reduce pain). Heroin which is morphine diacetate is a widely used narcotic analgesic. Drugs which are used to cure diseases caused by microbes are called antimicrobials. These may be either sulpha drugs such as sulphapyridine, sulphadiazine, sulphaguanidine or they may be antibiotics. Antibiotics such as erythromycin, tetracycline, chloramphenicol which inhibit or arrest the growth of microbes are called bacteriostatic while others which kill the microbes are called bactericidal antibiotics. Which of the following is/are antibacterials ? |
|
Answer» PENICILLIN |
|
| 44. |
Synthetic tranquillisersare mostly barbituric acid derivatives (e.g. veronal, luminal, seconal, amytal and nembutal). Other tranquillisers which are not barbituric acid derivatives are chlordiazepoxide, equanil meprobamate, valium (diazepam), etc. Opium alkaloids such as morphine and codeine are powerful analgesics (reduce pain). Heroin which is morphine diacetate is a widely used narcotic analgesic. Drugs which are used to cure diseases caused by microbes are called antimicrobials. These may be either sulpha drugs such as sulphapyridine, sulphadiazine, sulphaguanidine or they may be antibiotics. Antibiotics such as erythromycin, tetracycline, chloramphenicol which inhibit or arrest the growth of microbes are called bacteriostatic while others which kill the microbes are called bactericidal antibiotics. Among the following the narcotic analgesic is |
|
Answer» heroin |
|
| 45. |
Synthetic tranquillisersare mostly barbituric acid derivatives (e.g. veronal, luminal, seconal, amytal and nembutal). Other tranquillisers which are not barbituric acid derivatives are chlordiazepoxide, equanil meprobamate, valium (diazepam), etc. Opium alkaloids such as morphine and codeine are powerful analgesics (reduce pain). Heroin which is morphine diacetate is a widely used narcotic analgesic. Drugs which are used to cure diseases caused by microbes are called antimicrobials. These may be either sulpha drugs such