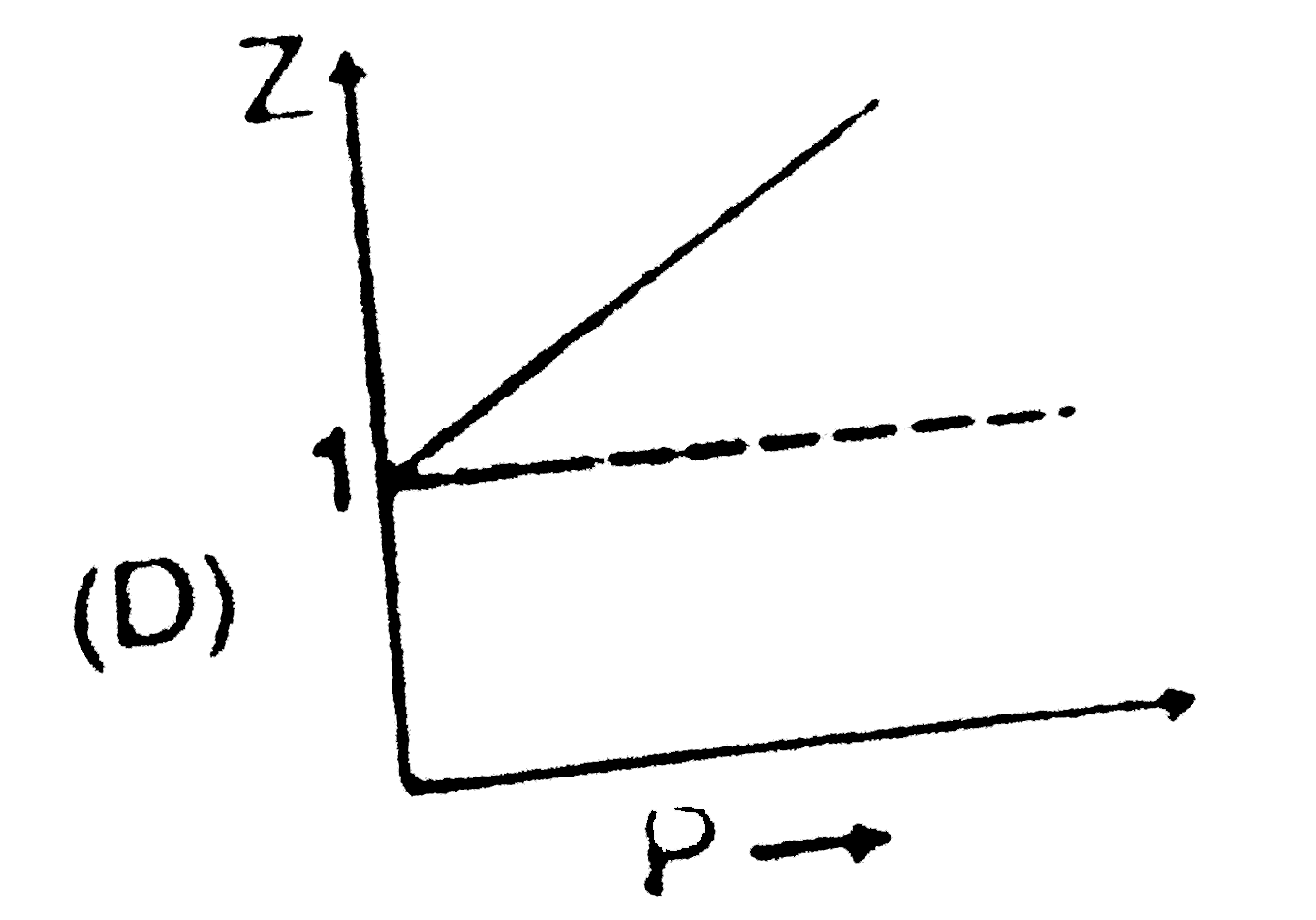Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Smelting is usually carried out in |
|
Answer» BLASTFURNACE |
|
| 2. |
Smelting is termed to the proces in which |
|
Answer» The ORE is heated in the ABSENCE of AIR |
|
| 3. |
Smelting is the process of reduction of metal oxide by : |
|
Answer» C |
|
| 4. |
Smelting is the reduction of oxide to metal by : |
|
Answer» C |
|
| 5. |
Smelting is done in: |
|
Answer» BLAST furnace |
|
| 6. |
Smeltinginvolvesreductionof metal oxide with |
|
Answer» CARBON |
|
| 7. |
Smelting. |
| Answer» SOLUTION :The process of EXTRACTION of a METAL from its ore by HEATING and melting at high temperature is called smelting. | |
| 8. |
Smelting involves reduction of metal oxide with |
|
Answer» ALUMINIUM |
|
| 10. |
Smallest among these spicies is |
|
Answer» lithium |
|
| 11. |
Smell of H_2O_2 resembles: |
|
Answer» Alcohol |
|
| 12. |
Small spherical ball of silver metal used in jewellery having diameter 0.1 cm, which is obtained by the electrolytic deposition.It total number of balls in jewellery is 10,000, then calculate the applied amount of electricity in coulombs, which is used on the deposition on electrodes having entire surface 0.12 m^2.[Density of Ag = 10.5] It is assumed that 3.5% electricity consumed as wastage during electrolysis and 60% of electrode body immersed in electrolyte.[Give your answer in multiple of 10^4] |
|
Answer» Surface area of electrodes =`0.12 m^2=1200 cm^2` Weight of jewellery =`4/3 pir^3xx10,000xx"DENSITY"=4/3xx3.14xx(0.05)^3xx10000xx10.5=54.9` GRAM Now amount of electricity in faraday =`("wt. of Ag")/("EQUIVALENT wt. of Ag")=54.9/108=0.50 F = 48250` coulomb So applied electricity amount =`(48250xx100)/96.5` =50,000 coulomb =`5xx10^4` coulomb |
