Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Statement 1 : Entropy of solution is less than entropy of pure solvent. Statement 2 : The freezing point of water is depressed by the addition of glucose. |
|
Answer» Statement 1 is true, statement 2 is true, statement 2 is a correct explanation for statement 1 ENTROPY of solution is GREATER than entropy of solvent due to PRESENCES of greatere number of particles in solution. |
|
| 2. |
Statement-1 : Electrons releasing group at para position of a migrating aryl group enhances. The rate of reaction in Hofmann's preparation of primary amine from amides. Statement-2: |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1 |
|
| 3. |
Statement-1 : Energy is released in the nuclear fusion of hydrogen nuclei to form helium nuclei Because Statement-2 : Binding energy per nucleon of helium greater than hydrogen. |
|
Answer» STATEMENT-1 is TRUE, Statement-2 is TURE, statement-2 is a correct explanation for statement -1 |
|
| 4. |
Statement-1 : Electronegativity of an element depends on ionisation energy of that element. Statement-2 : Electroegativity regularly decreases along the group. Statement-3 : Fluorine is the most electronegative element. |
|
Answer» T F T |
|
| 5. |
Statement - 1 : Durning the reduction of ZnO to Zn, C is more efficient than CO. And Statement - 2 : The standard free energy of formation of CO_(2) from CO is always higher than that of ZnO. |
|
Answer» Statement - 1 is TRUE, Statement - 2 is True, Statement - 2 is correct explanation for statement - 1. |
|
| 6. |
Statement 1: During the electrolysis of water,two faraday of charge will produce a total of 336 litre of gases at S.T.P. at electrodes. Statement 2: Two faraday of charge will produce half mole of H_(2) and one fourth mole of O_(2) gas. |
|
Answer» Statement 1 is true, statement 2 is true, statement 2 is a correct explanation for statement 1 `V_(EQ)(H_(2))=11.2L` at S.T.P. `V_(eq)(O_(2))=5.6L` at S.T.P. Total volume liberated by 2 faraday cahrge `=2xx11.2+2xx5.6` `=33.6L ` at S.T.P. |
|
| 7. |
Statement-1: Electrolysis of an aqueous solution fo KI gives I_(2) at the anode but that of KF gives O_(2) at the anode and not F_(2). Statement-2: F_(2) is more reactive than I_(2). |
|
Answer» Statement-1 is True, statement-2 is true, statement-2 is a correct explanation of statement-1 |
|
| 8. |
Statement- 1: Duringthereductionof ZnOto Zn, C is moreefficientthan CO. Statement- 2 : Thestandardfree energyofformationofCO_ 2from COisalwayshigherthatthat of ZnO. |
|
