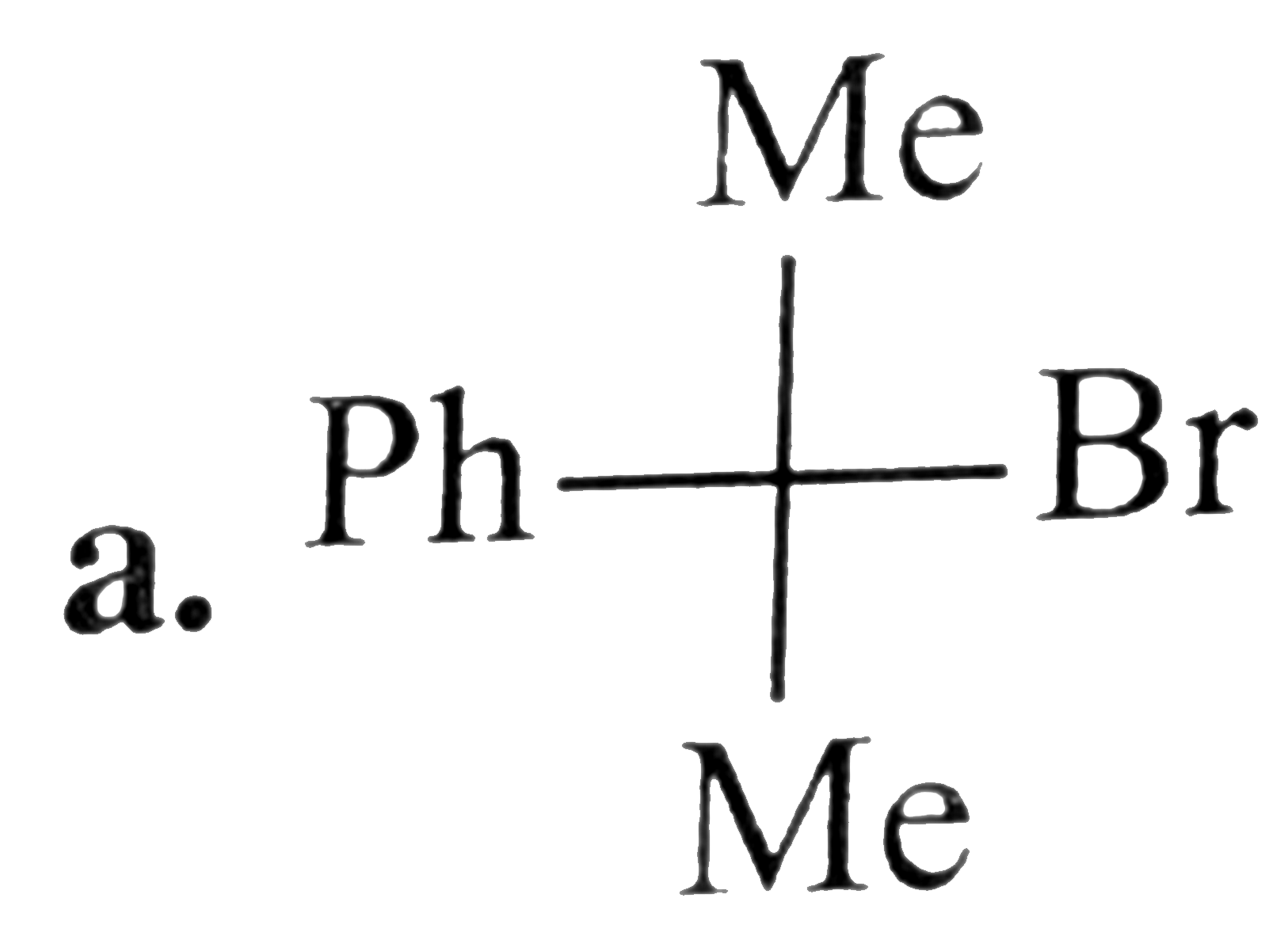Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 2. |
SO_2 can be obtained from which of the following reaction? |
|
Answer» Reaction with DIL. `H_2SO_4` with `O_2` |
|
| 3. |
SO_2 can be used as: |
|
Answer» BLEACHING agent |
|
| 6. |
SO_2 can act as strong oxidising agent in: |
|
Answer» ACIDIC medium |
|
| 7. |
SO_(2) and CO_(2) both lime water (A) milky ,SO_(2) alsoturns K_(2)Cr_(2)O_(7)//H^(o+)(B) green O_(2) is solutionin pyrogallal (C ) turmingit black .Thesegasesareto the detected in order by usingthesereagents .The order is |
|
Answer» (A),(B) ,(C ) |
|
| 8. |
SO_2 and CH_4 are introduced in a vessel in the molar ratio 1:2.The ratio of molecules of two gases present in the container when their rate of effusion becomes equal is : |
|
Answer» `1:2` `p_1/sqrtM_1=p_2/sqrtM_2` `p_1/p_2=sqrtM_1/sqrtM_2=n_1/n_2 IMPLIES n_(SO_2)/n_(CH_4)=SQRT(M_(SO_2)/M_(CH_4))=sqrt(64/16)=2` |
|
| 9. |
SO_2acts as a bleaching agent. How? |
|
Answer» Solution :In the PRESENCE of moisture, `SO_2` gives nascent hydrogen which REDUCES the colouring MATTER to a colourless compound. `SO_2 + 2H_2 O RARR H_2SO_4 + 2H` Vegetable colouring matter ` +H rarr ` Colouless matter |
|
| 10. |
SO_(2) acts as a/an |
|
Answer» bleaching agent |
|
| 11. |
SnO_(2) forms a positively charged colloidal sol in acidie medium and a negatively charged sol in the basic medium. Why ? Explain . |
|
Answer» Solution :`SnO_(2)` is ampotericin nature. It reacts with ACID, e.g, with HCL to form `SnCl_(4)` in the solution. The common `Sn^(4+)` ions are adsorbed on the SURFAC of `SnO_(2)` particles thereby giving them a positive charge `SnO_(2)+4HCl to SnCl_(4)+2H_(2)O` `SnO_(2)+Sn^(4+)to underset("POSITIVELY charged colloidal particles")([SnO_(2)]:Sn^(4+))` `SnO_(2)` reacts with a base, e.g., NaOH to form sodium stannate `(Na_(2)SnO_(3))` in the solution. The stannate ions are adsorbed on the surface of `SnO_(2)` particles giving them an negative charge. `SnO_(2)+2NAOH to underset("Sodium stannate")(Na_(2)SnO_(3)+)H_(2)O` `SnO_(2)+SnO_(3)^(2-)to underset("Negatively charged colloidal particles")([SnO_(2)]:SnO_(3)^(2-))` |
