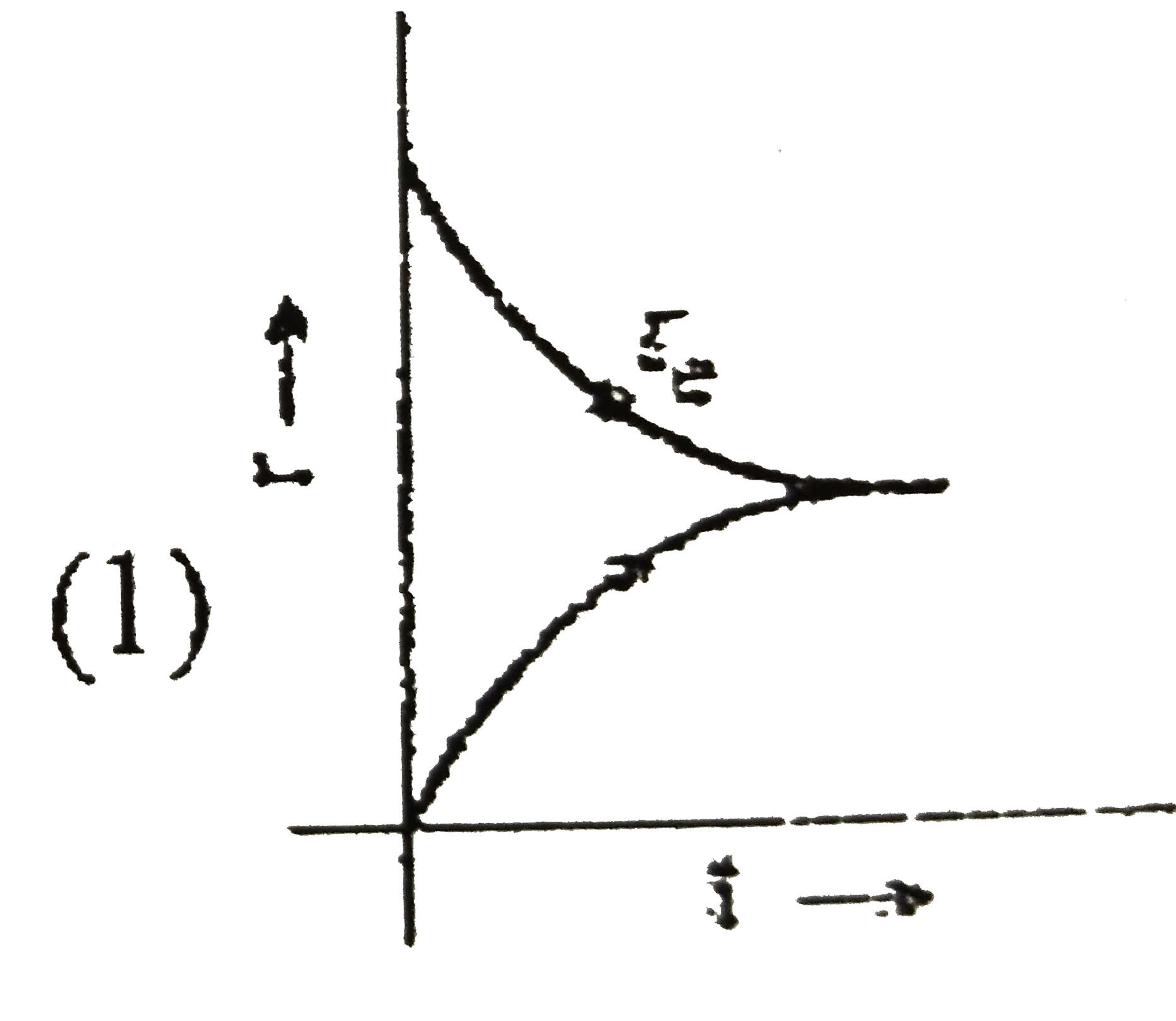Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Some of the metals with BCC structures are not close packed. Therefore, their densities would be greater if they were to change to a cubic close packed structure. What would be density of a metal if it had a cubic close packed structure rather than BCC? Its actual density is 19.3 g/ cc. |
|
Answer» Solution :`rho_("CCP")=(4M)/(N_(A)a_("CCP")^(3))` and`rho_("BCC")=(2M)/(N_(A)a_("BCC")^(3))`, `implies (rho_("CCP"))/(rho_("BCC"))=2((a_("BCC"))/(a_("CCP")))^(3)"`: Also in FCC `4r=sqrt(2a_("CCP"))` in BCC, `4r=sqrt(3)a_("BCC")` `implies (rho_("CCP"))/(19.3)=2((2)/(3))^(3//2)`=1.08867 `implies rho_("CCP")=21g//cc` |
|
| 2. |
Some of the metals occur in the native state in nature because they : |
|
Answer» Are REACTIVE |
|
| 3. |
Some of the lower quality ore are mixed with Haematite for Fe extraction like Siderate, Limonite and Magnetic. Roasted ore are mixed with coke and lime stone, finally heated in the blast furnace to get molten Fe. Choose the correct statement reagarding the roasting process here. |
|
Answer» It is illogical because there is no SULPHIDE ORE in extraction of Fe. |
|
| 4. |
Some of the following reagents are used as primary standard I. KMnO_(4) II. NaOH III . K_(2)Cr_(2)O_(7) IV. FeSO_(4)(NH_(4))_(2)SO_(4).6H_(2)O V. H_(2)C_(2)O_(4).2H_(2)O Select the primary standard |
| Answer» ANSWER :C | |
| 5. |
Some of the following statements are correct and some are incorrect. i) Both MnS and ZnS are soluble in dil. CH_(3)COOH and HCl ii) On adding of hot dilute acetic acid to a solution of barium oxalate and calcium oxalate only calcium oxalate dissolves. iii) Hg_(2)^(2) on reaction with ammonia solution give a black ppt. iv) Both Hg_(2)Cl_(2) and SnCl_(4) does not respond to chromyl chloride test. The wrong statements are : |
|
Answer» `(i),(III),(iv)` |
|
| 6. |
Some of the following sets of quantum numbers are correct for a 4d electron. Which are the correct sets? |
|
Answer» `4,3,2,+1/2` `l=2,m=-2,-1,0+1,+2,s=+1/2` or `-1/2` |
|
| 7. |
some of the following acids lack -COOH group but liberate CO_(2) with NaHCO_(3). These acids are |
|
Answer» ASCORBIC acid |
|
| 8. |
Some of the common steps involved in the metallurgical operations are I. refining II. Dressing of the ore III. Conversion of the ore into its oxides or other desired compounds. Choose the CORRECT order in which these steps are carried out. |