Answer» STATEMENT - 1 is True, Statement -2 ISTRUE, Statement-2iscorrectexplanationofStatement -1. |
|
| 9. |
Statement-1 : During irreversible adiabatic expansion of ideal gas entropy must increase. Statement-II : Increase in entropy of the system due to expansion is only partially compensated by decrease in temperature. |
|
Answer» Statement-I is TRUE, statement-II is true and statement-II is CORRECT EXPLANATION for statement-I |
|
| 10. |
Statement-1: Dipole moment of H_(2)O is more than that of OF_(2) Statement-2: In H_(2)O , the resultant bond dipole of O-H bond and the resultant lone pair moment are in opposite direction. |
|
Answer» STATEMENT -1 is TRUE, Statement-2 is true and statement-2 is CORRECT explanation for statement -1. |
|
| 11. |
STATEMENT-1: During calcinations of haematite ore, ferrous oxide is oxidized to ferric oxide, and STATEMENT-2: Ferric iron does not form slag during smelting and loss of iron is prevented. |
|
Answer» STATEMENT - 1 is TRUE, STATEMENT-2 is True, STATEMENT -2 is correct explanation for STATEMENT-1 |
|
| 12. |
Statement-1 : Due to adiabatic free expansion, temperature of a real gas always increases Statement-II : If a real gas is at inversion temperature then no change in temperature is observed in adiabatic free expansion. |
|
Answer» Statement-I is TRUE, statement-II is true and statement-II is CORRECT EXPLANATION for statement-I |
|
| 13. |
Statement-1 Diamond is tetrahedral, graphite is planar and C_(60) has bucky ball structures. Statement-2 Carbon in diamond, graphite and C_(60) is sp^(3),sp^(2) and sp hybridised respectively. |
|
Answer» If both the statement are TRUE and Statement -2 is the correct explanation of Statement-1: |
|
| 14. |
Statement 1: Diisopropyl ketone on reaction with isopropyl magnesium bromide followed by hydrolysis gives 2^(@) alcohol. Statement 2: Grignard reagent acts as a reducing agent. |
|
Answer» a.Statement 1 and Statement 2 are true and Statement 2 is the correct explanation of Statement 1. 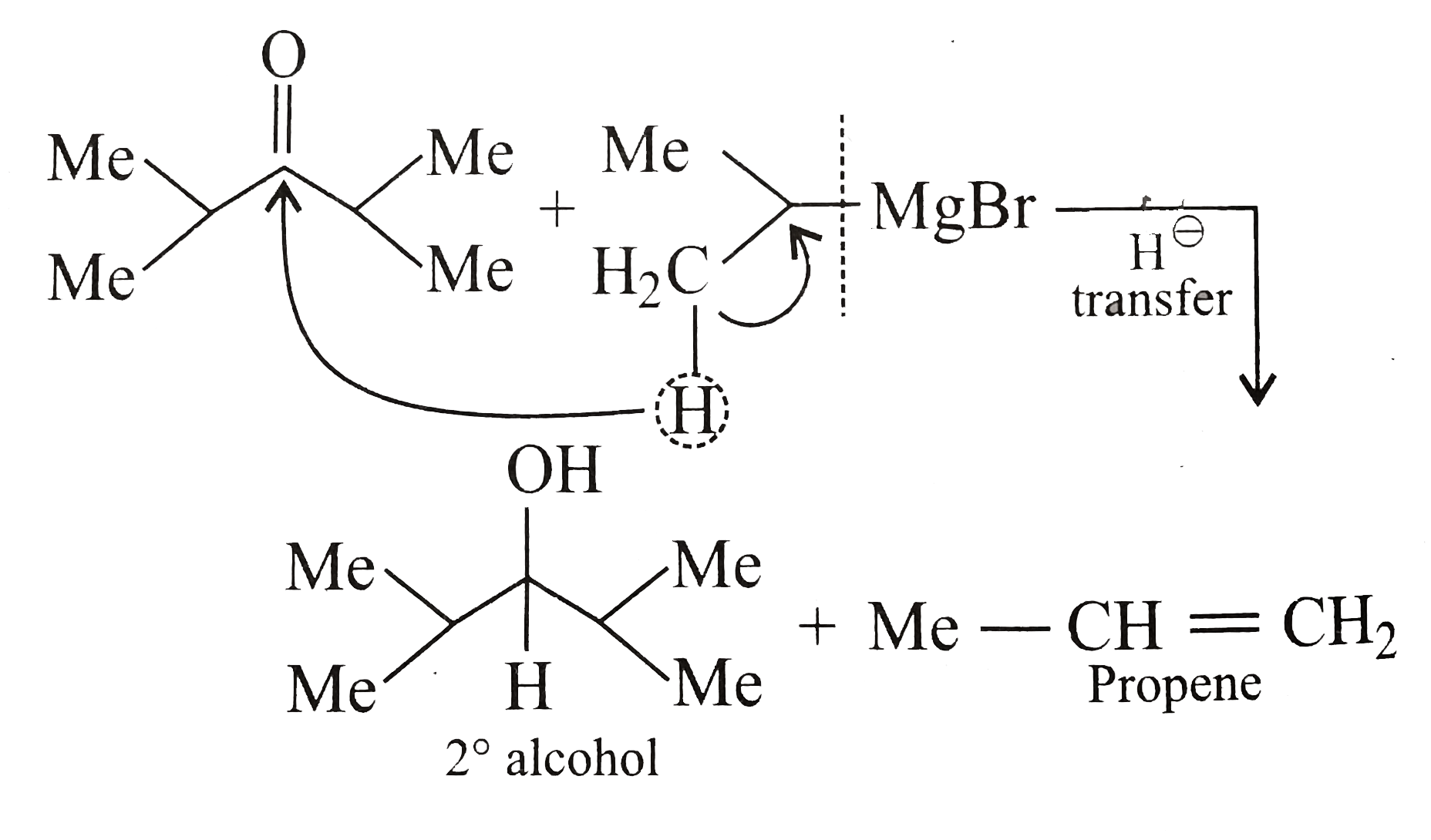 Hence, the G.R. acts as a REDUCING agent because the REACTION takes place via HYDRIDE ION `(H^(ɵ))` transfer to KETONE and G.R. itself is converted to alkene. Both atatements are true, so the answer is `(a)`. |
|
| 15. |
Statement-1: Diamonds are forever is generally quoted for diamond as rate of conversion of diamond to graphite at room condition is nearly zero. Statement-2 :At room condition, conversion of diamond into graphite is spontaneous. |
|
Answer» Statement-1 is True, Statement-2 is True, Statement-2 is a CORRECT explanation for Statement-1 |
