Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Sulpha drugs work like antibiotics but they are not. Is this a valid statement and why ? |
| Answer» Solution : This is a VALID STATEMENT since sulpha drugs LIKE antibiotics kill microorganisms. However, sulpha drugs are purely synthetic but antibiotics may be EITHER wholly or partially OBTAINED from microorganisms. For example, penicillin is wholly obtained from microorganisms while amoxycillin and ampicillin are semi-synthetic. | |
| 2. |
Sulphaguadine is used for |
|
Answer» Dysentery |
|
| 3. |
Sulpha drugs work like antibiotics but they are not antibiotics. Is this a valid statement and why? |
| Answer» Solution : This is a VALID statement since sulpha drugs like ANTIBIOTICS kill microorganisms. However, sulpha drugs are purely synthetic but antibiotics may be EITHER wholly or partially obtained from microorganisms. For example, PENICILLIN is wholly obtained from microorganisms while amoxycillin and ampicillin are semi-synthetic. | |
| 4. |
Sullphurdioxide can be used for …………….and…………….in agriculture. |
| Answer» SOLUTION :DISINFECTING CROPS and PLANTS | |
| 5. |
Sulpha drugs are used for : |
|
Answer» Precipitating BACTERIA |
|
| 6. |
Sulpha drugs are derivatives of |
|
Answer» <P>BENZENE sulphonic acid |
|
| 7. |
Sulfuric acid having concentration greater than 98.5% H_2SO_4cannot be obtained by distillation of an aqueous solution of H_2SO_4. Why? |
| Answer» SOLUTION :Sulfuric acid having 98.5% `H_2SO_4` FORMS a constant boiling mixture having B.P. of 611 K. Hence, dilute sulfuric acid cannot be CONCENTRATED beyond 98.5% by distillation. | |
| 8. |
Sulfur disappears when boiled with sodium sulfite. Why? |
|
Answer» Solution :On boiling SULFUR with sodium sulfite, sulfur disappears DUE to the FORMATON of sodium thiosulfate. `S + Na_2SO_3 OVERSET("Boil") RARR underset("Sodium thiosulfate")(Na_2S_2O_3)` |
|
| 9. |
Which form of sulphur shows paramagnetic behaviour ? |
|
Answer» Solution :On boiling sulfur with SODIUM SULFITE, sulfur DISAPPEARS due to the formaton of sodium thiosulfate. `S + Na_2SO_3 OVERSET("Boil") rarr underset("Sodium thiosulfate")(Na_2S_2O_3)` |
|
| 10. |
Sulphur sol contains |
|
Answer» DISCRETE S- atoms |
|
| 11. |
Suitable reagents A and B for the following reactions respectively are |
|
Answer» BR and `Br_(2)` 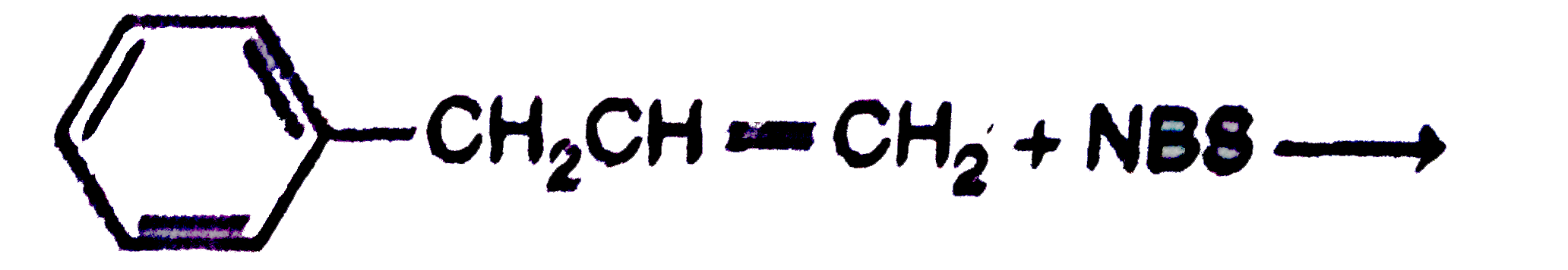
|
|
| 12. |
Suitable reaction to prepare chlorbenzene is |
|
Answer» SANDMEYER's REACTION |
|
| 13. |
Suggest two materials other than hydrogen that can be used as fuels in fuel cells. |
| Answer» SOLUTION :METHANE and METHANOL. | |
| 14. |
Suggest two material other than hydrogen that can be used as fuels in fuels cells. |
| Answer» SOLUTION :METHANE, METHANOL | |
| 15. |
Suggest the structure of nylon 2-nylon 6. |
|
Answer» Solution :Nylon 2-nylon 6 is a BIODEGRADABLE polymer.It is an alternating copolymer obtained by the condensation of glycine and AMINO caproic acid. It is CHEMICALLY a polyamide. Structure of nylon 2- nylon 6 is given as : `[--underset(H)underset(|)N-CH_(2)-underset(O)underset(||)C-underset(H)underset(|)N-(CH_(2))_(5)-underset(O)underset(||)--]_(n)` |