|
| 12. |
SnO_2 forms a positively charged colloidal sol in the acidic medium and negatively charged sol in the basic medium. Explain. |
|
Answer» SOLUTION :`SnO_2` is of AMPHOTERIC in nature which means that it can react with both acids and bases. With an acid such as HCL, if forms `SnCl_4`. The` Sn^(4+)` ions are adsorbed on the surface of `SnO_2` to give a POSITIVELY charged colloidal sol. `SnO_2+4HCl to SnCl_4+2H_2O and SnO_2+Sn^(4+) to [SnO_2]Sn^(4+)` Similarly with base like NaOH, if forms SODIUM stannate `(Na_2SnO_3)`. The stannate ions get adsorbed on the surface of SnO2 to give negatively charged colloidal sol. `SnO_2+2NaOH to Na_2SnO_3 and SnO_2+SnO_3^(2-) to [SnO_2] SnO_3^(2-)` |
|
| 13. |
SnCl_(4)stannic chloride hydrolyses in dilute solution as per given reaction SnCl_(4)+4H_(2)Oto[Sn(OH)_(4)]+4HCl. Hydrolysis can be repressed by adding compound "A".SnCl_(4)+A to B, B can be: |
|
Answer» `Sn(OH)_(4)` |
|
| 14. |
SnCl_(2) + HCl convert |
|
Answer» `-CHO` to `-CH_(2),-OH` |
|
| 15. |
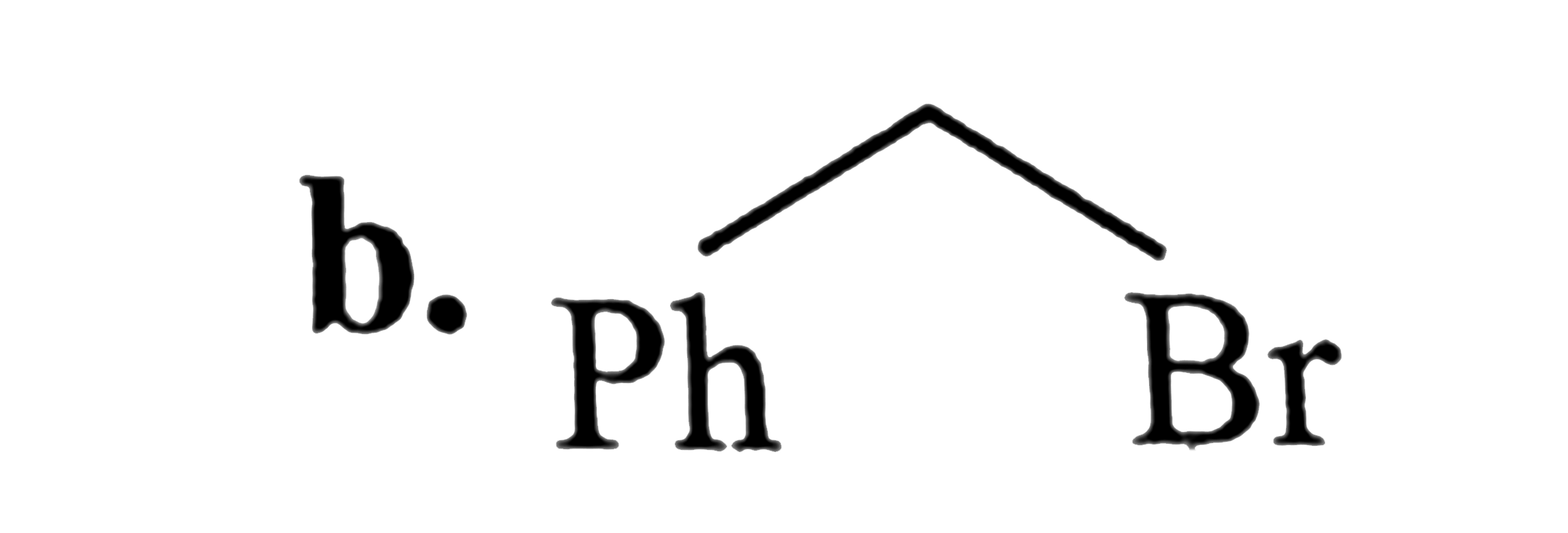
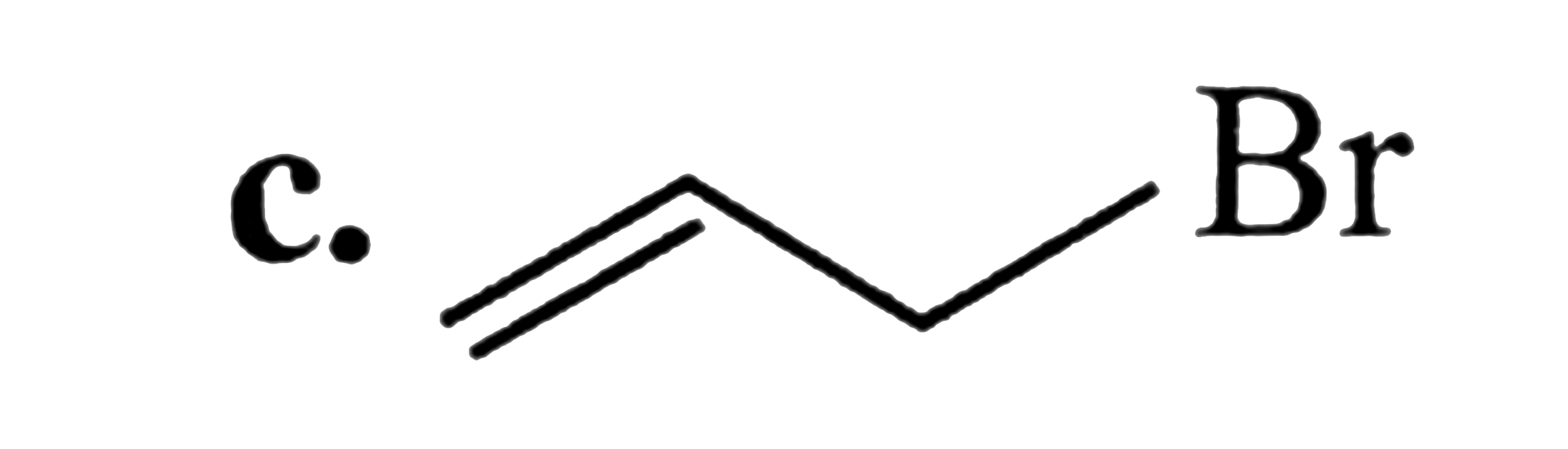
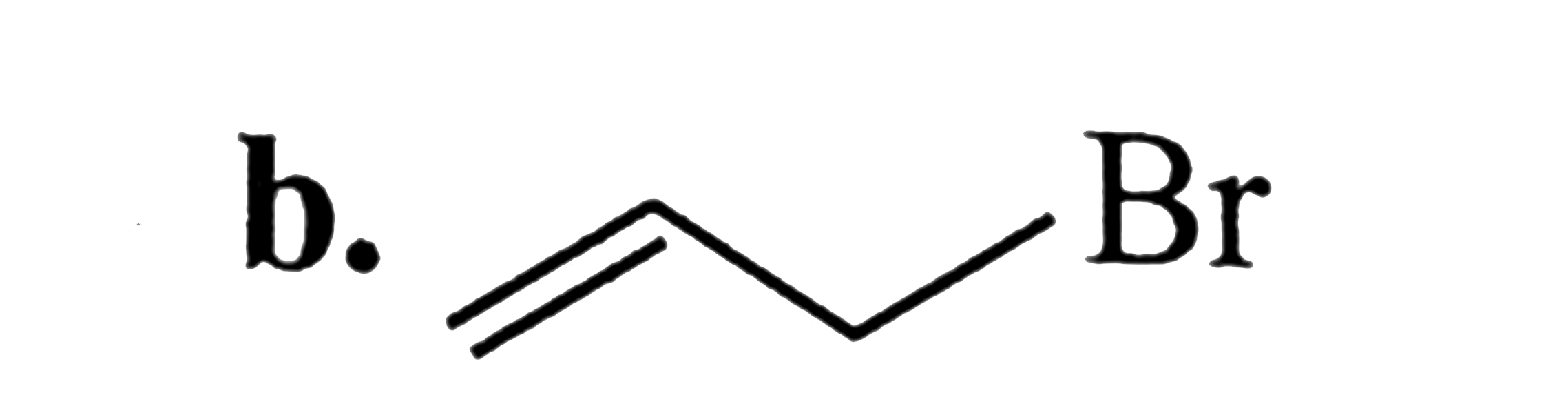
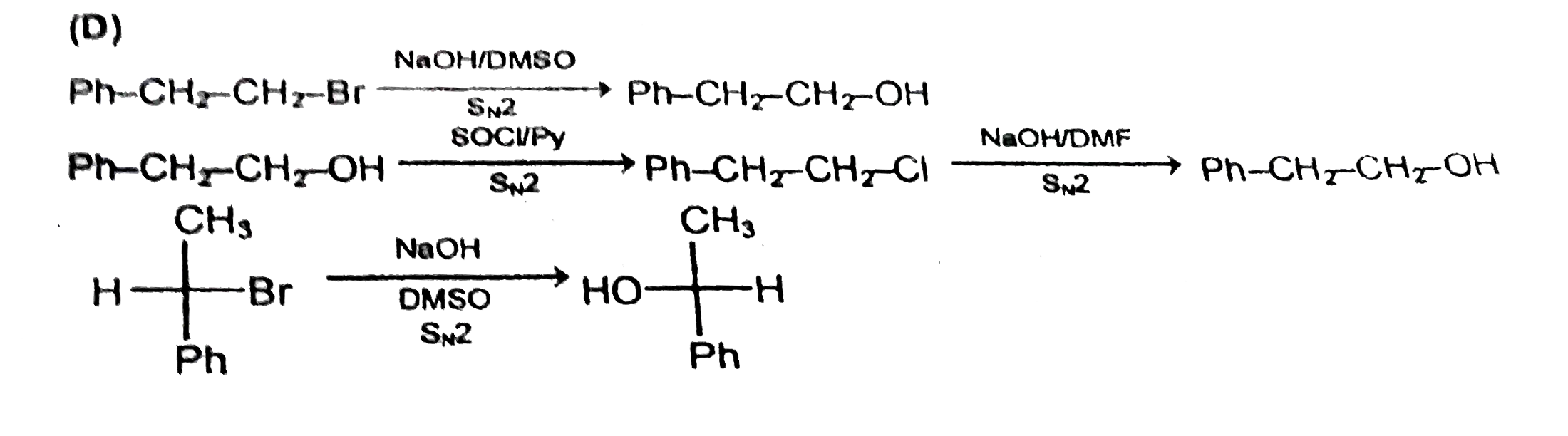
SN^2 reactions of alkyl halides is a bimolecular reaction which take place through formation of a transition state. The rate of reaction depends on the concentration of alkyl halide andnucleophile.The reaction is favoured by strong nucleophlie in polar aprotic solvents.Which of the following undero substitution by SN^2 mechanism at a faster rate from other |
|
Answer» `CH_3CH_2CH_2Br`<BR>`CH_2 = CH - CH_2 - Br` |
|
| 16. |
S_(N)2 reactions are much faster in the solvents like |
