Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Standard electrode potential are given as below: Fe^(+2)//Fe,E^(o)=-0.44"" Fe^(+3)//Fe^(+2),E^(o) = 0.77 IfFe^(+2),Fe^(+3)and Fe block are kept together then which is true? |
|
Answer» `FE^(+3)` increases |
|
| 2. |
Standard electrode potential are E_(Fe^(2+)|Fe)^(@)=-0.44V,E_(Fe^(3+)|Fe^(2+))^(@)=0.77V. If Fe,Fe^(2+) and Fe^(3+) are kept together, then |
|
Answer» `FE^(3+)` INCREASES |
|
| 3. |
Standard electrode potential. |
| Answer» Solution :It is defined as the DIFFERENCE of electrical potential between metal electrode and the solution around it at EQUILIBRIUM when all the SUBSTANCES involved in the electrode reaction are in their standard states of unit activity or CONCENTRATION at constant temperature. | |
| 4. |
StandardE^@ of the half cell Fe/Fe^(2+) is +0.44V and standard E^@ of half cell Cu /Cu^(2+) is -0.32V then: |
|
Answer» Cu oxidises `Fe^(2+)` ION |
|
| 5. |
Standard cell electrode potential is expressed as |
|
Answer» `E_(cell)^(0)=E_(red(R.H.S.))^(0)-E_("Oxi"(L.H.S))^(0)` |
|
| 6. |
Standaard electrode potential of three metals X , Y and Z are - 1.2 V, +0.5 V and - 3.0 V respectively . The reducing power of these metals will be |
|
Answer» `YgtZgtX` `ZgtXgtY` |
|
| 7. |
Standard boiling point of water is |
|
Answer» 100°C |
|
| 8. |
Stainless steel is non-corrosive. This character is more prominent in |
|
Answer» MN steel |
|
| 9. |
Stainless steel in an alloy steel of the following metals |