|
| 16. |
Suggest the reagent for conversion of ethanol to ethanoic acid. |
|
Answer» Solution :`to` Acidified `KMnO_(4)` or acidified `K_(2)Cr_(2)O_(7)` oxidizes the ethanol to ethanoic ACID. `CH_(3)CH_(2)OH overset([O])to CH_(3)CHO overset([O])to CH_(3)COOH` |
|
| 17. |
Suggest the most important type of intermolecular interaction in the following pairs : (iv) methanol and acetone |
| Answer» Solution :Both are POLAR MOLECULES. Hence, intermolecular INTERACTIONS in them will be dipole-dipole interactions. | |
| 18. |
Suggest the most important type of intermolecular interaction in the following pairs : (v) Acetonitrile (CH_(3)CN) and acetone (C_(3)H_(6)O). |
| Answer» SOLUTION :Both are POLAR MOLECULES. Hence, intermolecular INTERACTIONS in them will be dipole-dipole interactions. | |
| 19. |
Suggest the most important type of intermolecular interaction in the following pairs : (iii) NaClO_(4) and water |
| Answer» Solution :`NaClO_(4)` gives `NA^(+) and ClO_(4)^(-)` IONS in the solution while water is a polar MOLECULE. Hence, intermolecular interactions in them will be ion-dipole interactions. | |
| 20. |
Suggest the most important type of intermolecular interaction in the following pairs : (i) n-hexane and n-octane |
| Answer» SOLUTION :Both are non-polar. Hence, INTERMOLECULAR interactions in them will be LONDON dispersion FORCES | |
| 21. |
Suggest the most important type of intermolecular interaction in the following pairs : (ii) I_(2) and "CCl"_(4) |
| Answer» Solution :Both are non-polar. Hence, INTERMOLECULAR INTERACTIONS in them will be London DISPERSION FORCES | |
| 22. |
Suggest the most important type of intermolecular attractive interaction in the following pairs :(i) n - hexane and n - octane(ii) I_(2) and C Cl_(4)(iii) NaClO_(4)and water(iv) Methanol and acetone(v) Acetonitrile (CH_(3)CN)and acetone (C_(3)H_(6)O). |
|
Answer» Solution :(i) Van der wall FORCES of attraction as both are non - polar. (ii) Van der Wall forces of attraction as they are non - polar. (iii)ION - diople interaction as `NaClO_(4)` is IONIC in nature. (iv) Dipole - dipole interaction as they are polar. (v) Dipole - dipole interaction as they are polar. |
|
| 23. |
Suggest the most important type of intermolecular attractive interaction in the following pairs : (i) n-hexane and n-octane(ii) I_2 and "CCl"_4 (iii) NaClO_4 and water (iv) methanol and acetone (v) acetonitrile (CH_3CN) and acetone (C_3H_6O). |
|
Answer» SOLUTION :(i) Both are non-POLAR. Hence, intermolecular interactions will be London DISPERSION forces. (ii) Same as (i). (iii) `NaClO_4`gives `NA^+` and `ClO_4^-`ions in the solution and water is a polar molecule. Hence,intermolecular interactions will be ion-dipole interactions. IV) Both are polar molecules. Hence, intermolecular interactions in them will be dipole-dipole interactions. (v) Same as (iv). |
|
| 24. |
Suggest the most important type of intermolecular attractive interaction in the following pairs : (i) n-hexane and n-octane (ii) I_2 and C Cl_4 (iii) NaC1O_4 and water (iv) methanol and acetone (v) acetonitrile (CH_3 CN) and acetone (C_3H_6O). |