|
Answer» dimethyl formamide |
|
| 17. |
SN^2 reactions of alkyl halides is a bimolecular reaction which take place through formation of a transition state. The rate of reaction depends on the concentration of alkyl halide andnucleophile.The reaction is favoured by strong nucleophlie in polar aprotic solvents. In which of the following solvents SN^2reaction is more favourable |
|
Answer» `CH_3COOH` |
|
| 18. |
SN^(2) reaction is a bimolecular reaction which takes places by the formation of T.S. Veolcity of the reaction depends on the concentration of the substrate as well as the nucleophile. The reaction is favoureed by strong Nu^(o-) and in the presence of polar aprotic sovlent, optically active halides give. Walken inversion by SN^(2) mechanism. The presence of hero group (atom) at beta-C atom,unsaturation at beta-C and(-overset(O)overset(||)(C)-) group at alpha-C atomn favor SN^(2) mechanism. Allyl halides and benzyl halides give SN^(1) andSN^(2) reactions. Allyl halides alos give SN^(2) mechanism EDG at ortho- and para-postions in benzyl halides favors SN^(1)mechanism, wheras EWG favors SN^(2) mechanism. Which of the following will give SN^(1) reaction? |
|
Answer»
`(C) implies`benzyl halide with `EDG` |
|
| 19. |
S_N2 reactions are |
|
Answer» Stereospecific but not stereoselective |
|
| 20. |
S_(N^(2)) reaction will be fastest in : |
| Answer» Answer :D | |
| 21. |
SN^(2) reaction is a bimolecular reaction which takes places by the formation of T.S. Veolcity of the reaction depends on the concentration of the substrate as well as the nucleophile. The reaction is favoureed by strong Nu^(o-) and in the presence of polar aprotic sovlent, optically active halides give. Walken inversion by SN^(2) mechanism. The presence of hero group (atom) at beta-C atom,unsaturation at beta-C and(-overset(O)overset(||)(C)-) group at alpha-C atomn favor SN^(2) mechanism. Allyl halides and benzyl halides give SN^(1) andSN^(2) reactions. Allyl halides alos give SN^(2) mechanism EDG at ortho- and para-postions in benzyl halides favors SN^(1)mechanism, wheras EWG favors SN^(2) mechanism. Which of the following will gives SN^(2) mechanism gt |