|
| 13. |
Small quantities of solutions of compounds TX, TY and TZ are put into separete test tubes containing X,Y and Z solution. TX does not react with any of these. TY reacts with both X and Z. Does not react with any of these. TY reats with both X and Z. TZ reacts with X. the decreasing order of state of oxidation of the anions X^(-),Y^(-),Z^(-) |
|
Answer» `Y^(-),Z^(-),X^(-)` `Txto`No reaction `TY to X,Z` `TZtoX` `implies`Order of electrode potential is `TY lt TZ lt TX` `implies`Order of oxidation of the anion is `Y^(-) GT Z^(-) gt X^(-)`. |
|
| 14. |
Small quantities of solutions of compounds TX, TY and TZ are put into separate test tubes containing X, Y andZ solutions. TX does not react with any of these. TY reacts with both X and Z. TZ reacts with X. the decreasing order of oxidation of the anions X^(-),Y^(-),Z^(-) is: |
|
Answer» `Y^(-),Z^(-),X^(-)` `(Y^(-)+XtoY+X^(-),Y^(-)+ZtoY+Z^(-))` TZ reacts with X only `(Z^(-)+XtoZ+X^(-))` `Y^(-)`, thus, DECREASING order of OXIDATION of anions `X^(-),Y^(-),Z^(-)` will be : `Y^(-),Z^(-),X^(-)`. |
|
| 15. |
Sm^(3+) acts as |
|
Answer» an oxidising agent |
|
| 16. |
Slow oxidation of chloroform in air leads to the formation of : |
|
Answer» FORMYL CHLORIDE |
|
| 17. |
Slow oxidation of chloroform in air leads to form: |
|
Answer» formly chloride |
|
| 18. |
Slow decomposition of complex organic compounds into simpler ones by enzymes is known as |
|
Answer» CONDENSATION |
|
| 19. |
Slope of the straight line obtained by plotting log_(10)k against 1/T represents what term? |
|
Answer» `-E_(a)` Where m `to` SLOPE |
|
| 20. |
Slow acting nitrogenous fertilizer among the following is |
|
Answer» `NH_(2)CONH_(2)` `CaNCN +2H_(2)Oto CO_(2) +2NH_(3)` `NH_3 overset("Nitrifying")underset("bacteria")to ` soluble nitrates `to ` plants |
