Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Square planar complexes with coordination number 4 exhibit geometrical isomerism whereas tetrahedral complexes do not. Why ? |
| Answer» SOLUTION :TETRAHEDRAL complexes do not exhibit geometrical isomerism because the RELATIVE positions of the ligands with respect to each other is the same. The SQUARE planar complexes on the other hand show geometrical isomerism. If same kind of ligands occupy positions adjacent to each other, it is called cis- form and if these are OPPOSITE to each other, it is called trans- form. | |
| 2. |
Square planar complexes with coordination number of four exhibit geometrical isomerism whereas tetrahedral complexes do not. Why ? |
| Answer» Solution :TETRAHEDRAL complexes do not GEOMETRICAL isomerism because the RELATIVE positions of the ligands attached to the central metal atom are same with respect to each other. | |
| 3. |
Square planar complexes of the type MABXL (where A, B, X and L are unidentates) show |
|
Answer» TWO CIS and ONE TRANS isomer 
|
|
| 4. |
Square planar complexes are generally formed by d^(8) ions with strong field ligands. The crystal field splitting is larger for second and third row transition elements and for more highly charged species. All the complexes having 4d^(8)" and "5d^(8) configuration are mostly square including those with weak field ligands such halide ions. Square planar complexes with coordination number four can show diastereoisomerism. Which one of the following square planar complexes will show geometrical isomerism ? |
|
Answer» `[Pt(EN)_(2)]^(2+)` |
|
| 5. |
Square planar complexes are generally formed by d^(8) ions with strong field ligands. The crystal field splitting is larger for second and third row transition elements and for more highly charged species. All the complexes having 4d^(8)" and "5d^(8) configuration are mostly square including those with weak field ligands such halide ions. Square planar complexes with coordination number four can show diastereoisomerism. Which of the following staements is true for the complex [Ni(PPh_(3))_(2)Br_(2)]? |
|
Answer» Hybridisation is the same as found in NICKEL `(+2)` complex with STRONG field ligands like CN. `[Ni(CO)_(4)]""sp^(3)` `[NiCl_(4)]^(2-) "" implies sp^(3)` |
|
| 6. |
Square planar complexes are generally formed by d^(8) ions with strong field ligands. The crystal field splitting is larger for second and third row transition elements and for more highly charged species. All the complexes having 4d^(8)" and "5d^(8) configuration are mostly square including those with weak field ligands such halide ions. Square planar complexes with coordination number four can show diastereoisomerism. Amongst the following complexes which has square planar geometry ? |
|
Answer» `[RhCl(CO)(PPh_(3))_(2)]` |
|
| 7. |
Square planar complexes have………… type of hybridisation. |
|
Answer» `SP^(3)` |
|
| 8. |
Square planar complexes are generally formed by d^(8) ions with strong field ligands. The crystal field splitting is larger for second and third row transition elements and for more highly charged species. All the complexes having 4d^(8)" and "5d^(8) configuration are mostly square including those with weak field ligands such halide ions. Square planar complexes with coordination number four can show diastereoisomerism. Which of the ions do not form the square planar complexes amongst the following ions ? |
|
Answer» `NI(+II)" and "Rh(+I)` with strong field ligands |
|
| 9. |
Square planar complex of the type MAXBL (where A, B, X and L are unidentate ligands) shows following set of isomers |
|
Answer» two cis and ONE trans 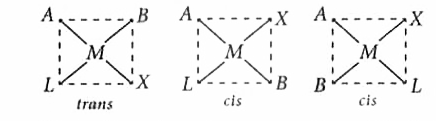 These above three isomers are formed by fixing the position of one LIGAND, A. |
|
| 10. |
Spring's reaction is / are |
|
Answer» `Na_(2)S+Na_(2)SO_(3)+I_(2) to Na_(2)S_(2)O_(3)+2NaI` |
|
| 12. |
Sprouting barley is the main source of ___ sugar. |
|
Answer» GLUCOSE |
|
| 13. |
Spraying of DDT on crops produces pollution of |
|
Answer» AIR only |
|
| 14. |