|
Answer» `ETBR` `(c) implies allyl halide. |
|
| 22. |
SN^(2) reaction is a bimolecular reaction which takes places by the formation of T.S. Veolcity of the reaction depends on the concentration of the substrate as well as the nucleophile. The reaction is favoureed by strong Nu^(o-) and in the presence of polar aprotic sovlent, optically active halides give. Walken inversion by SN^(2) mechanism. The presence of hero group (atom) at beta-C atom,unsaturation at beta-C and(-overset(O)overset(||)(C)-) group at alpha-C atomn favor SN^(2) mechanism. Allyl halides and benzyl halides give SN^(1) andSN^(2) reactions. Allyl halides alos give SN^(2) mechanism EDG at ortho- and para-postions in benzyl halides favors SN^(1)mechanism, wheras EWG favors SN^(2) mechanism. Which of the following will give Walden inversion ? |
|
Answer» `C_(4)H_(9) - CHD - Br`, |
|
| 23. |
SN^(2) reaction is a bimolecular reaction which takes places by the formation of T.S. Veolcity of the reaction depends on the concentration of the substrate as well as the nucleophile. The reaction is favoureed by strong Nu^(o-) and in the presence of polar aprotic sovlent, optically active halides give. Walken inversion by SN^(2) mechanism. The presence of hero group (atom) at beta-C atom,unsaturation at beta-C and(-overset(O)overset(||)(C)-) group at alpha-C atomn favor SN^(2) mechanism. Allyl halides and benzyl halides give SN^(1) andSN^(2) reactions. Allyl halides alos give SN^(2) mechanism EDG at ortho- and para-postions in benzyl halides favors SN^(1)mechanism, wheras EWG favors SN^(2) mechanism. Which of the following will give SN^(2) mechanism? |