|
| 21. |
Sleeping pills are recommended to patients suffering from sleeplessness but it is not advisable to take them without consulting the doctor. |
| Answer» Solution :All types of sleeping pills WHETHER they contain chlordiazepoxide, MEPROBAMATE, equanil, valium or barbiturates have side effects. Sleeping pills make you to breathe more slowly and less deeply. That can be dangerous for people who have asthma or some other heart problems. Sleeping pills also cause burning or tingling in the hands, feet or LEGS, changes in appetite, constipation, diarrhoea, dizziness and drowsiness. Further, dosage of a particular sleeping pill is also important since WOMEN clear the drug from their systems more slowly than men. In view of the above side effects of the sleeping pills, it is ADVISABLE to consult the doctor before taking a particular sleeping pill. | |
| 22. |
Slope of the plot between V and P at constant temperature |
|
Answer» zero |
|
| 23. |
Sleeping pills are recommended to patients suffering from sleeplessless but is not advisable to take them without consulting the doctor. |
| Answer» Solution :All types of sleeping pills whether they contain chlordiazepoxide, meprobamate, EQUANIL, valium or barbiturates have side effects. Sleeping pills make you to breathe more SLOWLY and less deeply. That can be dangerous for people who have asthma or some other HEART problems. Sleeping pills also cause burning or tingling in the hands, feet or legs, changes in appetite, constipation, DIARRHOEA, dizziness and drowsiness. Further, dosage of a particular sleeping pill is also important since women clear the DRUG from their systems more slowly than men. In view of the above side effects of the sleeping pills, it is advisable to consult the doctor before taking a particular sleeping pill. | |