|
Answer» III , II , I |
|
| 9. |
Some metals are extracted from their oxides by reducing with aluminium instead of carbon-why? |
| Answer» SOLUTION :Because they FORM METAL CARBIDES, | |
| 10. |
Some meta-directing substituents in aromatic substitution are given which one is the most deactivating? |
|
Answer» `-NO_2` `-NO_2gt -C-= N gt -SO_3 H gt COOH` |
|
| 11. |
Some meta-directing substituents in aromatic substitution are given. Which is most deactivating- |
|
Answer» `-COOH` |
|
| 12. |
Some meta-directing substituents in aromatic substitution are given. Which one is most deactivating? |
|
Answer» `-SO_(3)H` `-NO_(2)gt-CNgt-SO_(3)Hgt-COOH` `-NO_(2)` group is most deactiveting group due to strong -E, -I and -M EFFECTS. |
|
| 13. |
Some large white transparent crystals are left out in bowel for several days. They are then observed to have changed their form into white powder. The crystal may have been of: |
|
Answer» AMMONIUM chloride |
|
| 14. |
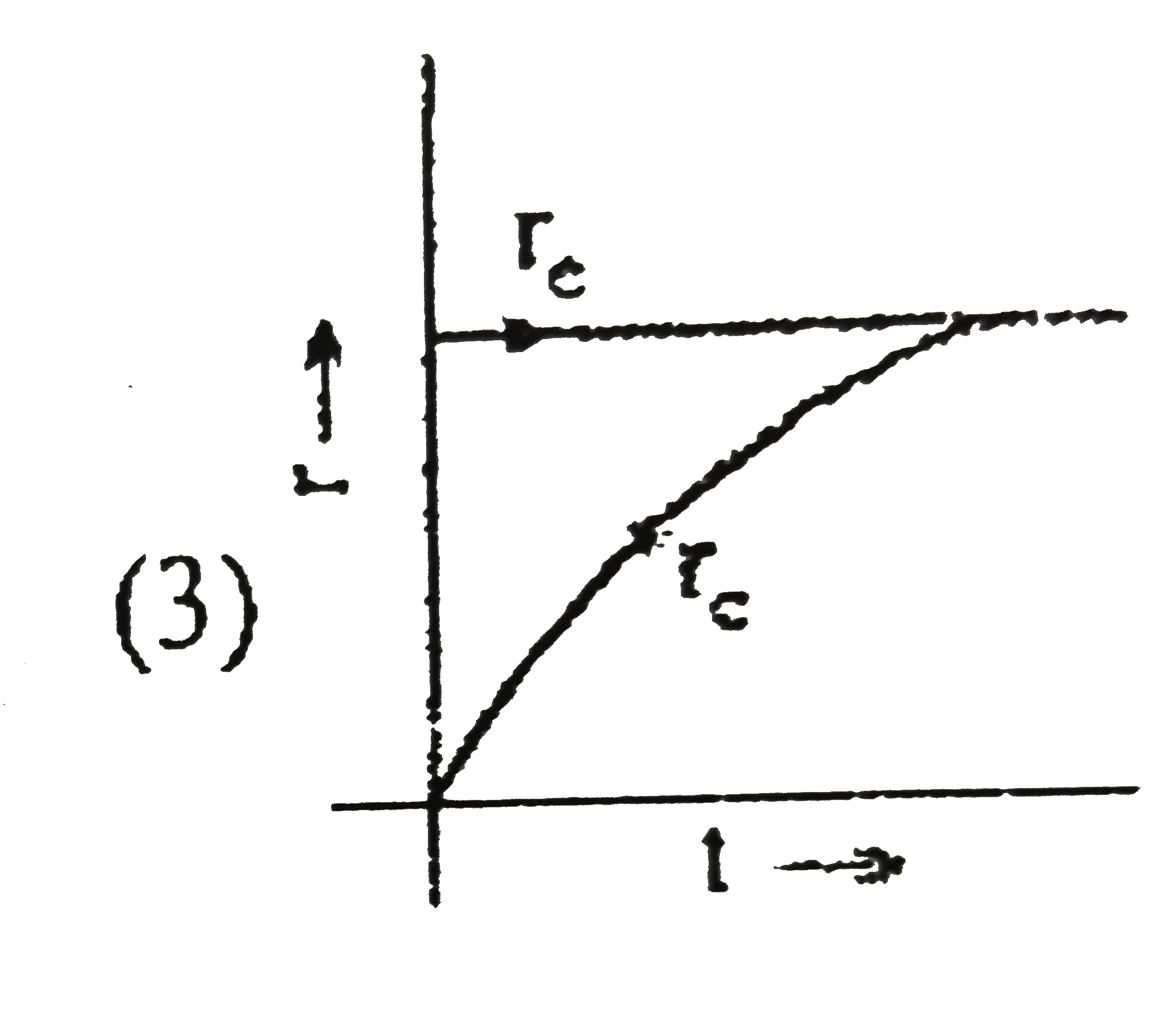
Some liquid is taken in an evacuted vessel and the vessel is seated. Which of the following graph will correctly represent the achievement of equilibrium of liquid with its vapour at constant temperature?[r_(c)= rate of vaporation r_(c)= rate of condensation] |
|
Answer»
`r_(expreation")=` constant `r_("conderation")=` INCREASES till equilibrium. 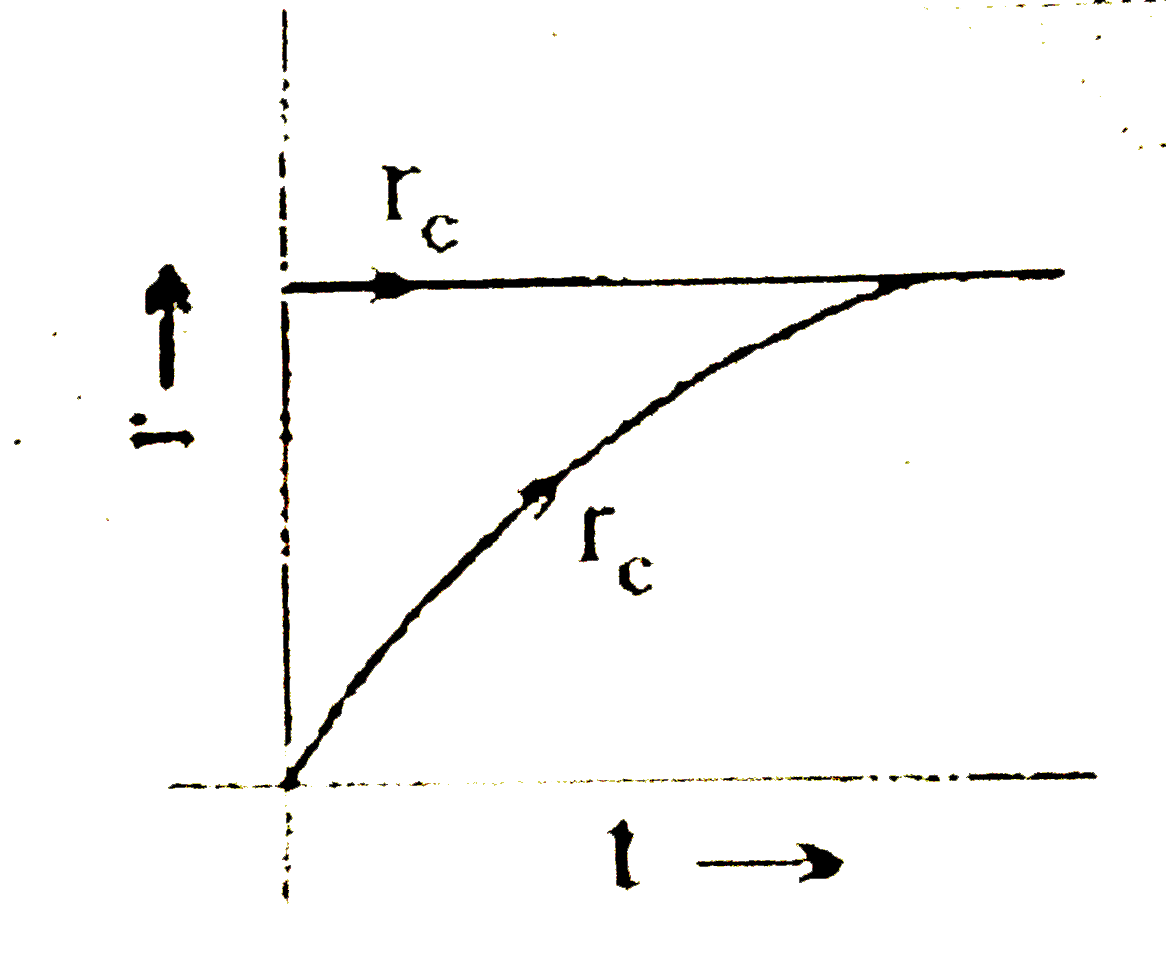
|
|
| 15. |
Some liquids on mixing form 'azeotropes'. Wh.at are 'azeotropes' ? |
| Answer» SOLUTION :Azeotropes : BINARY mixtures having same COMOPSITION in LIQUID and vapour PHASE and boil at a constant temperature. | |
| 16. |
Some halogens show +7 oxidation state in its exited state of configuration . What is its exited state. |
Answer» 
|
|
| 17. |
Some examples are given in List - II and their type is given in List - I{:("List - I","List - II"),("(1) Lipid","(A) Histidine"),("(2) Protein","(B) Ascorbic acid"),("(3) Amino acid","(C ) Cephalin"),("(4) Hormone","(D) Insulin"),("(5) Vitamin",):} |
|
Answer» `{:(A,B,C,D),(4,1,2,5):}` |
|
| 18. |
Some halogen containing compounds are useful in daily life. Some compounds of this class are responsible for exposure of flora and fauna to more and more of UV light which causes destruction to a great extent. Name the class of these halocompounds. In you opinion, what should be done to minimise harmful effects of these compounds. |