|
Answer» Fe only |
|
| 10. |
Stainless steel does not rust because |
|
Answer» N' present in it does not rust |
|
| 11. |
Stainless steel does not rust because : |
|
Answer» Nickel present in it does not RUST |
|
| 12. |
Stainless steel contain |
|
Answer» Au |
|
| 14. |
stage comes when no more sugar dissolves, instead it settles down at the bottom of the solution is now said to be |
|
Answer» condensed |
|
| 15. |
Stachyose on hydrolysis gives two moles of |
|
Answer» glucose |
|
| 16. |
Stachyose is which of the following type of saccharide ? |
|
Answer» OLIGOSACCHARIDE |
|
| 17. |
Stachyose is an example of |
|
Answer» MONOSACCHARIDES |
|
| 18. |
Stachyose is a |
|
Answer» monosaccharide |
|
| 19. |
Stachyose has formula |
|
Answer» `C_(12)H_(22)O_(11)` |
|
| 20. |
Stable oxidation state of Yb is |
|
Answer» `+2` |
|
| 24. |
Stable nuclides are those whose n/p ratio is |
|
Answer» `n//p = 1` |
|
| 25. |
Stable compounds in +1 oxidation state ore formed by : |
|
Answer» B |
|
| 26. |
Diazotisation means the conversion of |
|
Answer» any primary AMINE into diazonium salt USING `NaNO_(3)+HCI` at ice cold temperature |
|
| 27. |
Stable complex based on EAN rule (i) K_(4)[Fe(CN)_(6)] (ii) [Co(NH_(3))_(5)Cl]Cl_(2) (iii) [Ni(CO)_(4)] (iv) K_(3)(Ni(CN)_(4)] |
| Answer» Answer :A | |
| 28. |
Stabilization of oxidation state in Lanthanoid elements depends on which of the following? |
|
Answer» HYDRATION ENTHALPY and IONIZATION ENERGY |