|
| 16. |
Statement 1: Diamond and graphite do not have the same crystal structure. Statement 2: Diamond is crystalline while graphite is amorphous. |
|
Answer» Statement 1 is true, statement 2 is true: statement 2 is a correct EXPLANATION for statement 1. |
|
| 17. |
Statement 1: D-Cl bond is stronger than H-Cl bond. statement 2: Chlorine reacts more rapidly with H_(2) than with D_(2). |
|
Answer» STATEMENT 1 is true, statement 2 is true, statement 2 si a correct explanation for statement 15 |
|
| 18. |
Statement -1: DeltaH of a reaction is independent of temperature. and Statement -2: DeltaH^(@) of a reacction =sum H^(@) ("products") - sum H^(@)("reactants") |
|
Answer» STATEMENT -1 is true, Statement -2 is true, Statement -2 is correct explanation for statement -4 |
|
| 19. |
STATEMENT - 1 : Cyclohexanol is less souble in water than 1-hexanol. STATEMENT - 2 : 1- hexanol can from intermoleculae H-bond with H_(2)O. |
|
Answer» Statement - 1 is TRUE, Statement - 2 is Ture, Statement-2 is a correct explanation for Statement-1. |
|
| 20. |
Statement-1: Cyanides are more polar than isocyanides. and Statement-2: Isocynides show addition of oxygen or sulphur with HgO or HgS respectively. |
|
Answer» Statement-1 is TRUE, Statement-2 is true, statement-2 is a CORRECT EXPLANATION for statement-9 |
|
| 21. |
Statement-1: [Cu(NH_(3))_(4)]^(2+)" and "[Cu(CN_(4))]^(3-) ions are coloured. Statement-2: [Cu(NH_(3))_(4)]^(2+) involves dsp^(2) hybridization with one unpaired electron. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a correct EXPLANATION for Statement-1. |
|
| 22. |
Statement 1:CuSO_(4).5H_(2)O(s)hArrCuSO_(4).3H_(2)O(s)+2H_(2)O(g). For this equlibrium partial pressure of H_(2)O(g) increases continously with increasing volume. Statement 2: Vapour pressure depends only on temperarture. |
|
Answer» Statement 1 is TRUE, stetement 2 is true,statement 2 is a CORRECT explanation for statement 3 |
|
| 23. |
Statement-1: CuCl_(2) gives s deep blue coloured solution with ethylamine. Statement-2: Ethylamine molecules coordinate with cupric ions forming a blue coloured complex. |
|
Answer» Statement-1 is TRUE, statement-2 is true, statement-2 is a CORRECT EXPLANATION for statement-1 |
|
| 24. |
STATEMENT - 1 : Cu^(+2) is unstable in presence of Cl^(-) ion. and STATEMENT - 2 : Formation of CuCl is more favourable. |
|
Answer» Statement-1 is TRUE, Statement-2, is True, Statement-2 is a CORRECT EXPLANATION for Statement-9 |
|
| 25. |