|
Answer» Solution : Halogen compounds are extremely useful in daily life and also in inductry. alkyl halides are regarded as synthetic tools for the synthesis of a variety of organic compounds in the laboratory. Similarly, compounds such as chloroform, iodoform, westron, westrosol and perfluorocarbons (P.F.C's) have found utility in industry. But at the sametime, class of compounds known as freons or chlorofluorocarbons (CFC's) which are otherwise quite useful as refrigerants, have done immense harm to our ENVIRONMENT. they have played a major role in depleting ozone layer. Even carbon tetrachlodie is a culprit to some extent. the depletion of ozone layer has exposed both plants and animals to the harmaful ultraviolet radiations (U.V. radiations). these radiations lead to aging of skin, early cataract, sunburns, skin CANCER etc. skin cancer is very common in a country like Australia where the effect of UV radiations is very high. Measures to reduce the harmful effect of these compounds . ltBrgt We have studied that the major cause of environmental pollution is the release of toxic chemicals which are formed as a result of the processes and reactions carried at various levels. in other words, chemists are mainly responsible for polluting the atmosphere although they also manufacture products that are source of our confort. This has forced them to change their outbook. since 1990, a new concept called green chemistry has been introduced. by green chemistry we mean "the production of substances of daily use by CHEMICAL reaction which neither emmploy toxic chemicals nor release the same to the atmosphere"No double, this is altogether a new field but some success has been achieved. efforts have been made to carry the reactions in the presence of ultra violet sunlight (known as photochemistry) and with sound waves (called sonochemistry), microwaves have been used to carry reactions which neither need toxic solvents nor release such vapours into the atmosphere. automobile engineers have been fitted with CATALYTIC converters which prevent the release of the vapours of hydrocarbons and oxides of nitrogen into the atmosphere. these are the real culprits since they form poisonous substances such as formaldehyde, acrolein and peroxyacetyl nitrate. these measures have CHECKED the release of chemicals into