|
Answer» `MEBR` |
|
| 24. |
SN^(2) mechanism proceeds through the formation of |
|
Answer» CARBONIUM ion |
|
| 25. |
S_(N^(2)) reaction are |
|
Answer» stereoselective as WELL as atereopecific |
|
| 26. |
S_(N^(2)) mechanism proceeds through the formation of |
|
Answer» CARBONIUM ION |
|
| 28. |
S_N1 + S_N2 mixed mechanism is observed in the reaction |
|
Answer» <P>(I)(iii)(P) 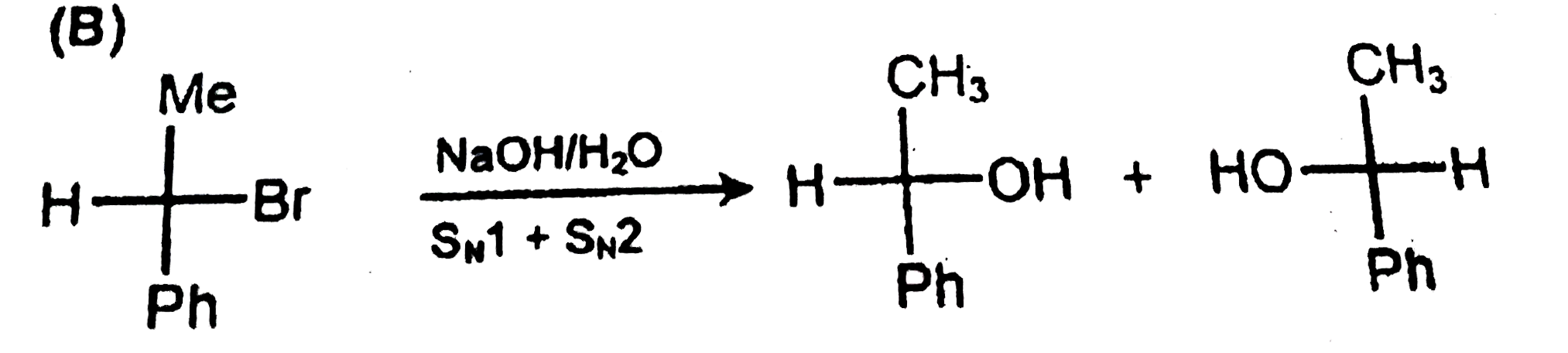
|
|
| 29. |
S_N1 reactions occur through the intermediate formation of |
|
Answer» CARBOCATIONS |
|
| 30. |
SN^1reactions never occur on |
|
Answer» `SP^3C - X` |
|
| 31. |
S_(N)1 reactions are accompanied by racemization in optically active halides. |
| Answer» SOLUTION :In `S_(N)1` reactions, carbocations are the INTERMEDIATES. Since carbocations are planar species, therefore, attack of the nucleophile on them cann occur from both the sides (front and rear) with ALMOST equal ease GIVING a 50:50 mixture of the TWO enantiomers, i.e., a racemix mixture is obtained. | |
| 32. |
S_(N)1 reaction undergoes in which type of fission ? |
|
Answer» Homolytic fission 
|
|
| 33. |
S_(N)1 reaction of alkyl halides leads to : |
|
Answer» retentionof CONFIGURATION |
|
| 34. |
S_N^1 reaction on optically active substrates mainly gives: |
|
Answer» RETENTION in configuration |
|
| 35. |
SN^1 reaction is given by alkylhalide which forms stable carbocation during reaction. The carbonation, has sp^2 hybridisation. In SN^1reaction, the attack of the nucleophile to carbocation inter mediate is from either side. In SN^2reaction the attacking nucleophile attacks from the back leading to the formation of inversion complex. Alcohol reacts with PCI_5to give alkyl chloride byan internal attack of nucleophile with in the molecule. In the following reaction . If R is H - undersetoverset(|)(D)oversetunderset(|)(CH_3)(C ) - R - OH underset(Py)overset(SOCl_2)(to) R - Cl + SO_2 + HCl |