Statement-1: Cs_(2)[CoF_(6)] is an inner orbital complex with one unpaired electron. Statement-2: According to the spectrochemical series F^(-) is weak field ligans. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a correct EXPLANATION for Statement-1. |
|
| 26. |
STATEMENT-1 : CrO_(2)Cl_(2) has tetrahedral shape . and STATEMENT-2: CrO_(3) reacts with HCl to formCrO_(2)Cl_(2) . |
|
Answer» Statement-1 is TRUE , Statement-2 is True , Statement-2 is a CORRECT explanation for Statement-1 |
|
| 27. |
Statement 1 : Critical temperature is the temperature at which a real gas exhibits ideal behaviour for considerable range of pressure. Statement 2 :At critical point the densities of a substance in gaseous and liquid states are same. |
|
Answer» Statement 1 is true, statement 2 is true , statement 2 is a correct explanation for statement 1 Critical temperature is the temperature above which the gas cannot be liquefied, how so ever HIGH pressure may be applied. `T_(C ) =(8a)/(27Rb)` |
|
| 28. |
Statement-1: [Cr(H_(2)O)_(6)]Cl_(2)" and "[Fe(H_(2)O)_(6)]Cl_(2) are reducing in nature. Statement-2: Unpaired electrons are present in their d-orbitals. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 29. |
STATEMENT-1: Cro, reacts with HCl to form chromyl chloride gas. and . STATEMENT-2: Chromyl chloride (CrO_(2)Cl_(2)) has tetrahedral shape. |
|
Answer» STATEMENT - 1 is TRUE, STATEMENT-2 is True, STATEMENT -2 is correct explanation for STATEMENT-1 |
|
| 30. |
Statement 1 : C_(P)-C_(V)=R for an ideal gas. Statement 2 :[(delE)/(delV)]_(T)=0 for an ideal gas. |
|
Answer» <P>STATEMENT 1 is true, statement-2 is true, statement 2 is a correct explanation for statement 6 |
|
| 31. |
STATEMENT-1: Considering the Vander Waal's equation of state (P+(a)/(V^(2)))(V-b) =RT for ammonia (NH_(3)) and nitrogen (N_(2)). The value of a for NH_(3) is larger than that for N_(2) and STATEMENT-2: Ammonia has a lower molecular weight than nitrogen. |
|
Answer» STATEMENT-1 is TRUE, STATEMENT-2 is True, STATEMENT-2 is CORRECT EXPLANATION for STATEMENT-1 |
|
| 32. |
Statement-1: Copper is dissolved at anode and deposited at cathode when Cu electrodes are used and electrolyte is 1M CuSO_(4)(aq) solution. Statement-2: SOP of Cu is less than SOP of water and SRP of Cu is greater than SRP of water. |
| Answer» SOLUTION :S.O.P of CU is greater than S.O.P of water `(H_(2)OtoO_(2))` and S.R.P. of Cu is greater than S.R.P of water `(H_(2)OtoH_(2))`. | |
| 33. |
