Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Suggest a suitable reagent to prepare secondary alcohol with identical group using Grignard reagent. |
|
Answer» `underset("(Acetaldehyde)")(CH_(3)-overset(O)overset("||")C-H)+underset("(methyl magnesium bromide)")(CH_(3)-Mg-Br)rarrunderset(""CH_(3))underset("|")overset(""OMgBr)overset("|")(CH_(3)-C-H)overset (H^(+)//H_(2)O)rarrunderset("(isopropyl alcohol)")(underset(CH_(3)"")underset("| ")(CH_(3)-CH-OH)+MgBr(OH)` |
|
| 2. |
Suggest a suitable reagent to distinguish acetaldehyde from acetone. |
|
Answer» Solution :Acetaldehyde reduces the Tollens reagent to form silver mirror, while acetone does not give this test. `MeCHO+2[Ag(NH_(3))_(2)]^(o+)+3overset(ɵ)(OH)rarr underset(mirror)underset(Silver)(2Ag) darr+4NH_(3)+2H_(2)O` |
|
| 3. |
Suggest a suitable carbonyl compound for the preparation of pent-2-en-1-ol using LiAlH_4. |
| Answer» SOLUTION :`underset(("2 - pentenal"))(CH_3-CH = CH - CH_2 - CHO) underset(2[H])overset(LiAlH_4)tounderset("pent - 2- 1 - OL")(CH_3-CH = CH - CH_2 - CH_2-OH)` | |
| 4. |
Suggesta structuralformulaof a compoundhavingmolecularC_(6) H_(11)N (A) whichis opticallyactivedissolvesin dil aqueous HCI and releases N_(2) withnitrousacid |
|
Answer»
 hasone asymmetricC - ATOMAND ISA primaryamine |
|
| 5. |
Suggest a scheme to convert CH_(3)CH_(2)OH into an acid containing 1 more carbon atom. |
| Answer» Solution :`underset("ethanol")(CH_(3)CH_(2)OH)overset(SOCl_(2))tounderset("ETHYLCHLORIDE")(CH_(3)CH_(2)Cl)underset(KCN)overset(ALC)tounderset("ethyl CYANIDE")(CH_(3)CH_(2)CN)overset(H_(2)O //H^(+))tounderset("Propionic ACID")(CH_(3)CH_(2)COOH)` | |
| 6. |
Suggest a scheme to convert alcohol into an acid with one more carbon atom. |
| Answer» Solution :`underset(1^(@)" Alcohol")(RCH_(2)OH) underset(-SO_(2),-HCL)overset(SOCl_(2))to RCH_(2)Cl underset(-KCL)overset(Alc." "KCN)to RCH_(2)CN underset("Hydrolysis")overset(H^(+)//H_(2)O)to underset("Carboxylic ACID")(RCH_(2)COOH)` | |
| 7. |
Suggest a reason why HF is a weak acid, wheres binary acids of the all other halogens are strong acids. |
|
Answer» SOLUTION :The hydrogen halides are extremely soluble in water due to the ionisation. `underset("(X = F, Cl, Br or I)")(X+H_(2)Orarr H_(2)O^(+)+X^(-))` Solutions of hydrogen halides are therefore acidic and known as hydrohalic acids. Hydrochloric, hydrobromic and hydroiodic acids are almost completely ionised and are therefore STRONG acids but HF is a weak acid. For HF, `HF+H_(2)OhArr H_(3)O^(+)+F^(-)` `HF+F^(-)rarr HF_(2)^(-)` At high concentration, the equilibrium involves the REMOVAL of fluoride ions is IMPORTANT. Since it affects the dissociation of hydrogen fluoride, therefore it is a weak acid.. |