|
| 29. |
Stability order of these benzyl carboncations is |
| Answer» Answer :A | |
| 30. |
Stability order of monovalent cations Ga^+, In^+,Tl^+ is given as |
|
Answer» `Ga^+gtIn^+gtTl^+` |
|
| 31. |
Stability of the species Li_(2) , Li_(2)^(-)and Li_(2)^(+) increases in the order of :- |
|
Answer» `Li_(2)^(-) LT Li_(2)^(+) lt Li_(2)` |
|
| 32. |
Stability of the species Li2 , Li2−and Li2+increases in the order: |
|
Answer» `Li_(2)lt Li_(2)^(+)lt Li_(2)^(-)` `Li_(2) : KK (sigma_(2s))^(2)B.O = 1` `Li_(2)^(+) : KK(sigma_(2s))^(1)B.O=0.5` `Li_(2)^(-): KK (sigma_(2s))^(2)(sigma_(2s)^(**))^(1)B.O=0.5` `Li_(2)^(+)` is more stable than `Li_(2)^(-)` because it has LESS NUMBER of antibonding electrons. HENCE,`Li_(2)^(-)lt Li_(2)^(+)lt Li_(2)`. |
|
| 33. |
Stability of monovalent and trivalent cations of Ga, In, TI lie in the following sequence |
|
Answer» `Ga^(3+) lt ln^(3+) gt Tl^(3+)` |
|
| 34. |
Stability of lyophilic coloids is due to |
|
Answer» same CHARGE on all the colloidal PARTICLES |
|
| 35. |
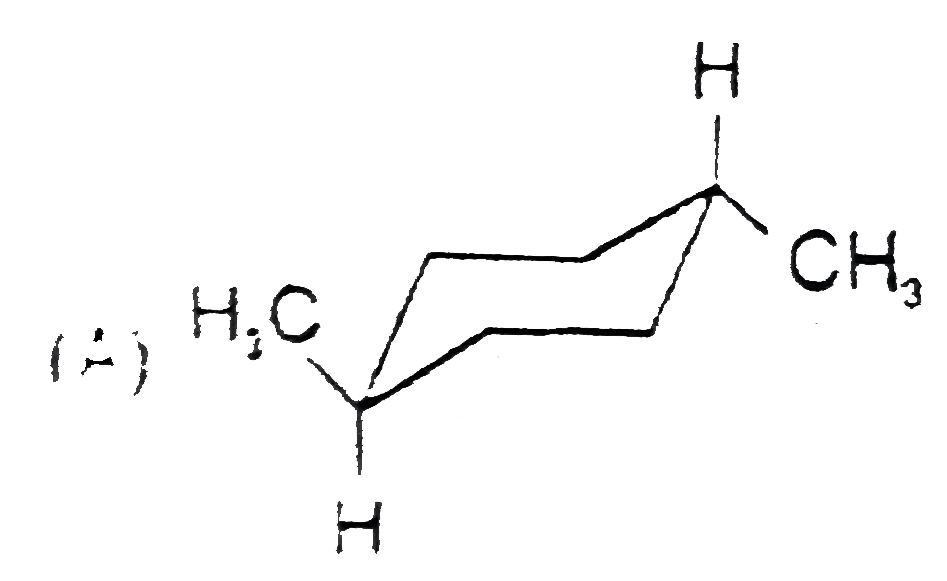
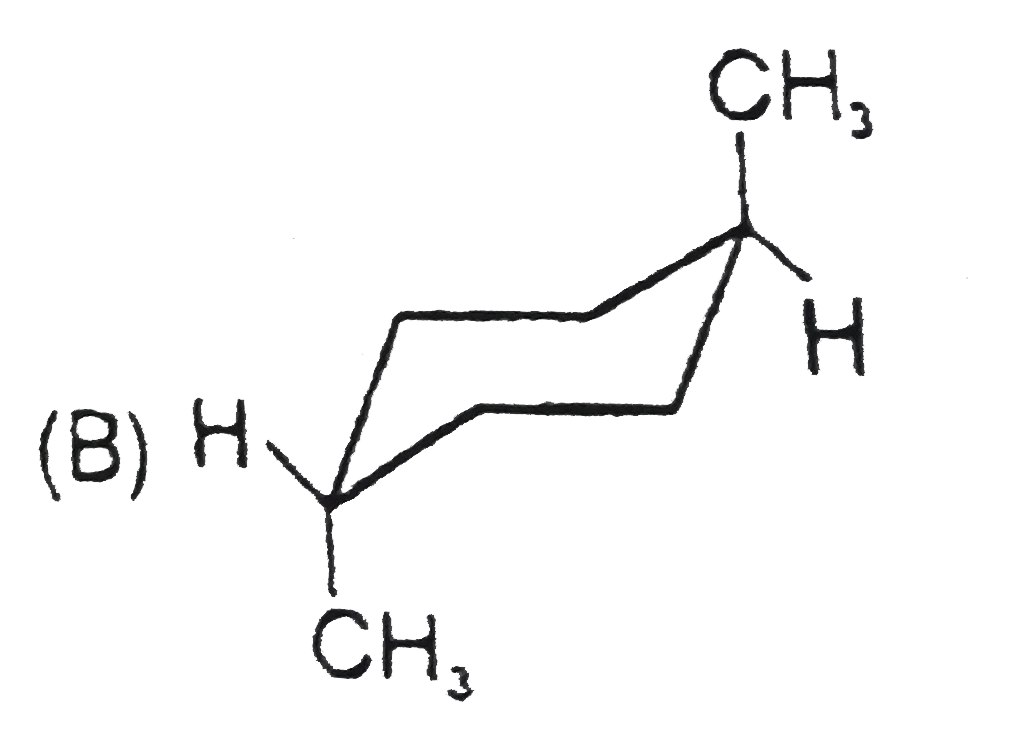
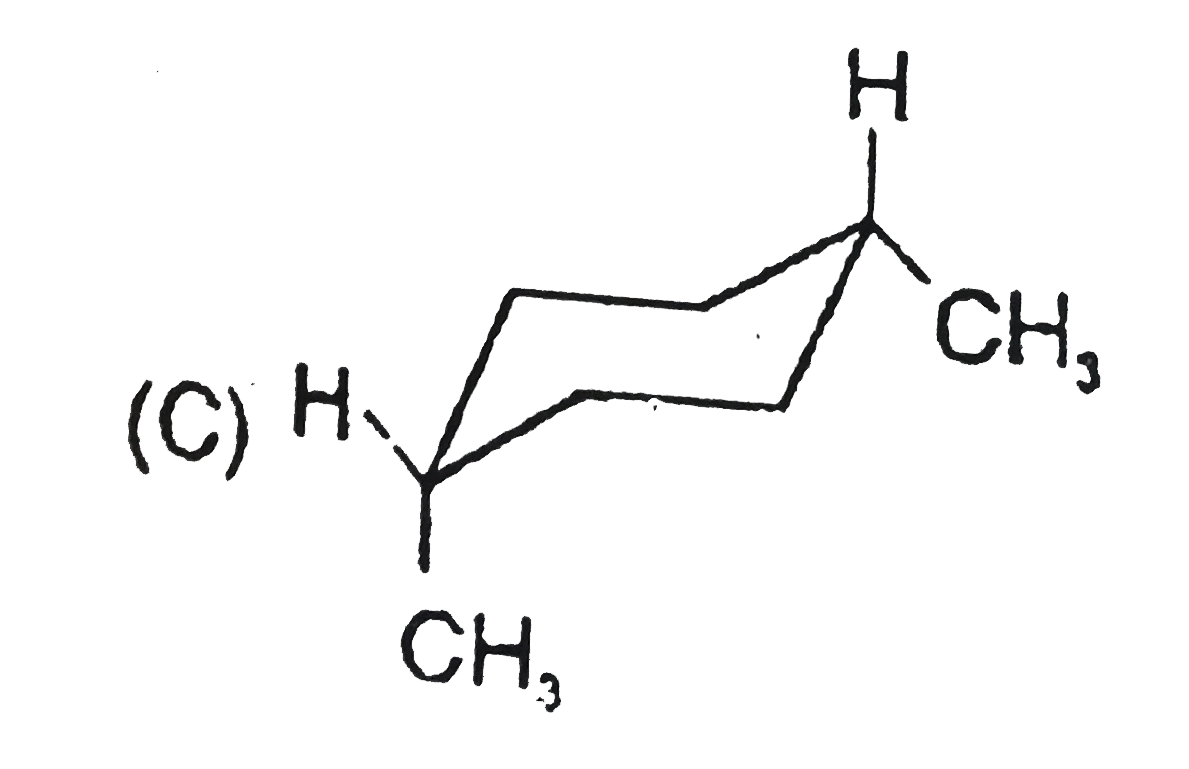
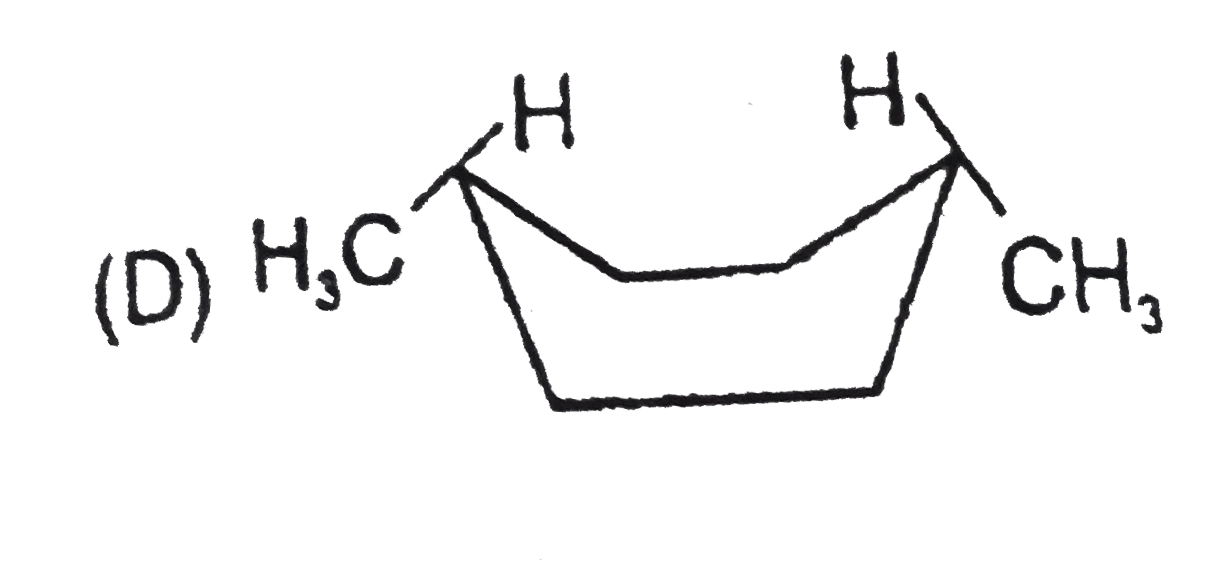
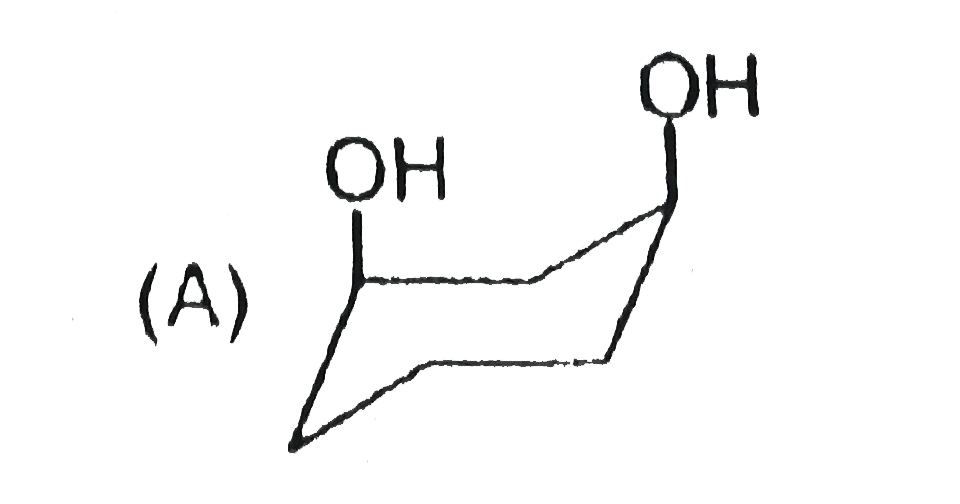
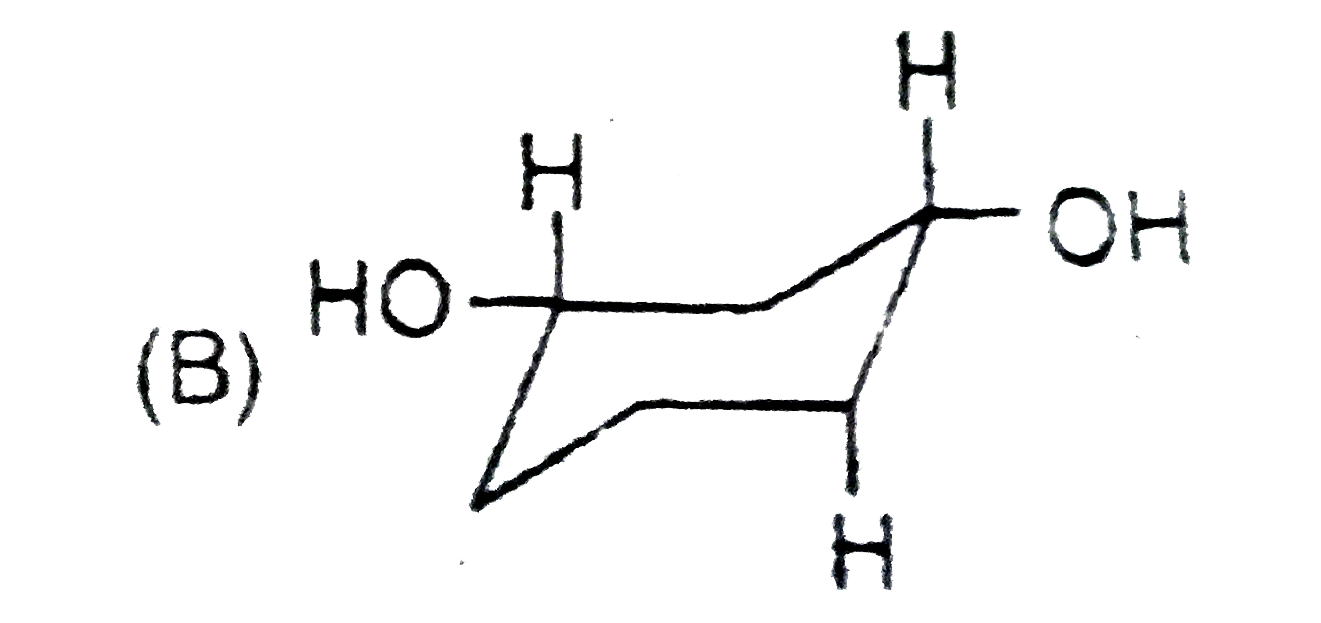
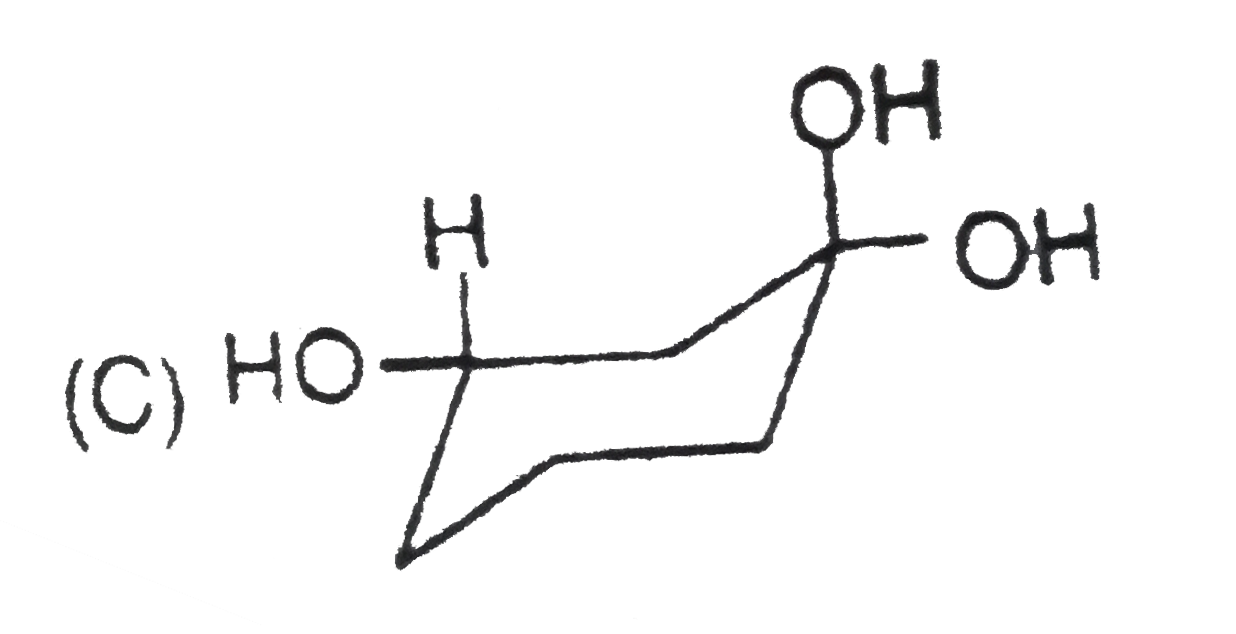
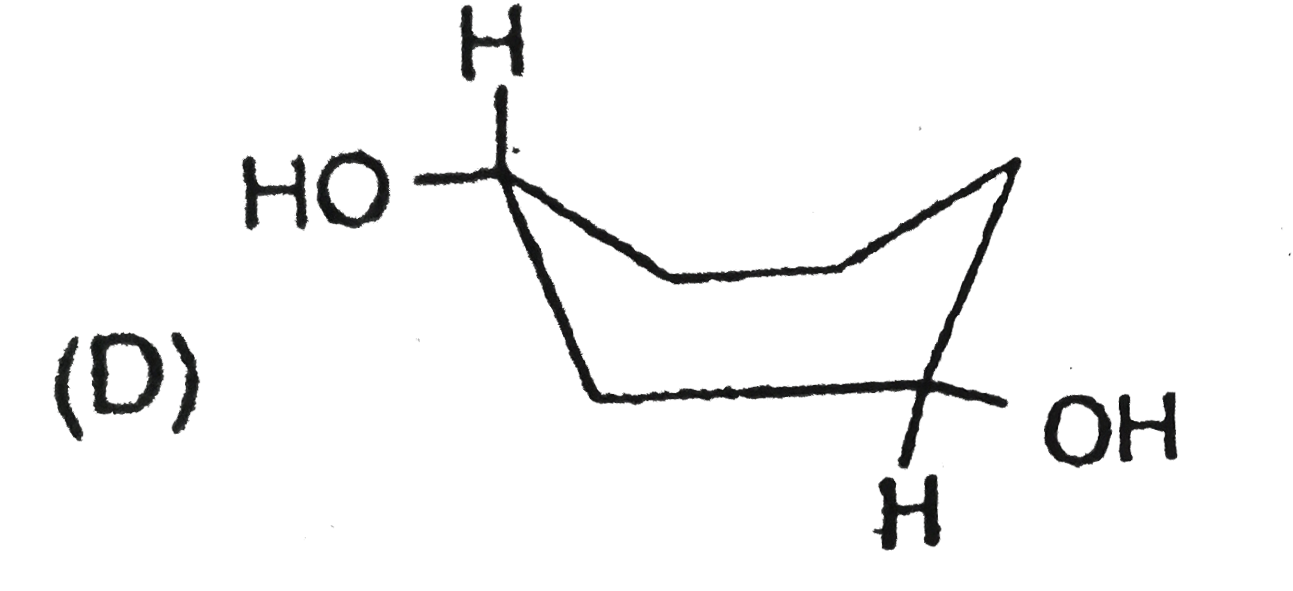