Statement-1. Coordination entities with d^(4) to d^(7) ions are more stable for high spin state. Statement-2. If Delta_(0)ltP, high spin state is more stable. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a correct EXPLANATION for Statement-3 |
|
| 34. |
Statement-1: [Co(NO_(2))_(6)]^(4-)" and "[Co(NO_(2))_(6)]^(3-) both complex involve d^(2)sp^(3) hybridization but former is paramagnetic and later one diamagnetic. Statement-2: In [Co(NO_(2))_(6)]^(4-) one of the 3d electrons jumps to 4d orbital making the two d-orbitals empty for d^(2)sp^(3) hybridization. NO_(2) is a stronger ligand which favours the pairing of electrons and therefore, d^(2)sp^(3) hybridization. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 35. |
Statement-1: [Co(NH_(3))_(6)]^(2+) when exposed to air, is more easily oxidised into [Co(NH_(3))_(6)]^(3+) as compared to oxidation of Co^(2+) into Co^(3+) in gaseous state. Statement-2: In [Co(NH_(3))_(6)]^(2+) electron is to be removed from 4d, and in Co^(2+)(g), electron is removed from 3d. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 36. |
Statement-1 : Conservation of a gammaphoton into an electron and a positron is an example of pair production. Statement-2: Pair production refers to the creation of an elementary particle and its antiparticle , usually when a photon interacts with a nucleus. |
|
Answer» Statement-1 is TRUE, statement-2 is True, Statement-2 is a CORRECT EXPLANTION for Statement-1. |
|
| 37. |
Statement-1. Co(NH_(3))_(2)Cl_(3) gives white precipitate with AgNO_(3) solution. Statement-2. Chlorine is not present in the ionising sphere. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a correct explanation for Statement-8 |
|
| 38. |
Statement-1: [Co(NH_(3))_(5)SO_(4)]Br" and "[Co(NH_(3))_(5)Br]SO_(4) are ionization isomers. Statement-2: They can distinguished by adding AgNO_(3) solution and BaCl_(2) solution respectively. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 39. |
Statement-1: Conc. Solution of Li in NH_(3) is diamagnetic. Statement-2: Mg//NH_3 works as anti-hydrogenation agent. Statement-3: LiCl forms LiCl.2H_(2)O. |
|
Answer» TFT |
|
| 40. |
Statement-1: Complex ion [Co(NH_(3))_(6)]^(2+) si readily oxidized to [Co(NH_(3))_(6)]^(3+). Statement-2: Unpaired electron in complex ion [Co(NH_(3))_(6)]^(2+) is present in 4d orbital. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 41. |
Statement-1: Complexes of MX_(6)" and "MX_(5)L type (X and L are unidentate) do not show geometrical isomerism. Statement-2: Geometrical isomerism is not shown by complexes of coordination number 6. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 42. |