|
| 8. |
Suggest a reason for the large difference in the boiling points of butanol and butanal, although they have the same solubility in water. |
| Answer» Solution :The b.p. of butanol is higher than that of BUTANAL because butanol has strong intermolecular H-bonding while butanal has weak dipole-dipole ATTRACTION. HOWEVER, both of them FORM H-bonds with water and HENCE are soluble. | |
| 9. |
Suggest a reason for the larger differenece between boiling points of butanol and butanal, although they have almost the same solubility in water. |
|
Answer» Solution :BUTANOL has greater capacity for INTER MOLECULAR hydrogen BONDING than butanal. |
|
| 10. |
Suggest a reason for the large difference in the boiling point of butanol and butanal, although they have the same solubility in water. |
| Answer» Solution :The boiling point of butanol is HIGHER than that of butanal because butanol has strong intermolecular H - bonding while butanal has weak dipole - dipole interactions. HOWEVER both of them will form H - bond with WATER and hence are soluble. | |
| 11. |
Suggest a reagent for conversion of ethanol to ethanoic acid. |
|
Answer» Solution :Any STRONG oxidising agent such as acidified `KMnO_(4)` or `K_(2)Cr_(2)O_(7)`. `underset("Ethanol")(CH_(3)CH_(2)OH) underset ("or "K_(2)Cr_(2)O_(7)//H_(2)SO_(4))overset(KMnO_(4)//H_(2)SO_(4))to underset("ETHANOIC acid")(CH_(3)COOH)` |
|
| 12. |
Suggest a reagent for convertion of ethanol to ethanal. |
|
Answer» Solution :Pyridinium chlorochromate in `CH_(2)Cl_(2)` `underset("ETHANOL")(CH_(3)CH_(2)OH) underset(CH_(3)Cl_(2))overset(C_(5)H_(5)NH^(+)CrO_(3)CL^(-))to underset("Ethanal")(CH_(3)CHO)` |
|
| 13. |
Suggest a quantitative method for estimation of the gas which protects us from UV rays of the sun. |
|
Answer» Solution :The GAS which protects us from UV rays of the sun is ozone. When ozone reacts with EXCESS of POTASSIUM iodide solution BUFFERED with a borate buffer (pH 9.2), iodine is LIBERATED which can be titrated against a standard solution of sodium thiosulphate. `O_3 + 2I^(-) + H_2O to O_2 + I_2 + 2OH^(-)` This is how ozone is quantitatively estimated. |
|
| 14. |
Suggest a possible reason for the following observations : (i) In the solid state, PCl_(5) behave as an ionic species. (ii) H_(2)S is more acidic than water. (iii) Fluorine forms the largest number of interhalogen compounds amongst the halogens. |
|