Stability of cycloalkanes can be explained on the basis of Bayer stain theory.This theory is applicable only for cyclopropane, cyclobutane and cyclopentane.Stability of cyclohexane and its derivatives can be explained by Mohr's theory According to this cyclohexane exists in two forms chair and boat.The chair form is more stable than the boat form.Every carbon of chair form has an axial and equatorial bonds.Bulky groups are generally present at equatorial position.The preferred conformation of cyclohexane ring is the chair form.but when intermolecular hydrogen bonding is possible between groups in 1 and 4 positions the molecule assumes a boat form. Which one of the following is most stable conformer of 1,4-dimethylcycohexane ? |
|
Answer»
|
|
| 36. |
Stability of hydrogen halide in the water |
|
Answer» `HF gt HCl gt HBr gt HI` |
|
| 37. |
Stability of cycloalkanes can be explained on the basis of Bayer stain theory.This theory is applicable only for cyclopropane, cyclobutane and cyclopentane.Stability of cyclohexane and its derivatives can be explained by Mohr's theory According to this cyclohexane exists in two forms chair and boat.The chair form is more stable than the boat form.Every carbon of chair form has an axial and equatorial bonds.Bulky groups are generally present at equatorial position.The preferred conformation of cyclohexane ring is the chair form.but when intermolecular hydrogen bonding is possible between groups in 1 and 4 positions the molecule assumes a boat form. Which one of the following is most stable ? |
|
Answer»
|
|