Assertion (A) Conductivity of all electrolytes decreases on dilution. Reason(R) On dilution number of ions per unit volume decreases. |
|
Answer» |
|
| 43. |
Statement-1 : Colour of lanthanide ions are pale in nature. Statement-2 : In the f-f transition change in subsidiary quantum number is zero. |
|
Answer» Statement-1 is TRUE, Statement-2 is True, Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 44. |
Statement-1: [Co^(II)(NH_(3))_(6)]^(2+) is not readily oxidised to [Co^(III)(NH_(3))_(6)]^(3+) When air is bubbled through it. Statement-2: Crystal field stabilization energy of Co(+III) with a d^(6) configuration is higher than for Co(+II) with a d^(7) arrangement. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 45. |
Statement-1: [Co(en)_(3)]^(3+) show stereoisomerism. Statement-2: EAN value of [Co(en)_(3)]^(3+) is 36. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT explanation for Statement-1. 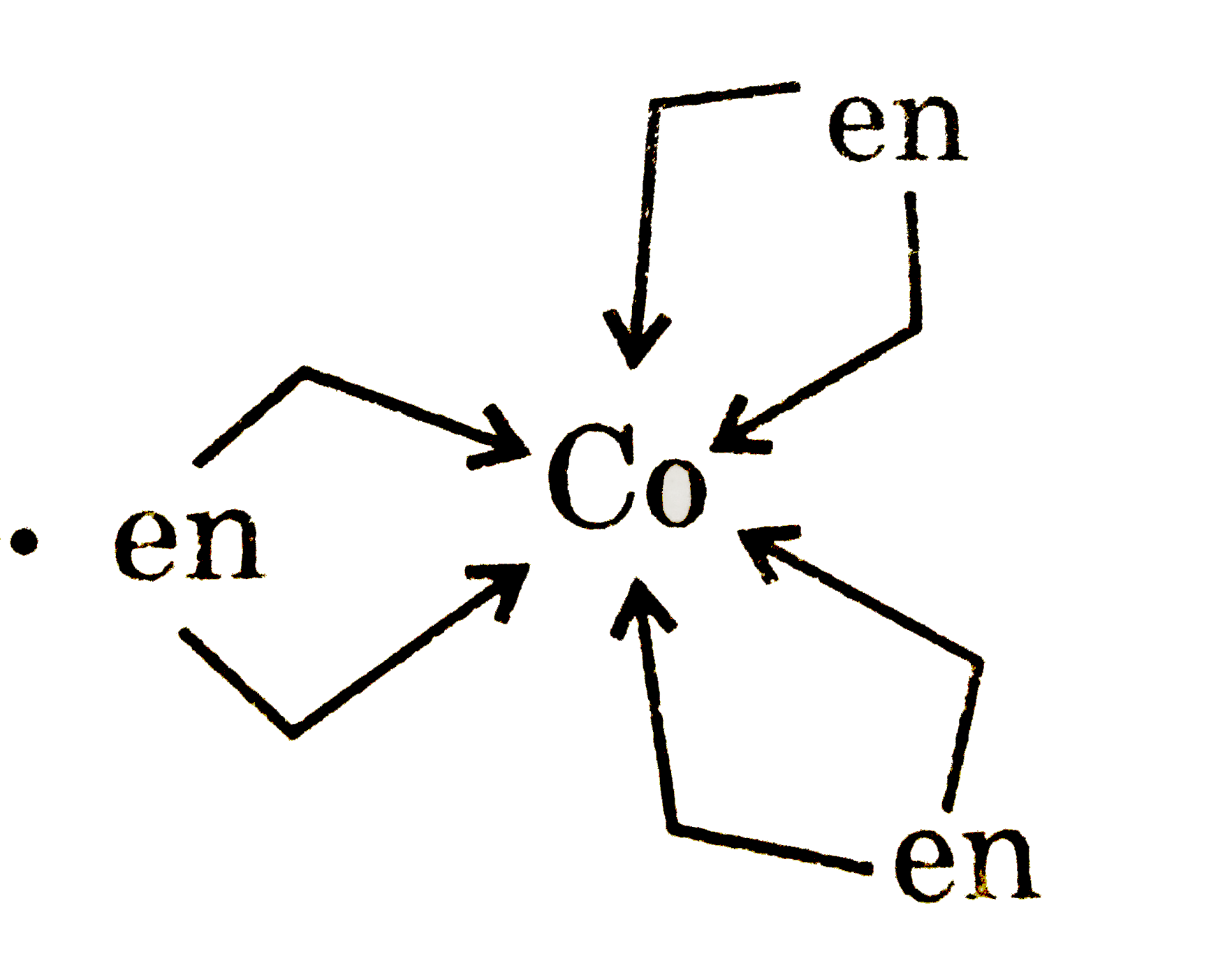 It is optically active so it cotains 2 stereoisomers `[Co(en)_(3)]^(3+)` EAN`rArr27-3+12=36` |
|
| 46. |
Statement-1: [Co^(II)(gly)_(3)] is called inner-metallic complex or inner complex. Statement-2: Both the coordination number and charge of the Both the coordination number and charge of the cation are satisfied simultaneously by ligands. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a correct explanation for Statement-1. |
|
| 47. |
Statement-1: CO, CN^(-), NO^(+) and phospines ligands act as pi acceptor. Statement-2: In [Mn^(VII)O_(4)]^(-)" and "[Cr^(VI)O_(4)]^(2-), the ligands act as pi donor and transfer charge from ligands to metal in pi interactions as well as sigma interactions. |
|
Answer» Statement-1 is TRUE, Statement-2 is True , Statement-2 is a CORRECT EXPLANATION for Statement-1. |
|
| 48. |
STATEMENT - 1 : Co^(2+) gives green colour with Br_(2) water in presence of KHCO_(3) and STATEMENT - 2 : Green colour is due to formation of CoCO_(3). |
|
Answer» Statement-1 is TRUE, Statement-2, is True, Statement-2 is a CORRECT EXPLANATION for Statement-5 |
|
| 49. |
Statement-1: Cleavage of anisole with HI at 373K gives phenol and CH_(3)I. Statement-2: due to resonance O-C_(6)H_(5) bond is stronger than O-CH_(3) bond. |
|
Answer» Statement-1 is TRUE, statement-2 is true, statement-2 is a CORRECT EXPLANATION for statement-1. |
|
| 50. |
Statement - 1 : ClF is more reactive than F_(2). Statement - 2 : The F-F bond is weaker than Cl - F bond. |
|
Answer» Statement-1 is True, Statement-2 is True , Statement-2 is a correct explanation for Statement-1. |
|