the environment which cause depletion of ozone layer. similarly, some enzymes have been employed as biocatalysts into the environment which cause depletion of ozone layer. similarly some enzymes have been emplyoyed as biocatalysts in the manufacure of certain antibiotics such as amppicillin and amoxycillin. a few noteworthy measures under the fold of green chemistry to check pollution are mentioned: (a). Dry cleaning of clothes. A commonly used dry cleaning solvent is tetrachloroethene `(Cl_(2)C=C Cl_(2))`. it pullutes water and is also carcinogenic. this has been replaced by some ther detergents which contains liquid carbon dioxide. these do not pollute water and give better results. these days, hydrogen peroxide is being used in the laundries for removing stains from clothes. (b). Bleaching of paper. chlorine gas was used earlier for bleaching of paper. it releases poisonous fumes in the atmosphere. At present, hydrogen peroxide is also used for this purpose. (c). Synthesis of chemicals. Ionic catalysts in the form of `Pd^(2+) and Cu^(2+)` salts have been empployed for the preparation of acetaldehyde (ethanal) from thene by carrying oxidation with oxygen. the yield of ethanal is excellent (about 90%) `underset("Ethene")(CH_(2)=CH_(2)+O_(2))overset(Pd^(2+)//Cu^(2+)" in water")tounderset("Ethanal")(CH_(3)CHO)` This is just the beginning and the results are encouraging. we are sure of our scientists, particularly the chemists will be successful in developing new techniques as well as chemicals to minimise pullution. all countries are giving importance to the study to biotechnology which has a major role in checking pollution. it is the duty of every individual to keep the environment free from pollution. |
|
| 19. |
Some grignard reagents react with ethyl orthoformate followed by acidic hydrolysis to give aldehydes. Propose mechanisms for the two steps in this synthesis. H-underset(O-CH_(2)CH_(3))underset(|)overset(O-CH_(2)CH_(3))overset(|)(C)-O-CH_(2)CH_(3)+R-Mg-X rarr R-underset(H)underset(|)overset(O-CH_(2)CH_(3))overset(|)(C)-O-CH_(2)CH_(3) overset(H_(3)O^(+))(rarr)R-overset(O)overset(||)(C)-H ethyl orthofomate""acetal"" aldehyde |
| Answer» SOLUTION : R —Mg—X | |
| 20. |
Some halogen containing compounds are useful in daily life. Some compounds of this class are responsible for exposure of flora and fauna to more and more of UV light which causes destruction to a great extent. Name the class of these halocompounds. In your opinion, what should be done to minimise harmful effects of these compounds. |