| 38. |
Stability of hydrates of carbonyl compounds depends on: |
|
Answer» Steric HINDRANCE |
|
| 39. |
Stability of carbanion follows the order. |
|
Answer» Q GT P gt S gt R |
|
| 40. |
‘Stability of a crystal is reflected in the magnitude of its melting points'. Comment. Collect melting points of solid water, ethyl alcohol, diethyl ether and methane from a data book. What can you say about the intermolecular forces between these molecules ? |
|
Answer» Solution : The stability of a crystal is reflected in the magnitude of MELTING points i.e., higher the melting point, stronger are the interparticle forces between the constituent particles and hence more is the stability of a crystal. The melting points of the given substances are as FOLLOWS : (i) Solid WATER = 273 K (ii) Ethyl alcohol : 155.7 K (iii) Diethyl ETHER = 156.8 K (iv) Methane = 90.5 K From these data, the magnitude of inter molecular forces are : Solid water > Diethyl ether `gt` Ethyl alcohol `gt` Methane The type of intermolecular forces present are : (i) Solid water `implies` H-bonding (4 H-bonds) (ii) Diethyl ether `implies` dipole-dipole interaction. (iii) Ethyl alcohol `implies`H-bond (3 H-bond) (iv) Methane `implies` London dispersion forces. |
|
| 42. |
"Stability of a crystal is reflected in the magnitude of its melting points.' Comment. Collect melting points of solid water, ethyl alcohol, diethyl ether and methane from a data book. What can you say about the intermolecular forces between these molecules ? |