as sulphapyridine, sulphadiazine, sulphaguanidine or they may be antibiotics. Antibiotics such as erythromycin, tetracycline, chloramphenicol which inhibit or arrest the growth of microbes are called bacteriostatic while others which kill the microbes are called bactericidal antibiotics. Which of the following tranquillisers is not a derivative of barbituric acid ? |
|
Answer» Veronal |
|
| 46. |
Synthetic tranquillisersare mostly barbituric acid derivatives (e.g. veronal, luminal, seconal, amytal and nembutal). Other tranquillisers which are not barbituric acid derivatives are chlordiazepoxide, equanil meprobamate, valium (diazepam), etc. Opium alkaloids such as morphine and codeine are powerful analgesics (reduce pain). Heroin which is morphine diacetate is a widely used narcotic analgesic. Drugs which are used to cure diseases caused by microbes are called antimicrobials. These may be either sulpha drugs such as sulphapyridine, sulphadiazine, sulphaguanidine or they may be antibiotics. Antibiotics such as erythromycin, tetracycline, chloramphenicol which inhibit or arrest the growth of microbes are called bacteriostatic while others which kill the microbes are called bactericidal antibiotics. The bactericidal and bacteriostatic antibiotics respectively are |
|
Answer» PENICILLIN, ofloxacin |
|
| 47. |
Systamatic name of Pb-CH_(2)-COOH is: |
|
Answer» benzeneceticacid |
|
| 48. |
Synthetic testosterone promotes |
|
Answer» MENSTRUAL CYCLE |
|
| 49. |
Synthetic rubber is a vulcanisable rubber like polymer Write the method of preparation of the above synthetic rubber |
Answer» SOLUTION :
|
|





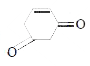





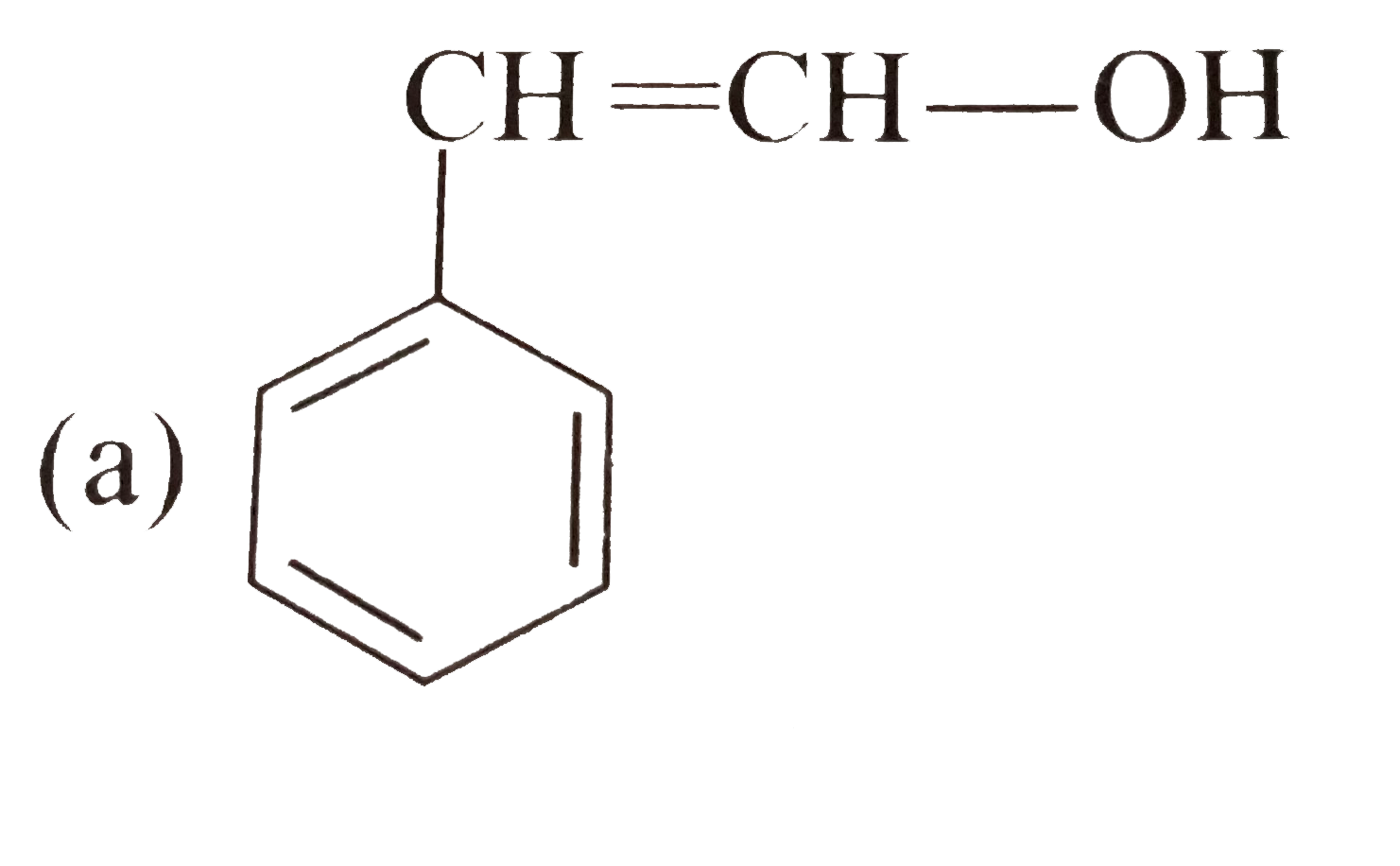
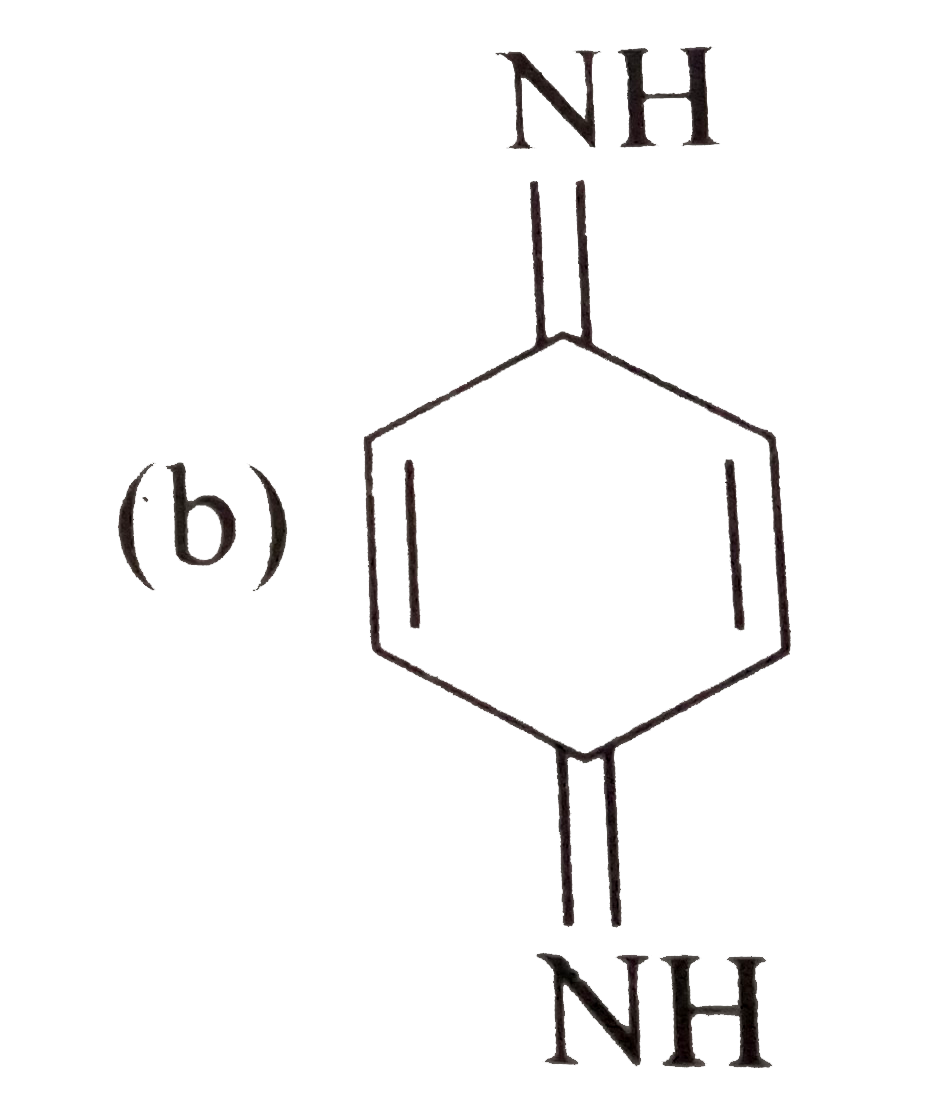
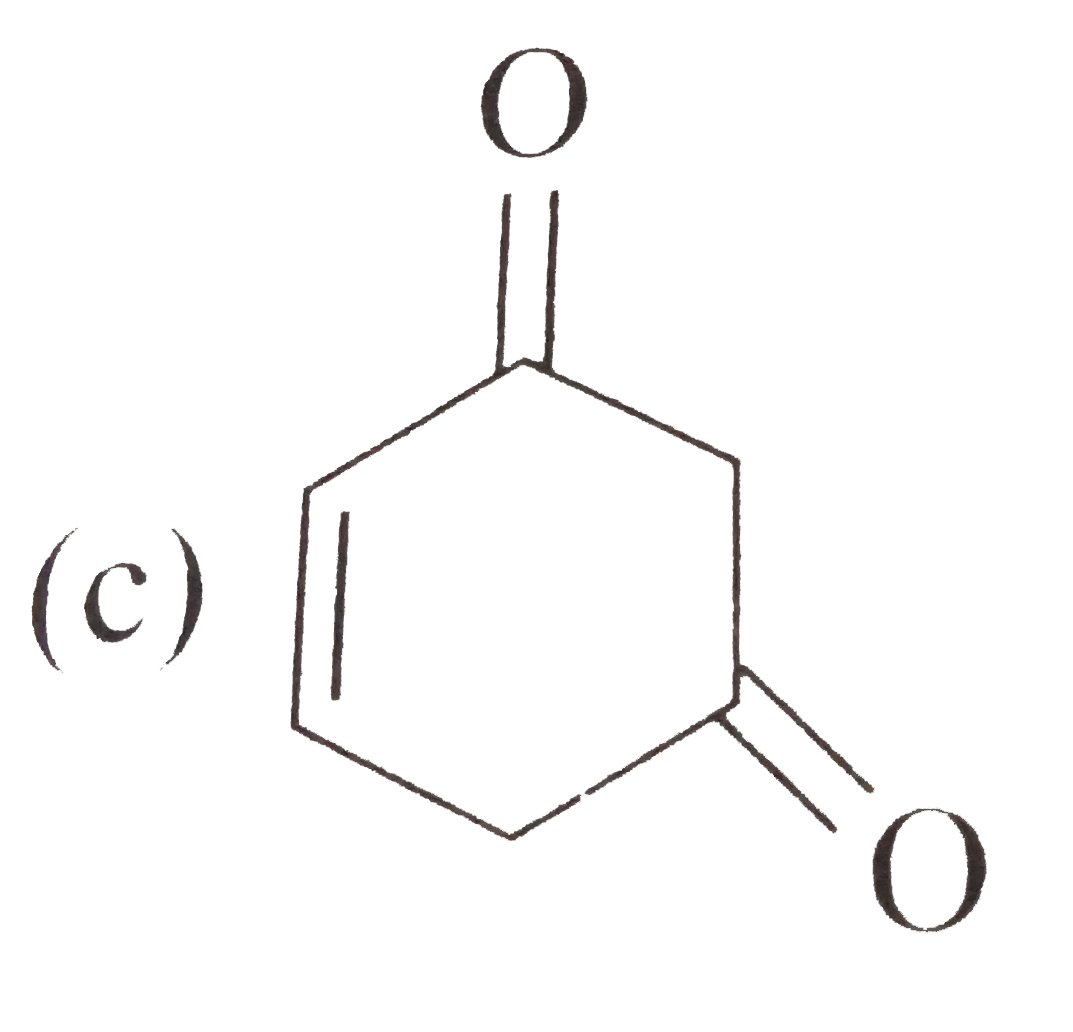
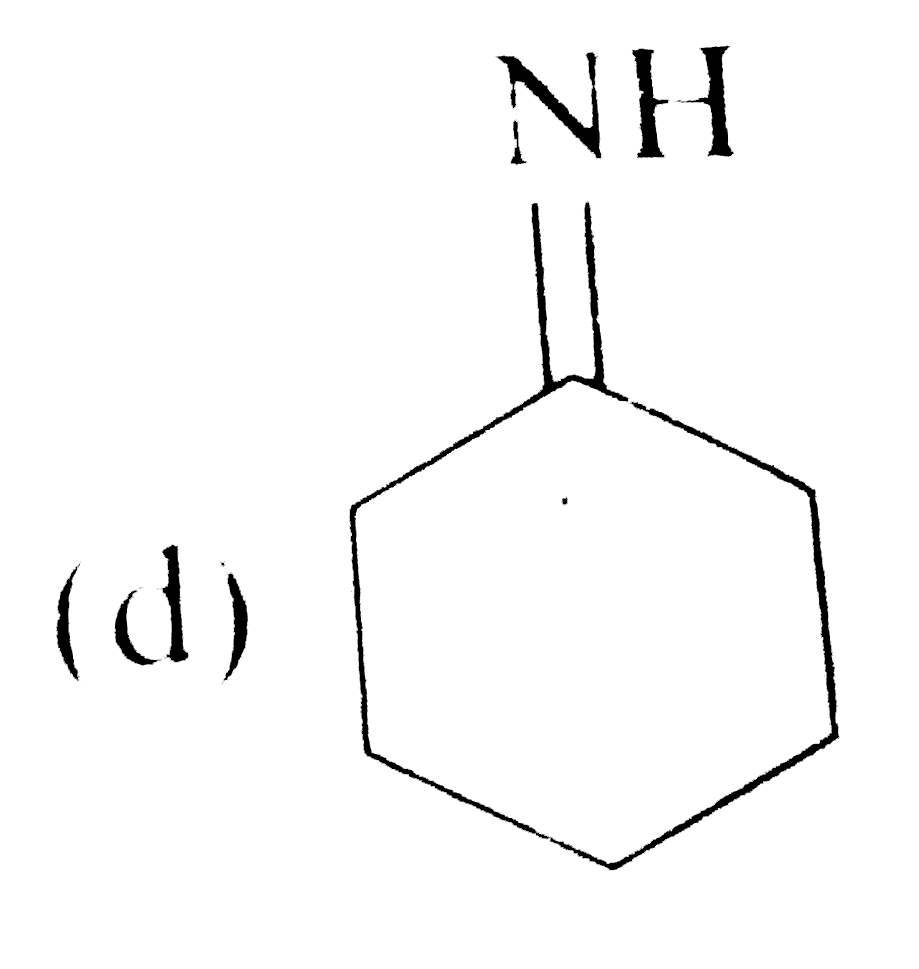



` <br> Availability of acidic `alpha` H - atoms at these position (shown by asterisk marks) enable the compounds A,B and D to exhibit keto - enol tautomerism.)