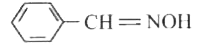Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Tendency for catenation is strongest in: |
|
Answer» C |
|
| 2. |
Ten grams of a gas occupies 2 L at STP. At what temperature will the volume of the same quantity of a gas becomes double at the same pressure ? |
|
Answer» Solution :`( V_(1))/( T_(1)) = (V_(2))/( T_(2))` (Charles. law) `T_(2) = ( 4L xx 273)/( 2L) = 546K` |
|
| 3. |
Ten grams of potassium chloride are dissolved in 10^(3) kg of solution, its strength may be expressed as |
|
Answer» Solution :Ten grams are present in `10^(3)kg=1` MILLION gram `:.` STRENGTH `=10` ppm |
|
| 4. |
Ten grams of a fairly concentrated solution of cupric sulphate is electrolysed using 0.01 faraday of electricity. Calculate (i) the weight of the resulting solution and (ii) the number of equivalent of acid or alkali in the solution. (Cu = 63.5, F = 96500 coulombs) |
|
Answer» Solution :(i) Since mole of electric charge used = 0.01 F `therefore` eq/ of Cu deposited = 0.01 `therefore` WT. of Cu deposited `= 0.01 xx 31.75 = 0.3175g`. `("eq. wt. of Cu " = (63.5)/(2) = 31.75)` Eq. of oxygen discharged = 0.01 `therefore` wt. of oxygen discharged `= 0.01 xx 8 = 0.08g`. `("eq. wt. of Cu " = (16)/(2) = 8)` Total loss in weight due to electrolysis = 0.3175 + 0.08 = 0.3975 g `therefore` the weight of the resulting solution = 10 - 0.3975 = 9.6025 g. (ii) Electrolysis of `CuSO_(4)` solutions follows through `CuSO_(4) = Cu^(2+) + SO_(4)^(2-)` At cathode : `Cu^(2+) + 2E^(-) RARR Cu` At anode : `SO_(4)^(2-) + H_(2)O rarr H_(2)SO_(4) + (1)/(2)O_(2) uarr + 2e^(-)` Thus `H_(2)SO_(4)` is produced during electrolysis. `therefore` eq. of `H_(2)SO_(4)` produced = 0.01 (due to passage of 0.01 faraday) |
|
| 5. |
Temporary hardness of water can be removed by |
|
Answer» ADDITION of potassium permanganate `CA(HCO_(3))_(2) overset("Boil") underset("(insoluble)")(CaCO_(3))+H_(2)O +CO_(2)` |
|
| 6. |
Temporary hardness may be removed from water by adding ? |
|
Answer» `CaCO_(3)` |
|
| 7. |
Temporary hardness in water is due to which one of the following of Calcium and Magnesium ? |
|
Answer» Hydrogencarbonates |
|
| 8. |
Temporary hardness is caused due to the presence of : |
|
Answer» `CaSO_4` |
|
| 9. |
Temporary hardness is caused due to the presence of |
|
Answer» `CaCO_(3)` |
|
| 10. |
Temperature range used for the reduction of FeO by overall effect of the given reactions is related to which points shown in the adjacent graph. (i) C+O_(2)rarrCO_(2) (ii) 2C+O_(2)rarr2CO (iii) 2CO+O_(2)rarr2CO_(2) |
| Answer» Answer :B::D | |
| 11. |
Temperature of stars is several times that of sun . Which of the following cycles are reponsible for the stellar energy of stars ? |
|
Answer» <P>Porton- porton CYCLE |
|
| 12. |
Temperature coefficient of most of the reactions lies between…………………….and…………………….. . |
|
Answer» |
|
| 13. |
Temperature coefficient of a reaction is 2. when temperature is increased from 30^(@)C to 100^(@)C, rate of the reaction increases by |
|
Answer» 128 times i.e., `2^(7)=128` times. |
|
| 14. |
The temperature coefficient of a reaction is 2. When the temperature is increased from 30^(@)C to 90^(@)C, the rate of reaction is increased by |
|
Answer» 60 times |
|
| 15. |
Temperature at the foot of a mountain is 30^(@)C and pressure is 760 mm whereas at the top of the mountain these are 0^(@)C and 710 mm. compare the densities of the air at the foot and at the top of the mountain. |
|