|
Answer» There is no CHANGE in CONFIGURATION 
|
|
| 36. |
SN^(1) reaction is. |
|
Answer» SINGLE STEP REATION |
|
| 37. |
S_(N)1 mechanism of alkyl halides is favoured by : |
|
Answer» HIGHER concentration of nucleophile |
|
| 38. |
SN reaction is givenby these compounds, whichhave a nuclophilic group and a good leaving EWG. It should be stable after leaving with bonding pair of overline(e)'s and it should have high polarisability. Nucliphilic aliphatic substituion reaction is mainly of two types SN^(-1) and SN^(2). SN^(-1) mechanism is a two step process. Reaction velocity of SN^(-1) depends only on the concentration of the subtrate. It proceeds via the formation of carbocation, optically active substrate gives (o+) and (o-) froms of the product. In most of the cases, the product usually consists of 5-20% inverted and (95-80%) racemised species. The more stable is the carbocation, the greater is the propotion of racemission. In solvolysis reaction, the more nucleophillic is the solvent, the greater is the proportion of inversion. Which of the following gives SN^(1) reaction? |
|
Answer»
|
|
| 39. |
SN reaction is givenby these compounds, whichhave a nuclophilic group and a good leaving EWG. It should be stable after leaving with bonding pair of overline(e)'s and it should have high polarisability. Nucliphilic aliphatic substituion reaction is mainly of two types SN^(-1) and SN^(2). SN^(-1) mechanism is a two step process. Reaction velocity of SN^(-1) depends only on the concentration of the subtrate. It proceeds via the formation of carbocation, optically active substrate gives (o+) and (o-) froms of the product. In most of the cases, the product usually consists of 5-20% inverted and (95-80%) racemised species. The more stable is the carbocation, the greater is the propotion of racemission. In solvolysis reaction, the more nucleophillic is the solvent, the greater is the proportion of inversion. Which of the following will give SN reaction? |