Spontanelity of any process can be predicated withthe help of DeltaS_("total").But this requires calculation of changes in system as will as surrounding. If some criteria (depanding upon the system only) can be developed for checking spontaneity unde specific conditions, then that would be a more useful parameter.The criteria can be derived from Clausius inequality. T ds ge q,(gt sign for irreversible process= sign for rev. process) or T dsgtdU-W for an irreversible process or T dS_(sys)gt dU_(sys)+p dV [consider no non-PV work] If U and V are constant, T dS_(sys)gt0 or (dS)_(u.v)gt0 for spontaneous process if V and T are constant T dS gt dU or dU-T dS lt 0 As temperature is constant, dU-d(TS)lt0 or d(U-TS)lt0 Another state function A(Helmholtz's function)=U-TS A decrease in Helmholtz function (A) under constant volume and temperature is the criteria of spontaneity of a process. For an irreversible cyclic process oint (dq)/T of system is : |
|
Answer» EQUAL to zero `oint(dq)/T=0` for REVERSIBLE `oint(dq)/Tgt0` for reversible |
|
| 15. |
Spontaneous adsorption of a gas on solid surface is an exothermic process because |
|
Answer» `DELTAH` increases for SYSTEM |
|
| 16. |
Spontanelity of any process can be predicated withthe help of DeltaS_("total").But this requires calculation of changes in system as will as surrounding. If some criteria (depanding upon the system only) can be developed for checking spontaneity unde specific conditions, then that would be a more useful parameter.The criteria can be derived from Clausius inequality. T ds ge q,(gt sign for irreversible process= sign for rev. process) or T dsgtdU-W for an irreversible process or T dS_(sys)gt dU_(sys)+p dV [consider no non-PV work] If U and V are constant, T dS_(sys)gt0 or (dS)_(u.v)gt0 for spontaneous process if V and T are constant T dS gt dU or dU-T dS lt 0 As temperature is constant, dU-d(TS)lt0 or d(U-TS)lt0 Another state function A(Helmholtz's function)=U-TS A decrease in Helmholtz function (A) under constant volume and temperature is the criteria of spontaneity of a process. If a state function G is defined such as G=H-TS, then dG must decrease for a spontaneou process (involving only PV work ) occuring at |
|
Answer» Constant VOLUME and TEMPERATURE `G=H-TS("at" dG lt 0)` |
|
| 17. |
Spontanelity of any process can be predicated withthe help of DeltaS_("total").But this requires calculation of changes in system as will as surrounding. If some criteria (depanding upon the system only) can be developed for checking spontaneity unde specific conditions, then that would be a more useful parameter.The criteria can be derived from Clausius inequality. T ds ge q,(gt sign for irreversible process= sign for rev. process) or T dsgtdU-W for an irreversible process or T dS_(sys)gt dU_(sys)+p dV [consider no non-PV work] If U and V are constant, T dS_(sys)gt0 or (dS)_(u.v)gt0 for spontaneous process if V and T are constant T dS gt dU or dU-T dS lt 0 As temperature is constant, dU-d(TS)lt0 or d(U-TS)lt0 Another state function A(Helmholtz's function)=U-TS A decrease in Helmholtz function (A) under constant volume and temperature is the criteria of spontaneity of a process. For a spontaneous process, if entropy and volume are constant, the internal energy of system must |
|
Answer» Increases or `dU LT 0` So internal ENERGY of SYSTEM decreases |
|
| 18. |
splitting of spectral lines under the influences of magnetic field is called |
|
Answer» PHOTO ELECTRIC effect |
|
| 19. |
Splitting of spectral lines when atoms are subjected to strong electric field is called : |
|
Answer» ZEEMAN effect |
|
| 20. |