| Answer» Solution :A numbe rof polyhalogen coompounds, e.g., dichloromethane, CHLOROFORM, iodoform, carbon tetrachloride, FREON and DDT have many applications in our daily LIFE. Freions also cause depletion of ozone layer and thus allow more UV rays to reach the earth which, in turn, damage flora and fauna. to minimise the harmful effects of freons, hydrofluorocarbons and fluorocarbons which are stable in the STRATOSPHERE should be used in aerosol propellants and in air conditioning equipments. | |
| 21. |
Some glass objects from ancient civilisations look milky instead of being transparent. Explain. |
| Answer» SOLUTION :It is because of some CRYSTALLIZATION of the AMORPHOUS GLASS with TIME. | |
| 22. |
Some enzymes are named after the reaction, where they are used. What name is given to the class of enzymes which catalyse the oxidation of one substrate with simultaneous reduction of the other substrate ? |
| Answer» SOLUTION : Oxireductase. | |
| 23. |
Some enzymes are made after the reaction where they are used. What name is given to class of enzymes which catalyse the oxidation of one substrate with simultaneous reduction of another substrate ? |
| Answer» SOLUTION :OXIDOREDUCTASE ENZYMES. | |
| 24. |
Some enzymes are named after the reaction , where they are used. What is given to the class of enzymes whcih catalyse the oxidation of one substrate with simultaneous reduction of another substrate. |
| Answer» SOLUTION :Enzymes WHCIH catalyse the OXIDATION ofone substance with simultaneous reduction of another substance are called OXIDOREDUCTASE. | |
| 25. |
Some entropy change are represented in fig. Select correct entropy change: |
|
Answer» `DeltaS_(1),DeltaS_(2),DeltaS_(3)` |
|
| 26. |
Some elements in p-block shows allotropy. What are the allotropic forms of sulphur? |
| Answer» SOLUTION :Yellow RHOMBIC sulphur `(ALPHA- sulphur)`, monoclinic sulphur (p - sulphur). | |
| 27. |
Some elements exist in more than one crystalline or molecular forms in the same physical state is called………………. |
|
Answer» isomerism |
|
| 28. |
Some elements in p-block shows allotropy. What are inter halogen compounds? |
| Answer» SOLUTION :COMPOUNDS FORMED by the combination of two DIFFERENT halogens are called inter halogen compounds. E.g.,CLF,`BrF_3`. | |