| 24. |
Slaking is the process of adding water to : |
|
Answer» `Ca(OH)_(2)` |
|
| 25. |
Sleeping pills are recommended by doctors to the patients suffering from sleeplessness but it is not advisable to take its doses without consultation with the doctor. Why ? |
| Answer» SOLUTION :Most of drugs taken in doses higher than that recommended by the doctor may produce harmful EFFECTS and, act as poison and can cause even death. Therefore, a doctor MUST always be CONSULTED before taking the drug. | |
| 26. |
Sleeping pills are recommended by doctors to the patients suffering from sleeplessness but it is not advisable to take its doses without consultation with the doctor. Why? |
| Answer» Solution :Most of the DRUGS TAKEN in doses HIGHER than recommended may cause harmful effect and act as poison. Therefore, a doctor should ALWAYS be consulted before taking MEDICINE. | |
| 27. |
Slaked lime is used in the manufacture of: |
|
Answer» Cement |
|
| 30. |
Slag is a product of |
|
Answer» Fluxand COKE |
|
| 31. |
Slag is formed by reaction between |
|
Answer» IMPURITIES and coke |
|
| 32. |
Slag coming out at the bottom of a blast furance during extraction of iron from its ores , is used in making : |
| Answer» Answer :A | |
| 33. |
Slag is : |
|
Answer» flux and coke |
|
| 34. |
Slacked lime is used in the manufacture of |
|
Answer» CEMENT |
|
| 35. |
Skin turns yellow in contact with conc. HNO_3, because: |
|
Answer» PROTEINS are CONVERTED into xanthoproteins |
|
| 36. |
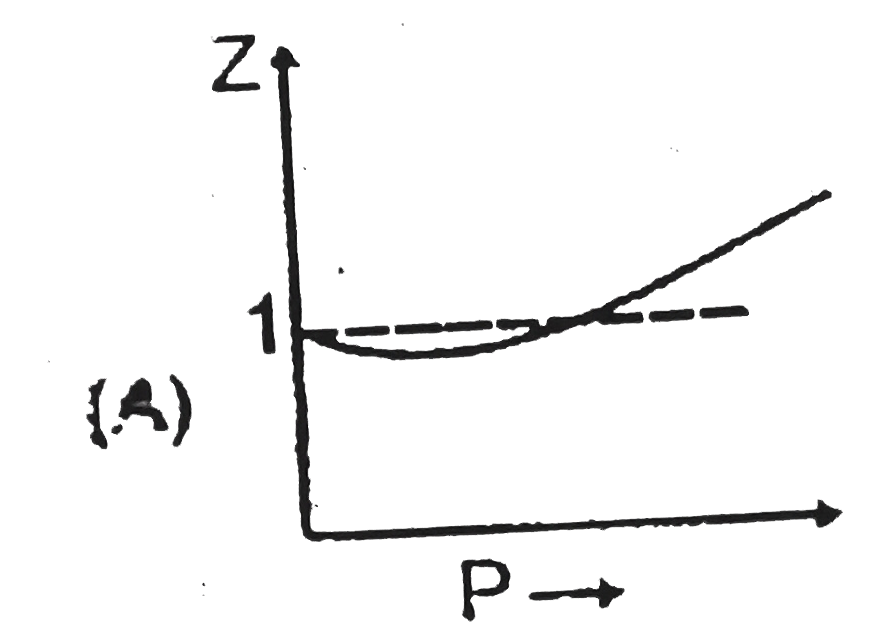
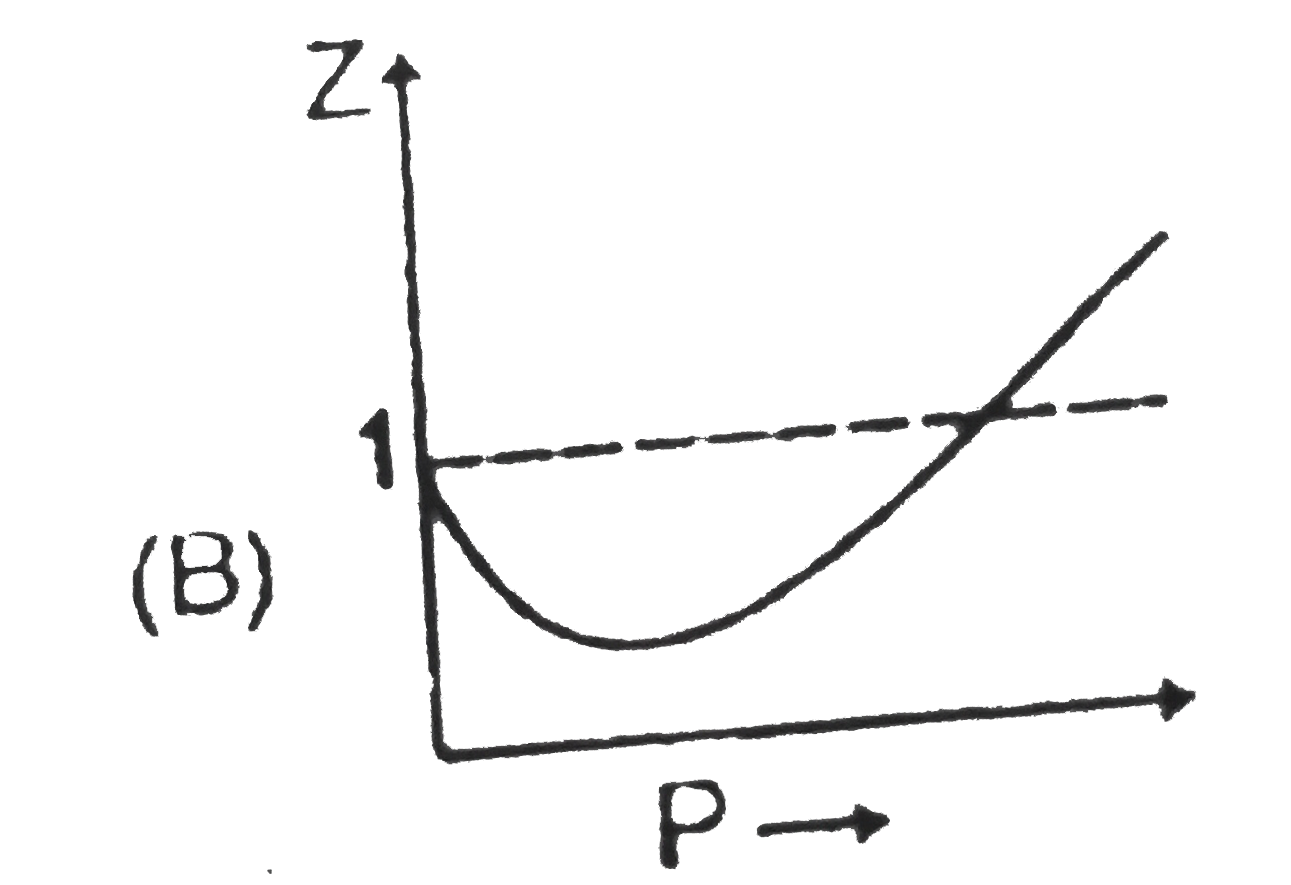
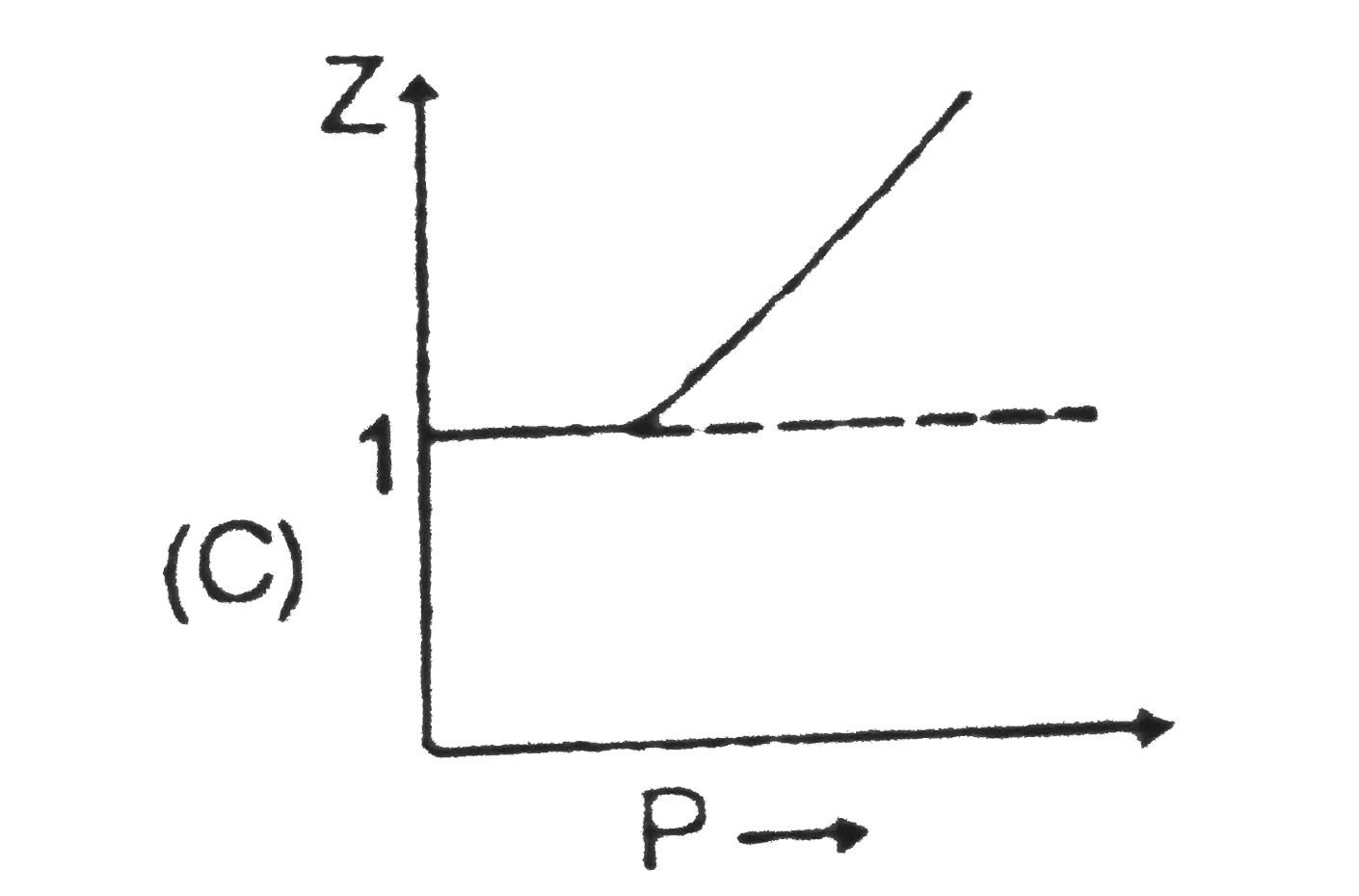