Splitting energy (Delta_(0)) can usually be measured form the absorption spectra of the complex ions. In simple cases when light is absorbed by a complex ion, an electron in one of the lower energy orbitals is excited to one of the higher energy orbital. The energy corresponding to the frequency of absorbed light is equal to Delta_(0). If value of Delta_(0) for the complex is in visible region, the complex is coloured and the value of Delta_(0) lies in ultraviolet or infrared region, the complex is colourless. For octahedral complexes, the cyrstal field stabilisation energy is given by CFSE= +[-0.4t_(2g)N+0.6e_(g)N_(1)]Delta_(0) Where N and N_(1) are number of electrons in t_(2g)" and "e_(g) orbitals respectively. The value of CFSE can be used for the correction of the experimental values of heats of hydration of divalent can be obtained by substracting the calculated CFSE values form the experimental values. The heat of hydration of Cr^(2+) ion is -450k" "cal//mol. For [Cr(H_(2)O)_(6)]^(2+), Delta_(0)=13,900" "cm(-1). What heat of hydration would be, if there were no crystal field stabilisation energy ? |
|
Answer» `-426 k" "CAL//"MOLE"` |
|
| 21. |
Splitting energy (Delta_(0)) can usually be measured form the absorption spectra of the complex ions. In simple cases when light is absorbed by a complex ion, an electron in one of the lower energy orbitals is excited to one of the higher energy orbital. The energy corresponding to the frequency of absorbed light is equal to Delta_(0). If value of Delta_(0) for the complex is in visible region, the complex is coloured and the value of Delta_(0) lies in ultraviolet or infrared region, the complex is colourless. For octahedral complexes, the cyrstal field stabilisation energy is given by CFSE= +[-0.4t_(2g)N+0.6e_(g)N_(1)]Delta_(0) Where N and N_(1) are number of electrons in t_(2g)" and "e_(g) orbitals respectively. The value of CFSE can be used for the correction of the experimental values of heats of hydration of divalent can be obtained by substracting the calculated CFSE values form the experimental values. The value of crystal field splitting (Delta_(0)) for [Ti(H_(2)O)_(6)^(3+) is 243 kj mol^(-1). The crystal field stabilisation energy (CFSE) in this complex is : (in kj mol^(-1)) |
|
Answer» `-(3)/(5)xx243` CFSE`=-0.4xx1xxDelta_(0)` |
|
| 22. |
Splitting energy (Delta_(0)) can usually be measured form the absorption spectra of the complex ions. In simple cases when light is absorbed by a complex ion, an electron in one of the lower energy orbitals is excited to one of the higher energy orbital. The energy corresponding to the frequency of absorbed light is equal to Delta_(0). If value of Delta_(0) for the complex is in visible region, the complex is coloured and the value of Delta_(0) lies in ultraviolet or infrared region, the complex is colourless. For octahedral complexes, the cyrstal field stabilisation energy is given by CFSE= +[-0.4t_(2g)N+0.6e_(g)N_(1)]Delta_(0) Where N and N_(1) are number of electrons in t_(2g)" and "e_(g) orbitals respectively. The value of CFSE can be used for the correction of the experimental values of heats of hydration of divalent can be obtained by substracting the calculated CFSE values form the experimental values. Which of the following statements is correct ? |
|
Answer» ZINC (II) ION has a zero CFSE for any geometry. CFSE`=-1.2 Delta_(0)+0.6Delta_(0)=-0.6 Delta_(0)` NOTE :`349.75 cm^(-1)=1 KCAL//"mol"` `implies CFSE=-0.6xx(13900)/(349.75)"kcal"//"mol"` `=-23.84 kcal//mol~=-24` Heat of hydration withoutCFSE will be : `-450+24=-426 "kcal"//"ml"` |
|
| 23. |
Spirit of salt is: |
|
Answer» HBr |
|
| 24. |
Spinel is a important class of oxides consisting of two types of metal ions with the oxide ions arranged in CCP pattern. The normal spinel has one-eight of the tetrahedral holes occupied by one type of metal ion and one half of the octahedral hole occupied by another type of metal ion. Such a spinel is formed by Zn^(2+), Al^(3+) and O^(2–), with Zn^(2+) in the tetrahedral holes. Give the formulae of spinel. |
|
Answer» |
|
| 25. |
Spirit is 95% solution of |
|
Answer» Methanol |
|
| 26. |
Spinel is a important class of oxides consisting of two types of metal ions with the oxide ions arranged in CCP pattern. The normal spinel has one-eight of the tetrahedral holes occupied by one type of metal ion and one half of the octahedral hole occupied by another type of metal ion. Such a spinel is formed by Zn^(2+), Al^(3+)andO^(2–), with Zn^(2+) in the tetrahedral holes. Give the formulae of spinel. |
|