| Answer» Solution :(i) van der Waal.s INTERACTIONS, (II) van der Waal.s interactions (III) ion-dipole (iv) HYDROGEN bonding (v) dipoledipole interactions. | |
| 25. |
Suggest the most important type of intermolecular attractive interaction in NaClO_(4) and water |
| Answer» SOLUTION :Ion-dipole INTERACTION | |
| 26. |
Suggest the most important type of intermolecular attractive interaction in n - hexane and n-octane |
| Answer» SOLUTION :VANDER WAALS INTERACTION | |
| 27. |
Suggest the most important type of intermolecular attractive interaction in I_(2) and C Cl_(4) |
| Answer» SOLUTION :VANDER WAALS INTERACTION | |
| 28. |
Suggest reagents for the transformations CH_(3)Ito CH_(3)OH |
| Answer» SOLUTION :`CH_(3)I OVERSET(NaOH)to CH_(3)OH` | |
| 29. |
Suggest reagents for the transformations |
Answer» SOLUTION :
|
|
| 30. |
Suggest reagents for the transformations |
Answer» SOLUTION :
|
|
| 31. |
Suggest reagents for the transformations |
Answer» SOLUTION :
|
|
| 32. |
Suggest onemethodfor separationof nickelfromcobalt ? |
|
Answer» Solution :CO ispassedover aheatedmixture ofNiand Co at330- 350KwhenNiformsvolatile` Ni(CO)_ 4`butCodoes not. The volatile` Ni(CO ) _4`isseparatedandthenheatedat450- 470 KtogetpureNi. `underset ("IMPURE nickel")(Ni (s) ) +4CO(g)overset ( 330 - 350 K ) tounderset ("Nickeltetracarbonyl") (Ni(CO)_4(g)) overset (450- 470 K ) tounderset ("Purenickel") (Ni(s))+ 4CO(g) ` |
|
| 33. |
Suggest one method for carrying out anti-Markovnikov's addition of water indirectly to propylene. Give one example. |
|
Answer» Solution :Hydroboration-oxidation gives ALCOHOLS which correspond to anti-Markovnikov's addition of `H_(2)O` to alkenes. `UNDERSET("propene")(6CH_(3)CH=CH_(2))underset(("Hydroboration"))overset(B_(2)H_(6)//THF)to underset("Tri-n-propylborane")(2(CH_(3)CH_(2)CH_(2))_(3)B) underset(("Oxidation"))overset(H_(2)O_(2)-NAOH)to underset("Propan-1-ol")(6CH_(3)CH_(2)CH_(2)OH)` |
|
| 34. |
Suggest four ways in which the concentration of hydrazine, N_(2)H_(4), could be increased in an equilibrium described by the equation. N_(2)(g)+2H_(2)(g)hArrN_(2)H_()(g)DeltaH=95k J |
| Answer» Solution :ADD `N_(2)`, add `H_(2)`, increase the PRESSURE, HEAT the reaction | |
| 35. |
Suggest epxplanation for the following The pK_(a1) for all of the dicarboxylic acids in Table 17.2 is smaller tyhan the pK_(a) for a monocarboxylic acid with the same number of carbon atoms. |
| Answer» Solution :The CARBOXYL gropu is electron- withdwaring, THUS, in a dicarboxylic ACID such as those in Table 17.2 onecarboxylic acid group INCREASES the acidtiy of the other. | |
| 36. |
Suggest epxplanation for the following The difference between pK_(a1) and pK_(a2) for dicarboxylic acid of the type HO_(2)C(CH_(2))_(n)CO_(2)H decreases as n increases. |
| Answer» SOLUTION :As the distance between the CARBOXYL gropus INCREASES, the acid-strengthening, inductive effect decreases. | |
| 37. |
Suggest appropriate reagents for following conversion. |
Answer» SOLUTION : . .