| Answer» Solution :All | |
| 40. |
SN reaction is givenby these compounds, whichhave a nuclophilic group and a good leaving EWG. It should be stable after leaving with bonding pair of overline(e)'s and it should have high polarisability. Nucliphilic aliphatic substituion reaction is mainly of two types SN^(-1) and SN^(2). SN^(-1) mechanism is a two step process. Reaction velocity of SN^(-1) depends only on the concentration of the subtrate. It proceeds via the formation of carbocation, optically active substrate gives (o+) and (o-) froms of the product. In most of the cases, the product usually consists of 5-20% inverted and (95-80%) racemised species. The more stable is the carbocation, the greater is the propotion of racemission. In solvolysis reaction, the more nucleophillic is the solvent, the greater is the proportion of inversion. Fugacity power of which group will be maximum? |
|
Answer» `CH_(3) - O - underset(O)underset(||)overset(O)overset(||)(S)-C_(2)H_(5)` Therfore, the order of leaving group: `(c) gt (B) gt (a) gt (d)`. |
|
| 41. |
Sn is dissolved in excess of NaOH solution, the compound obtained is |
|
Answer» `SN(OH)_(2)` |
|
| 42. |
S_N1 reaction is favoured by : |
|
Answer» Non POLAR solvents |
|
| 43. |
SN1 reaction is fastest in: |
|
Answer» ETHYL chloride |
|
| 44. |
Smoke is passed through charged surface before allowing it to come out of chimeys in factories. Assign reason. |
| Answer» | |
| 45. |
Smoke has generally blue tinge. It is due to: |
|
Answer» SCATTERING |
|
| 46. |
Smoke is a dispersion of: |
|
Answer» A solid in gas |
|
| 47. |
Smoke, cloud and gold sol are respectively |
|
Answer» AEROSOL, Hydosol and Aquasol |
|
| 48. |
Smog is : |
|
Answer» NOTHING but BLACK smoke |
|
| 49. |
Smetling is done in |
|
Answer» BLAST furnace |
|