|
Answer» Solution :Greater the intermolecular FORCES between the constituent particles, higher is the melting point and greater is the stability of the substance. Water and ethyl ALCOHOL have hydrogen bonds between the molecules. Water has higher melting point than alcohol because the former exhibits stronger hydrogen bonding. Intermolecular forces in diethyl ETHER are dipole-dipole interactions. There are only weak van der Waals forces acting in methane. Hence stability in DECREASING order can be arranged as : `"Water" lt" ethylalcohol "lt" diethyl ether "lt "methane"` |
|
| 43. |
Stability of a crystal is reflected in the magnitude of its melting points'. Comment. Collect melting points of solid water, ethyl alcohol, diethyl ether and methane from a data book. What can you say about the intermolecular forces between these molecules? |
|
Answer» Solution :For a crystal to melt, the attractive FORCES holding the constituent paticles in the crystal must be overcome. At the melting point of a crystal, thermal energies of the constiuent particles of the crystal overcome the attractice forces holding the particles, and in consequence the crystal STRUCTURE collapses and it melts. Therefore, a crystal in which the constituent particles are held by strong attractive forces requires more heat energy for its melting and hence, possesses a high melting point. The forces of attraction among the constituent particles in a crystal also determine the stability of the crystal because stronger forces of attraction favour the close-packing of the particle and which, in turn, increases the stability of the crystal. Therefore, a stable crystal must have a high melting point and we can say that a crystal's melting point reflects its stability.  (i) `H_(2)O` molecules are capable of forming H-bonds with one another. Hence, molecules in solid water experience intermolecular forces of attraction arising from H - bonds. In solid water experience dipole-dipole attraction and London forces. (ii) `C_(2)H_(5)OH` molecules are polar and capable oof forming hydrogen bonds with each other. So, the molecules in solid ETHANOL experience intermolecular forces of attraction due to hydrogen BONDING. In addition, the molecules in solid ethanol experience dipole-dipole attractions and London forces. (iii) `(C_(2)H_(5))_(2)O` molecules are polar but they cannot form hydrogen bond. In solid `(C_(2)H_(5))_(2)O`, molecules experience dipole-dipole attractions and London forces. (iv) In solid `CH_(4)`, molecules experience only London forces. Melting point of a solid depends on the strength of intermolecular forces holding the constituent particles in the solid. The stronger the forces of attraction in a solid, the higher the melting point it possesses. Strength of attractive forces due to hydrogen bonding is stronger than the dipole-dipole attractive forces, which in turn is