|
|
| 38. |
Suggest appropriate structures for the missing compounds. (The number of carbon atoms remains the same throughout the reactions). underset(Delta)overset( dil KMnO_(4))rarr(A)overset(HIO_(4))rarr(B)overset(OH^(-))rarr(C) |
|
Answer» |
|
| 39. |
Suggest chemical reactions for the following conversions: (i) Cyclohexanol to Cyclohexylamine, (ii) 1-Hexanenitrile to 1-Aminopentane |
Answer» SOLUTION :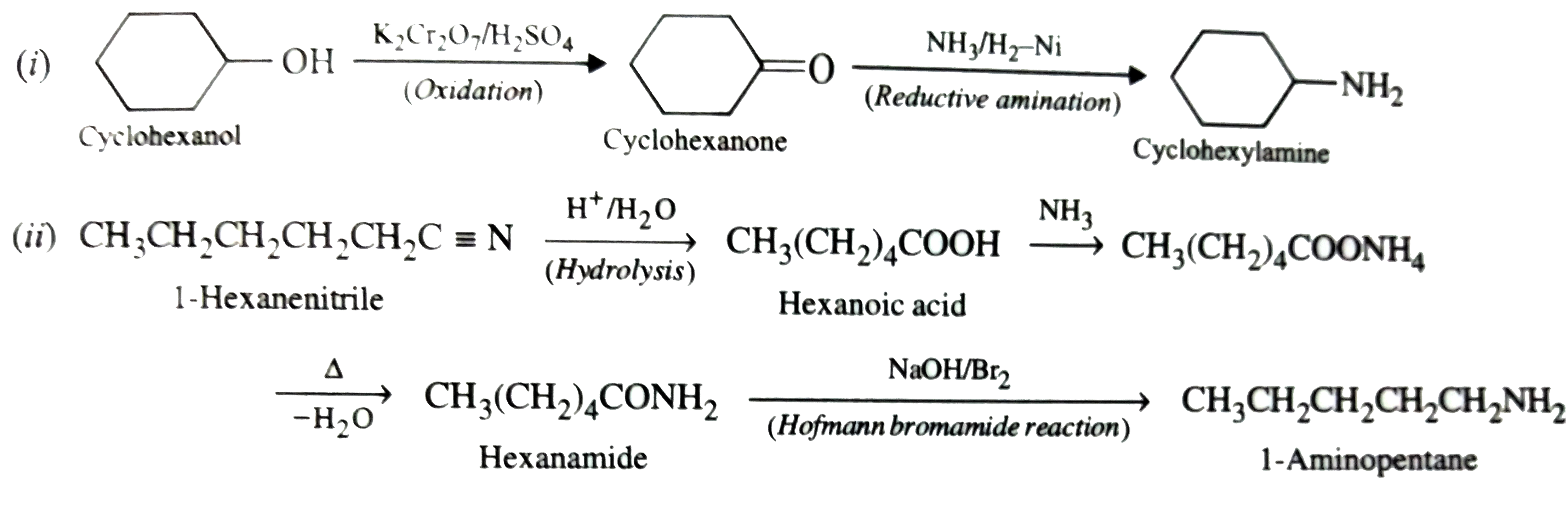 (ii) `CH_(3)CH_(2)CH_(2)CH_(2)CH_(2)C-=N underset(("HYDROLYSIS"))OVERSET(H^(+)//H_(2)O)to underset("Hexanoic ACID")(CH_(3)(CH_(2))_(4)COOH overset(NH_(3))to CH_(3)(CH_(2))_(4)COONH_(4) underset(-H_(2)O)overset(Delta)to underset("Hexanamide")(CH_(3)(CH_(2))_(4)CONH_(2)) underset(("Hofmann bromamide reaction"))overset(NaOH//Br_(2))to underset("1-Aminopentane")(CH_(3)CH_(2)CH_(2)CH_(2)CH_(2)NH_(2)` |
|
| 40. |
Suggest any two uses of carbohydrates. |
|
Answer» SOLUTION :Carbohydrates acts as biofuels to provide ENERGY for the functioning of living systems. They act as CONSTITUENTS of cell membrane. |
|
| 41. |
Suggest any two fluorides ofXenon |
| Answer» SOLUTION :XENON DIFLUORIDE `(XeF_2)`Xenon TETRAFLUORIDE `(XeF_4)` . | |
| 42. |
Suggest any two methods to prevent corrosion of iron. |
|
Answer» SOLUTION :BARRIER PROTECTION. Sacrifical protection ALLOYING CATHODE protection |
|
| 43. |
Suggest a simple method to remove peroxides from ethers. |
| Answer» Solution :By shaking the ETHER with an AQUEOUS solution of KI or a FERROUS SALT. | |
| 44. |