| 29. |
Some elements in p-block shows allotropy. Name two oxoacids of Sulphur. |
| Answer» SOLUTION :SULPHUROUS ACID `(H_2SO_3)` SULPHURIC acid `(H_2SO_4)` | |
| 30. |
Some drugs do not bind to the active site of enzyme but bind to a different site. This site is called |
|
Answer» SPECIFIC SITE |
|
| 31. |
Some drugs do not bind to active site of enzyme but bind to a different site. This site is called |
|
Answer» SUBSTITUTION SITE |
|
| 32. |
Some effects of sulphur dioxide and its transformation products on plants include |
|
Answer» Chlorophyll destruction |
|
| 33. |
Some current should be passed for 2 hours to liberated 0.504 gm H_(2) gas. For same time if same current is passed then how much gram of oxygen is liberated ? |
|
Answer» 2.0 gm To liberated 2 gm `H_(2)` required CURRENT=`2xx96500C` So, to liberate `0.504gm` `H_(2)` gas required current=(?) `=(2xx96500)/(2)xx0.504F=48636C` `4OH^(-) to 2H_(2)O+O_(2)+4e^(-)` On passing `4xx96500` current 1 mole `O_(2)=32GM` So, on passing 48636 C current=? `O_(2)` `=(32xx48646)/(4xx96500)=4.032gm~~4.0gm` |
|
| 34. |
Some drug do not bind to the enzyme's active site , instead bind to a different site of enzyme . This site is called. |
|
Answer» ALLOSTERIC SITE |
|
| 35. |
Some crystal defects are shown in the figures below : Indicate the types of defect shown by (a) and (b). |
|
Answer» Solution :(a) indicates VACANCY DEFECT. (B) indicates INTERSTITIAL defect. |
|
| 36. |
Some common salt isadded to ice taken in a flask kept at 0^(@)C. With time |
|
Answer» there is no change in the mass of the ice |
|
| 37. |
Some amount of NH_4 CI was boiled with 50ml of 0.75 N NaOH solution till the reaction was complete. After the completion of the reaction 10 ml of 0.75 N H_2 SO_4 was required for the neutralization of the remaining NaOH. The amount of NH_4 CI taken was: |
|
Answer» 1,6 gm |
|
| 38. |
Some amount of "20V" H_(2)O_(2) is mixed with excess of acidified solution of Kl. The iodine so liberated required 200 mL of 0.1 N Na_(2)S_(2)O_(3) for titration. The volume of O_(2) at STP that would be liberatedby above H_(2)O_(2) solution on disproportionation is : |
|
Answer» 56 mL |
|
| 39. |
Some amount of "20V" H_(2)O_(2) is mixed with excess of acidified solution of Kl. The iodine so liberated required 200 mL of 0.1 N Na_(2)S_(2)O_(3) for titration. The volumeof H_(2)O_(2) solution is : |