Answer» <P> Solution :`d=(Pm)/(RT) rArr (d_(1))/(d_(2))=(P_(1))/(T_(1))xx(T_(2))/(P_(2))=(760 mm)/(303 K) xx (273 K)/(710 mm) =(0.964)/(1) =0.964` |
|
| 16. |
Temperature and heat are: |
|
Answer» EXTENSIVE PROPERTIES |
|
| 17. |
Tellurium forms oxides of the formula Teo, TeO_(2) and Te_(3) . What is the nature of these oxides? |
|
Answer» SOLUTION :`TEO` (oxidation number of Te is `+2`) is basic. `TeO_(2)` (oxidation number of Te is `+4`) is AMPHOTERIC. `TeO_(2)` (oxidation number of Te is `+6`) is acidic. As the oxidation number of the element forming oxide increases, the acidic nature also increases. |
|
| 18. |
Teflon, styron and neoprene are all |
|
Answer» Copolymers |
|
| 19. |
Teflon, styrene and neoprene are all |
|
Answer» Copolymers |
|
| 20. |
Teflon, polystyrene and neoprene are called |
|
Answer» COPOLYMERS |
|
| 21. |
Teflon , neoprene , Nylon -6 are all |
|
Answer» co-POLYMER |
|
| 22. |
Teflon, Orlon and Neoprene are ……………….. |
|
Answer» HOMOPOLYMER |
|
| 23. |
Teflon is polymer of :- |
|
Answer» Tetrafluoro ETHENE |
|
| 24. |
Teflon is made by polymerisation of: |
|
Answer» Tetrafluoroethene |
|
| 25. |
Teflon is an example of: |
|
Answer» Fibres |
|
| 27. |
Teflon is a polymer of ________. |
|
Answer» ethylene |
|
| 28. |
Teflon is a polymer of the monomer |
|
Answer» Monofluoroethene |
|
| 29. |
Teflon is a type of _____. |
| Answer» SOLUTION :ADDITIONAL POLYMER | |
| 31. |
Teflon coating is done in a number of items of daily use. Name any two such items and for what purpose this coating is done? |
| Answer» SOLUTION :Teflon is an insulating material. Now a days, some PEOPLE go for teflon coating on the body of their car as it covers the painted SURFACE and protects from sratch, RUST etc. teflon coating is also done on the inner side of the non-stick pans as it has great CHEMICAL inertness and thermall stability. | |
| 32. |
Teflon is: |
|
Answer» Fluorocarbon |
|
| 33. |
Teflon is : |
|
Answer» `(-CF_2-CF_2-)_n` |
|
| 34. |
Teflon and neoprene are |
|
Answer» copolymers |
|
| 36. |
TeCl_(4)is expected to be |
|
Answer» TETRAHEDRAL |
|
| 37. |
Tear gas is also known as |
|
Answer» Chloropicrin |
|
| 38. |
Tear gas is |
|
Answer» `COCl_2` |
|
| 39. |
Teacher explained that due to a stoichiometric defect, the density of a crystal changes. What change can we observe? |
| Answer» SOLUTION :DENSITY DECREASE | |
| 40. |
Teacher explained that due to a stoichiometric defect, the density of a crystal changes. Name the defect. |
| Answer» SOLUTION :SCHOTTKY DEFECT | |
| 41. |
Teacher explained crystal defects in class room.What are the different types of crystal defects? |
| Answer» SOLUTION :STOICHIOMETRIC DEFECTS and Non-Stoichiometric defects | |
| 42. |
Te yield of product in the reaction A_(2(g))+B_(2(g))hArr2AB_((g)),DeltaH=+ve |
|
Answer» Unaffected by PRESSURE |
|
| 43. |
Te defect that is more likely to occur in almost all types of ionic crystals is |
|
Answer» Non-stoichiometric defects |
|
| 44. |
The reagents, NH_(4)Cl and aqueous NH_(3) will precipitate |
|
Answer» `Ca^(2+)` |
|
| 45. |
Tb ^(+4) has electronic configuration 4f ^(7) 5d ^(0) 6s ^(0). Because of this it hs strong tendency to |
|
Answer» loose ELECTRONS |
|
| 46. |
Tautomerism will be exhibited by |
|
Answer» `(CH_3)_3CNO` 
|
|
| 47. |
Tautomerism is possible in (1) CH_(3) - CHO""(3) H-CHO (2) CH_(3)-CO-CH_(3)""(4) (CH_(3))_(3)C-CHO |
|
Answer» `1, 2` |
|
| 48. |
Tautomerism is shown by |
|
Answer» `CH_(3)-O-NO` |
|
| 49. |
Tautomerism is not exhibited by |
|
Answer»
|
|
| 50. |
Tautomerism is NOT exhibit by _________. |
| Answer» Answer :A | |