stronger than London forces. Forces in solid `H_(2)O` due to hydrogen bonds are stronger than that in solid `C_(2)H_(5)OH` because the number of hydrogen bonds per molecule in solid `H_(2)O` is greater than that in solid `C_(2)H_(5)OH`. THus, the order of strength of intermolecular attractive forces in the given solids is `H_(2)OgtC_(2)H_(5)OHgt(C_(2)H_(5))_(2)OgtCH_(4)`. This result in the following order of melting points- `H_(2)OgtC_(2)H_(5)OHgt(C_(2)H_(5))_(2)OgtCH_(4)` |
|
| 44. |
Stabilisation energy of octahedral complex with d^7 configuration (A)1.8 Delta_0with one unpaired electron (B)1.8 Delta_0with three unpaired electron (C )0.8 Delta_0with one unpaired electron (D)0.8 Delta_0with threeunpaired electron |
| Answer» ANSWER :A | |
| 45. |
SRP of copper is less than that for silver. |
| Answer» SOLUTION :COPPER is above silver in electro chemical SERIES. | |
| 46. |
SRP of Sn^(2+)//Sn^(4+) is 0.5 , SRP of Au//Au^(3+) is 1.5, give reaction for the cell |
|
Answer» `2Sn^(2+)+3Au^(3+) to 2Sn^(4+)+3Au` THEREFORE `Au^(+3)` undergo reduction and `Sn^(2+)` undergo OXIDATION. |
|
| 47. |
S_("rhombic")+O_(2)(g)toSO_(2)(g), DeltaH = -297 .5 kcal S_("monoclinic")+ O_(2)(g) to SO_(2)(g) , DeltaH = -300 kJ |
|
Answer» rhombic sulphur is YELLOW in color |
|
| 48. |
SrCrO_(4)darr+2AcOH(excess)toSr(ArO)_(2)toNo dissolution |
|
Answer» For PRECIPITATE formation formation reaction |
|
| 49. |
Squashes are stored by adding |
|
Answer» Citric acid |
|