|
Answer» 11.2 mL |
|
| 40. |
Some amount of "20V" H_(2)O_(2) is mixed with excess of acidified solution of Kl. The iodine so liberated required 200 mL of 0.1 N Na_(2)S_(2)O_(3) for titration. The mass of K_(2)Cr_(2)O_(7) needed to oxidise the above volume of H_(2)O_(2) solution is : |
|
Answer» 3.6 g |
|
| 41. |
Some alkylhalides undergo substitution whereas some undergo elimination reaction on treatment with bases discuss the structural features of alkyl halides with the help of examples which are responsible for this difference. |
| Answer» Solution :Please note that primary alkyl halides generally prefer to take part in the substitution reaction by following `S_(N^(2))` mechanism while TERTIARY alkyl halides prefer to participate in elimination reactions by following `E_(1)` mechanism. However, other factors are also SIGNIFICANT. | |
| 42. |
Some alkyl halides undergo substitution whereas some undergo elimination reaction on treatment with bases. Discuss the structural features of alkyl halides with the help of examples which are responsible for this difference. |
Answer» Solution :Primary alkyl halides follow `S_(N)2` mechanism in which a NUCLEOPHILE attacks at `180^(@)` to the halogen atom. A transition state is formed in which carbon is bonded to two NUCLEOPHILES and finally halogen atom is pushed out. In `S_(N)2` mechanism, substitution of nucleophile takes place as FOLLOWS  THUS, in `S_(N)2` mechanism, substitution takes place. Tertiary alkyl halides follow `S_(N)1` mechanism. In this case, tert alkyl halides form `3^(@)` carbocations. Now, if the reagent used is a weak base then substitution occur while if it is a strong base than instead of substitution elimination occur.  here the reagent used is aq. KOH. It is a weak base so, substitution takes place.  As alc. KOH is a strong base, so elimination competes over substitution and alkene is formed |
|
| 43. |
Some alkyl halides undergo substitutionn whereas some undergo elimination reaction on treatment with bases. Discuss the structural features of alkyl halides with the help of examples which are responsible for this difference. |