Answer» Solution :(i) In solid state `PCl_(5)` BEHAVES as an ionic species : Because of the formation of `(PCl_(4))^(+) and (PCl_(6))^(-)` in which cation is `(PCl_(4))^(+)` is tetrahedral and anion `(PCl_(6))^(-)` octahedral. (ii) `H_(2)S` is more acidic than water : Because `H_(2)S` has lower bond dissociation energy than `H_(2)O` bond. (iii) Largest no. of interhalogen compounds are formed by fluorine due to its smaller size and higher electronegativity. State decreases down the group due to increase in size and metallic character. Bi (last member of the group) hardly forms any compound of `-3` oxidation state. The stability of `+5` oxidation state decreases down the group, whereas that of `+3` oxidation state increases down the group (due to inert PAIR effect). N reacts with oxygen showing `+1, +2, +3,+4 and +5` oxidations states. P shows `+1 and +4` oxidation states in some oxo - acids. (iii) Atomic size :Covalent and ionic radii (in a particular state) increase down the group. From N to P, covalent radius increase considerable. From As to Bi, there is only a small increase in covalent radius. The REASON is the presence of completely filled d - and /- or /f orbitals in heavier members. (iv) Ionization enthalphy : Ionization enthalpy decreases down the group due to gradual increase in atomic size. I.E., of group - 15 elements is greater than that of group - 14 elements and group - 16 elements in thecorresponding periods. |
|
| 15. |
Suggest a reagent for conversion of ethanol to ethanal. |
| Answer» SOLUTION :`to` PYRIDINIUM CHLOROCHROMATE. | |
| 16. |
Suggest a possible mechanism for the formation of R-Li from R-X by equation only. |
Answer» SOLUTION :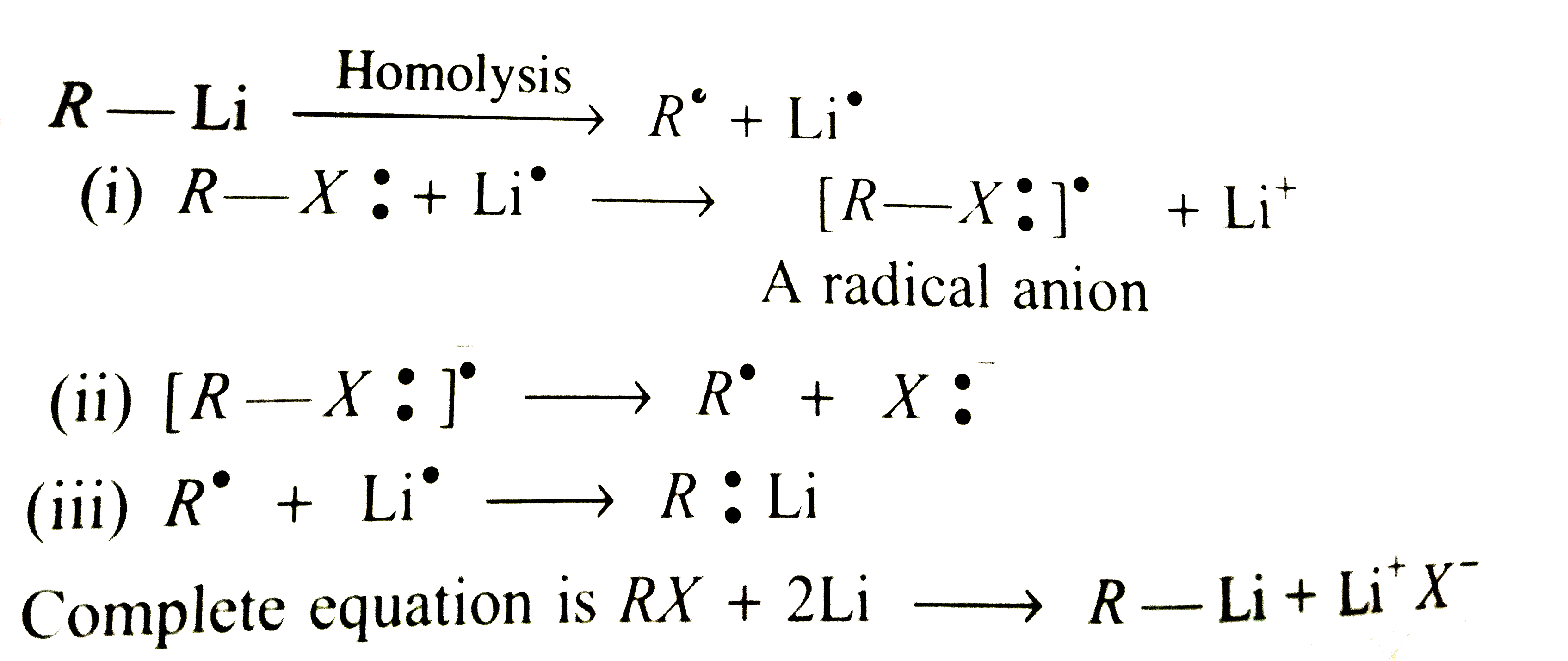
|
|
| 17. |
Suggest a method of preparation of benzaldehyde from toluene. |
|
Answer» |
|
| 18. |
Suggest a method for the conversion of CH_(3)-CH_(2)-NH_(3) into ethyl alcohol. |
|
Answer» SOLUTION :Primary alcohol is prepared from a primary amine using NITROUS ACID. `CH_3-CH_2NH_2overset(HNO_2)(rarr)CH_3-CH_2OH+N_2+H_2O`. |
|
| 19. |
Suggest a mechanism for the following reaction: |
Answer» SOLUTION :The protonated aldehyde is an ELECTROPHILE and the doubleis a nucleophile. The reaction between the two closes the ring, resulting CARBONIUM ion produced GIVES a diol on HYDRATION. 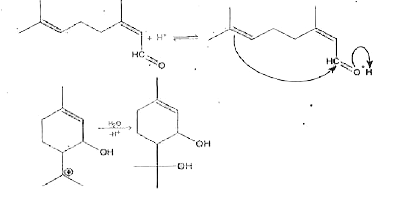
|
|
| 20. |
Suggest a list of metals that are extracted electrolytically. |
|
Answer» SOLUTION :* Al, Na, Mg * Metals that are on the top of the REACTIVITY series such as SODIUM, potassium, calcium, lithium, MAGNESIUM, ALUMINIUM are extracted electrolytically. * Na and Mg metal can be electrolysed by molten chloride. * Aluminium can be obtained by electrolysis of aluminium oxide in presence of cryolite. |
|
| 21. |