Answer» Solution :`ZN^(2+)=8//8=1` `Al^(3+)=4//2=2` `O^(2-)`=CCP(4) `ZnAl_(2)O_(4)` `Zu^(2+)=(1)/(8)xx8` octahedral voids `O^(2)`=CCParrangment(1+3)=4 |
|
| 27. |
Spinels are denoted with the formula MAl_2 O_4, where 'M' is |
|
Answer» Mg in + 2 state or Mn in + 2 state |
|
| 28. |
Spin only magnetic moment of the compound Hg(Co(SCN)_(4)is |
|
Answer» `sqrt3`  m`n=3` mu_(8)=SQRT(n(n+2))=sqrt(3xx5))=sqrt15` |
|
| 29. |
Spin only magnatic moment can be calculated by using mu=sqrt(n(n+2)) where 'n' represents |
|
Answer» PRINCIPAL QUANTUM No |
|
| 30. |
Spin only' magnetic moment of Ni in [Ni(dmg)_(2)] is same as that found in: |
|
Answer» `NI " in " [NiCl_(2)(PPh_(3))_(2)]` |
|
| 31. |
Spin only' magnetic moment of Ni in [Ni(dmg)_2] is same as that found in |
|
Answer» Ni in `[NiCl_2(PPh_3)_2]` `Pt(II)-5d^8` configuration .complex is square planar and THEREFORE, dimagnetic. So , `mu=0` |
|
| 32. |
Spin only' magnetic moment of Ni in [Ni(dmg)_(2)] is same as that found in : |
|
Answer» NI in `[NiCl_(2)(PPh_(3))_(2)]` `{MnO_(4)^(-),[Pt(H_(2)O)_(2)Br_(2)]" are diamagetic"}` |
|
| 33. |
Spinof elctron is |
|
Answer» ratation of electron about it 's own axis |
|
| 36. |
Spiegeleisen is an alloy of |
| Answer» Answer :A | |
| 38. |
Spherical gold paricles of radius 10nm, then the number of gold particles per mm^(3) of sol will be |
|
Answer» `1.9xx10^(12)` `=("mass")/("density") = (1.9xx10^(-4)g)/(19g cm^(-3)) = 1 xx 10^(5)cm^(-3)` Radius of gold sol particle `=10NM = 10 xx 10^(-9)m = 10xx10^(-7) cm = 10^(-6)cm` Volume of gold sol particle= `4/3 pi r^(3)` `4/3 xx 22/7 xx (10^(-6))^(3) = 4.19xx10^(-18) cm^(3)` No. of gold sol particle in `1 xx 10^(-5)cm^(3)` No, of gold sol particle in `1 xx 10^(-5) cm^(3)` `=(1xx10^(-5))/(4.19xx10^(-18)) = 2.38xx10^(12)` No. of gold sol particle in one `mm^(3)` `=(2.38xx10^(12))/(10^6) = 2.38 xx 10^(6)`. |
|
| 39. |
Sphaleriteis concentratedby |
|
Answer» GRAVITYSEPARATION |
|
| 40. |
Sphalerite and siderite are the ores of the metals |
|
Answer» AI and ZN |
|
| 42. |
Spermaceti is commonly used in : |
|
Answer» Fermentation of cane sugar |
|
| 43. |
Speeds of diffusion of carbon dioxide and unknown gas under identical conditions were 0.29 and 0.271 mL ("minute")^- respectively. Calculate the density of the gas if that of carbon dioxide is 1.964 gL^(-1) . |
|
Answer» |
|
| 44. |
Speigel, used in the manufacture of steel by the Bessemer process, is an alloy of..... |
|
Answer» iron, CHROMIUM and carbon. |
|
| 46. |
Specify the oxidation numbers of the metals in the following coordination entities : [PtCl_(4)]^(2-) |
|
Answer» Solution :Oxidation numbers can be obtained by adding the oxidation numbers of CONSTITUENTS and equating them with the overall charge on the ION, or zero, if it is a MOLECULAR complex. Assuming x as the oxidation number of the METAL, we have `x - 4 = - 2` or `x = +2` |
|
| 47. |
Spectomycin is |
|
Answer» an TRANQUILIZER |
|
| 48. |
Specify the oxidation numbers of the metals in the following coordination entities : (i) [Co(H_(2)O)(CN)(en)_(2)]^(2+) (ii) [PtCl_(4)]^(2-) (iii) [Cr(NH_(3))_(3)Cl_(3)] (iv) [CoBr_(2)(en)_(2)]^(+) (v) K_(3)[Fe(CN)_(6)] |
|
Answer» Solution :(i) x+(0)+(-1)+(0)=+2orx=+3 (ii) x-4=-2or x=+2 (III) x+0+3(-1)=0 or x=+3 (iv) x+2(-1)+0=+1 or x=+3 (V) `[Fe(CN)_(6)]^(3-)`, x+6(-1)=-3 or x=+3 |
|
| 49. |
Specify the oxidation numbers of the metals in the following coordination entities : K_(3)[Fe(CN)_(6)] |
|
Answer» Solution :Oxidation numbers can be OBTAINED by adding the oxidation numbers of constituents and equating them with the OVERALL charge on the ion, or zero, if it is a MOLECULAR COMPLEX. Assuming x as the oxidation number of the metal, we have `x + 6(-1) = - 3` or `x = +3.` |
|
| 50. |
Specify the oxidation numbers of the metals in the following coordination entities : [Co(H_(2)O)(CN)(en)_(2)]^(2+) |
|
Answer» Solution :Oxidation numbers can be obtained by adding the oxidation numbers of constituents and equating them with the overall CHARGE on the ion, or ZERO, if it is a molecular COMPLEX. Assuming x as the oxidation NUMBER of the METAL, we have `x + (0) + (-1) + (0) = +2` or `x = +3` |
|