Suggest a way to determine the wedge_(m)^(@) value of water. |
|
Answer» Solution :`wedge_(m)^(@)(H_(2)O)=lamda_(H^(+))^(@)+lamda_(OH^(-))^(@)` we find out `wedge_(m)^(@)(HCl),wedge_(m)^(@)(NaOH) and wedge_(m)^(@)(NaCl)`. Then `wedge_(m)^(@)(H_(2)O)=wedge_(m)^(@)(HCl)+wedge_(m)^(@)(NaOH)-wedge_(m)^(@)(NaCl)` |
|
| 45. |
Suggest a way to determine wedge_(m^(@)) value of water. |
|
Answer» SOLUTION :`Lambda_(m)^(@)(H_(2)O)=ambda_(mH^(+))^(@)+Lambda_(mOH^(-))^(@)` It can be determine from the value of `Lambda_(m)^(@)(HCL), Lambda_(m)^(@)(NAOH) and Lambda_(m)^(@)(NACL)` Then, `Lambda_(m)^(@)(H_(2)O)=Lambda_(m)^(@)(HCl)+Lambda_(m)^(@)(NaOH)-Lambda_(m)^(@)(NaCl)` |
|
| 46. |
Suggest a way to determine the Lambda_(m)^(0)value of water. |
|
Answer» Solution :`Lambda_(m)^(@)(H_(2)O) = Lambda_(H+)^(@) + Lambda_(OH^(-))^(@)` We find out `Lambda_(m)^(@) (HCL), Lambda_(m)^(@)(NaOH)` and `Lambda_(m)^(@) (NaCl)`. Then, APPLYING Kohlaursch LAW, we can calculate `Delta_(m)^(@)` `Lambda_(m)^(@)(H_(2)O) =Lambda_(m)^(@)(HCl) + Lambda_(m)^(@) (NaOH) - Lambda_(m)^(@)(NaCl)`. |
|
| 47. |
Suggest a way to determine the A_m^0 value of water. |
|
Answer» Solution :We know that water is a weak electrolyte. Its molar conductivity at infinite dilution i.e., `A_m^0` can be DETERMINED in terms of `A_m^0` for strong electrolytes. This is in accordance with Kohlrausch.s law. `A_m^0H_2O=A_m^0(HCl)+A_m^0(NAOH)-A_m^0(NACL)` |
|
| 48. |
Suggest a way to determine lambda_m^@ value of water. |
|