Suggesta conditionunders whichmagnesiumcouldreduce alumina. |
|
Answer» Solution :The tworedox REACTIONSARE : ` ""(4)/(3) Al+O_ 2to(2)/(3)Al_2 O _ 3 , Delta _f G ^(@)""_("("Al, Al_2O_3")") ""` ... (i) ` "" 2Mg +O _ 2to2Mg O,Delta _ f G^(@) ""_("("Mg, MgO ")")"" `...(ii) THEMG, MgOand Al, `Al_2O_ 3`curveintersectatpointA (1623K) where`Delta _ rG^(@) = 0 `. The ` Delta _fG^(@) `valueof` Al_2 O _ 3 `, attemperatures below1623 K , is LESS- vethan thatof MgO. Therefore, whenEq.(i) issubtrated fromEq. (ii),` Delta _r G^(@) ` of thetwocombinedredox reactions, i.e.,Eq. (iii) is -ve. ` 2 Mg +(2)/(3) Al_2O _ 3 to2 MgO+(4)/(3)Al , Delta _ rG^(@) = - ve"" `... (iii) Therefore, below 1623 K, Mg can reduce` Al_2O_ 3`to`Al`. However,above1623 K, the` Delta _f G^(@) `value for` Al_ 2O_3`is more-vethanthatofMgO . Therefore,when Eq. (ii)is subtractedfrom Eq.(i),` Delta _r G^(@) `of thetwocombinedredoxreactions , i.e.,Eq. (iv)is-ve. ` (4)/(3) Al +2 MgOto(2)/(3) Al_2O _ 3+2Mg , Delta_ rG^(@) =- ve"" `... (iv) Therefore,above 1623 K, AlcanreduceMgOtoMg. |
|
| 22. |
Suger is soluble in water due to |
|
Answer» HIGH solvation energy |
|
| 23. |
Suggest a condition under which magnesium could reduce alumina. The two equations are : (a) 4/3Al + O_2 to 2/3 Al_2O_3 (b) 2Mg + O_2 to 2MgO |
|
Answer» SOLUTION :At the point of intersection of the `Al_2O_3` and MGO curves (marked "A" in ELLINGHAM diagram), the `A,G^(Theta)`becomes ZERO for the reaction: `2/3Al_2O_3 + 2Mg to 2MgO + 4/3 Al` - Below that point magnesium can REDUCE ALUMINA. |
|
| 24. |
Suggest a combination of organic halide and cuprate regent appropriate for the prepaneof each of the followingcompounds : (a) 2- methybutane (B)i- Butylcyclohexene |
Answer» SOLUTION :`CH_(3)I+LiCu[CH_(2)CH(CH_(3))_(2)]_(2)to UNDERSET("2-Methylbultane ")(CH_(3)CH_(2)CH(CH_(3))_(2))` 
|
|
| 25. |
Sugars have the suffix |
|
Answer» ol |
|
| 26. |
Sugars that yield two to ten monosaccharide molecules on hydrolysis is |
|
Answer»
|
|
| 27. |
Sugars are charactrised by the preparation of osazone derivatives. Which sugar have identical osazones. |
|
Answer» GLUCOSE and lactose |
|
| 28. |
Sugars are |
|
Answer» OPTICALLY active POLYHYDROXY aldehydes |
|
| 29. |
Describe the preparation of ether by williamson synthesis. |
|
Answer» Glucose and LACTOSE |
|
| 30. |
Sugar which will not reduce Fehling's solution is |
|
Answer» maltose |
|
| 31. |
Sugar on fermentation with which enzyme convert into Glucose and Fructose ? |
|
Answer» INVERTASE |
|
| 32. |
Sugar present in fruits is |
|
Answer» glucose |
|
| 33. |
Sugar is the main source of energy as it produces energy on metabolic decomposition. But these days low calorie drinks are more popular, why? |