| Answer» Solution :Primary and secondary alkyl halides PREFER to undergo substitution reactions by `S_(N)2` mechanism while tert-alkyl halides prefer to undergo elimination reactions due to the formation of stable carbocations. For example 'n-butyl' bromide and sec-butyl bromide on treatment with aqueous KOH undergo substitution reactions to afford the corresponding alcohols, i.e., n-butyl alcohol and sec-butyl alcohol respectively. in contrast tert-butyl bromide on treatment with aqueous KOH preferentially undergoes elimination to form isobutylene as the MAJOR product. WRITE EQUATIONS yourself. | |
| 44. |
Some alkyl halides undergo substitution reaction whereas some undergo elimination reaction on treatment with bases. Discuss the structural features of alkyl halides with the help of examples which are responsible for this difference. |
|
Answer» Solution :A chemical reaction is the result of COMPETITION. An alkyl HALIDE, with hydrogen atom on carbons neighbouring to halogen carrying carbon, has two COMPETING routes: substitution and elimination. Which route will be taken depends upon the nature of alkyl halide, strength and size of base/nucleophile and reaction conditions. Thus a bulkier nucleophile like `C_(4)H_(9)O^(-)`will prefer to act as a base. It will abstract a proton rather than approach a tetravalent carbon atom for steric reasons. On the contrary, a smaller nucleophile will attach itself to the carbon carrying the halogen. A primary alkyl halide will prefer a `S_(N)2`reaction, a secondary halide will tend to give `S_(N)2`or elimination reaction depending upon the strength of the base/nucleophile and a tertiary alkyl halide will give `S_(N)1`or elimination reaction depending upon the stability of the carbocation or the more substituted alkene. |
|
| 45. |
Solvent used in dry cleaning of clothes is: |
|
Answer» Alcohol |
|
| 46. |
Solvay's process is used for the preparation of |
|
Answer» Ammonia |
|
| 48. |
Solutions with components which obey Raoult's law over the entire composition range are called |
|
Answer» REAL solutions |
|
| 49. |
Solutions which distil without change in composition or temperature are called |
|
Answer» IDEAL solutions |
|
| 50. |
Solutions of two electrolytes 'A' to 'B' are dilutesd. The wedge_(m) of 'B' increases 1.5 times while that of A increases 25 times. Which of the two is a strong electrolyte? Justify your answer. |
| Answer» Solution :B' is a STRONG ELECTROLYTE because of dilution of a strong electrolyte, number of ions remains the same. Only interionic attraction decreases and therefore INCREASE in `wedge_(m)` is SMALL. | |