Answer» Solution :`lambda_m^@ (H_2O) = lambda_H^@ + lambda_(OH^-)^@` We find out `lambda_m^@ (HCL) , lambda_m^@(NAOH) and lambda_m^@ (NACL)` Then `lambda_m^@(H_2O) = lambda_m^@ (HCl) + lambda_m^@ (NaOH) - lambda_m^@ (NaCl)`. |
|
| 49. |
Suggest a way to determine at Lamda_(m)^(@) value of water. |
|
Answer» Solution :* There is less IONIZATION of pure water. `H_(2)O_((l)) hArr H_((aq))^(+)+OH_((aq))^(-)` * Applying Kohlrausch.s law of INDEPENDENT migration of ions, the `Lamda_(m)^(@)` value of water can be DETERMINED as follows: `Lamda_(m)^(@)(H_(2)O)=lamda_(m)^(@)(H^(+))+lamda_(m)^(@)(OH^(-))` Now, `HCl +NAOH to NaCl+H_(2)O`, so, `Lamda_(m)^(@)(HCl)+Lamda_(m)^(@)(NaOH)-Lamda_(NaCl)^(@)=Lamda_(m)^(@)(H_(2)O)` or `Lamda_(m)^(@)(HCl)+Lamda_(m)^(@)(KOH)-Lamda_(m)^(@)(KCl)=Lamda_(m)^(@)(H_(2)O)` * Hence, by knowing the `Lamda_(m)^(@)` values of HCl, NaOH and NaCl, the `Lamda_(m)^(@)` value of water can be determined. |
|
| 50. |
Suggest a suitable oxidising agent for the conversion, (CH_(3))_(2)C=CHCOCH_(3)to(CH_(3))_(2)C=CHCO_(2)H |
Answer» Solution :ALKALINE `KMnO_(4)`, acidified `K_(2)Cr_(2)O_(7)` or `HNO_(3)` cannot be used since all of these will cleave the molecule at the SITE of the double bond giving a mixture of ketones/acid. The most suitable reagent for this oxidation is NaOI `(I_(2)//NaOH)` since methyl ketones on treatment with NaOI undergo iodoform reaction to GIVE iodoform along with the NA salt of a carboxylic acid having one carbon atom less than the starting methyl ketone. 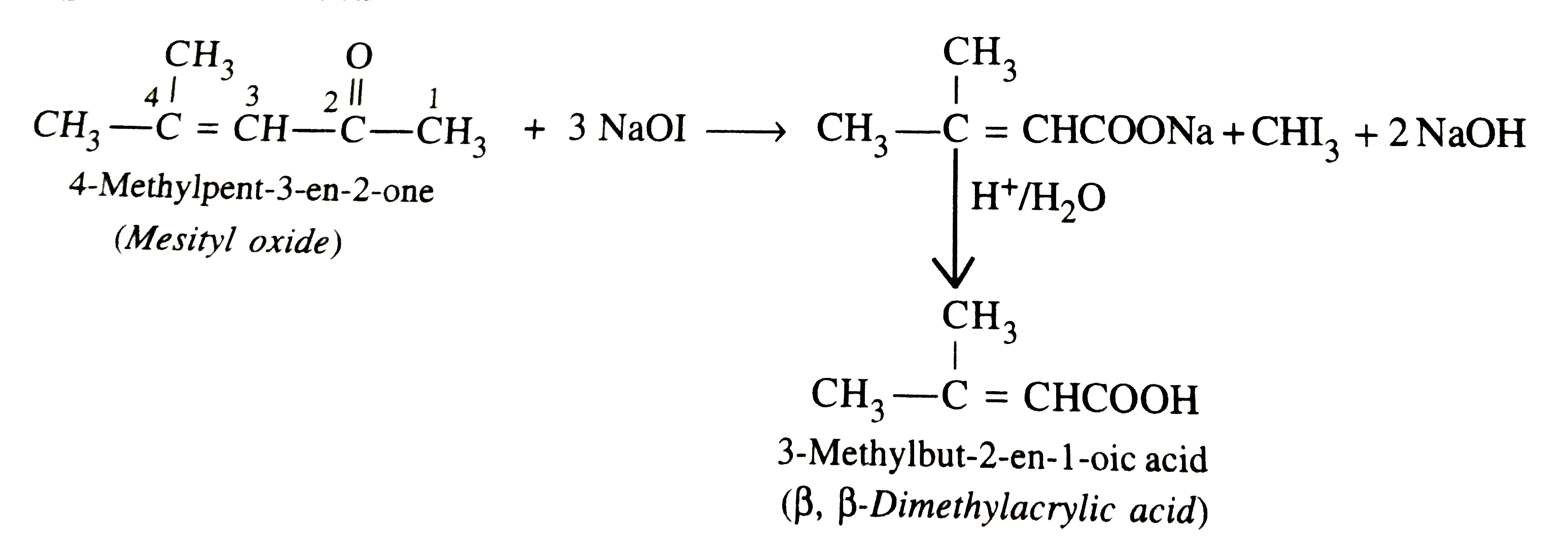 . .
|
|