| Answer» Solution :Natural sweeteners, e.g, sucrose add to CALORIE intake. Due to occurrence of more causes of diabetes and obesity among YOUNG and old PEOPLE, these DAYS people usually TAKE lowcalorie intake. | |
| 34. |
Sugar is the main source of energy as it produces energy on metabolic decomposition. But these days low calorie drinks are more popular, why ? |
| Answer» Solution :Due to OCCURRENCE of more cases of diabetes and obesity among young and old people, these DAYS people usually take low calorie drinks: These CONTAIN artificial sweetening agents which are often many hundreds times SWEETER than sugar but do not metabolize and hence do not produce any energy | |
| 35. |
Sugar is ……………………. |
|
Answer» MONO SACCHARIDE |
|
| 36. |
Sugar is/ are |
|
Answer» SWEET |
|
| 37. |
Sugar differingin configurationat an asymmetriccentre is knowas ……… . |
| Answer» Answer :A | |
| 38. |
Sufficient NaCN was added to 0.015 M AgNO_3to give a solution that was initially0.1 M in CN^-. What is the concentration of Ag^+ in this solution after Ag(CN)_2^-,forms? The formation constant K_ffor the complex ion Ag(CN)_2^-is 5.6 xx 10^18.Ag^(+) + 2CN^(-)iff Ag(CN)_2 |
| Answer» SOLUTION :`5.5 XX 10^(-19) M` | |
| 40. |
Sucross solution which is 40% by mass is heated till it becomes 50% by mass. Water lost form 100 g of the solution is |
|
Answer» 10 g To get `50%` solution, 100 g of solution WOULD CONTAIN 50 g of sucrose or 40 g of sucrose will be present in solution `=(100)/(50)xx40=80g`. Thus, water now present `=80-40g=4g.` Hence, water lost `=60-40g=20g`. |
|
| 41. |
Sucroseunderset ( H^(-)) overset (H_2O) to Y and X. The total no of optical isomers possible for Y and X are |
| Answer» Answer :D | |
| 42. |
Sucrose on treatment with conc. HCl produces |
|
Answer» glucose |
|
| 43. |
Sucrose reacts with acetic anhydride to form |
|
Answer» PENTAACETATE |
|
| 44. |
Sucrose on hydrolysis produces equimolar mixture of |
|
Answer» D(+)-GLUCOSE and D(+)-fructose |
|
| 45. |
Sucrose on hydrolysis gives |
|
Answer» glucose and MALTOSE `underset"Sucrose"(C_12H_22O_11)+H_2O overset"Invertase "to underset"Glucose"(C_6H_12O_6) + underset"Fructose"(C_6H_12O_6)` |
|
| 46. |
Sucrose molecule is formed by monosaccharide of |
|
Answer» `ALPHA`-D-glucofuranose and `BETA`-D-fructopyranose |
|
| 47. |
Sucrose molecule is made up of |
|
Answer» a gluco pyranose and a fructo pyranose |
|
| 48. |
Sucrosemolecule is made up of ……… . |
|
Answer» a gluco pyranoseand fructo PYRANOSE |
|
| 49. |
Sucrose molecule contains |
|
Answer» a GLUCOPYRANOSE and a fructopyranose UNITS |
|