Sketch shows the plot of Z v/s P for a hypothetical gas for one mole at three distint temperature. Boyle's temperature is the temperature at which gas shows ideal behaviour over a pressure range in the low pressure region.Boyle's temperature (T_b)=a/(Rb).If a plot is obtained at temperature well below Boyle's temperature then the curve will show negative deviation, in low pressure region and positive deviation in the high pressure region. Near critical temperature the curve is more likely as CO_2 and the temperature well above critical temperature curve is more like H_2 at 0^@C as shown above.At high pressure suppose all the constant temperature curve varies linearly with pressure according to the following equation : Z=1+(Pb)/(RT) (R=2 cal "mol"^(-1) K^(-1)) As shown in the figure at 200 K and 500 atm value of compressibility factor is 2 (approx).Then volume of the gas at the point will be |
|
Answer» <P>0.01 L |
|
| 37. |
Sketch shows the plot of Z v/s P for a hypothetical gas for one mole at three distint temperature. Boyle's temperature is the temperature at which gas shows ideal behaviour over a pressure range in the low pressure region.Boyle's temperature (T_b)=a/(Rb).If a plot is obtained at temperature well below Boyle's temperature then the curve will show negative deviation, in low pressure region and positive deviation in the high pressure region. Near critical temperature the curve is more likely as CO_2 and the temperature well above critical temperature curve is more like H_2 at 0^@C as shown above.At high pressure suppose all the constant temperature curve varies linearly with pressure according to the following equation : Z=1+(Pb)/(RT) (R=2 cal "mol"^(-1) K^(-1)) Plot at Boyle's temperature for the gas will be |
|
Answer»
|
|
| 38. |
Sketch shows the plot of Z v/s P for a hypothetical gas for one mole at three distint temperature. Boyle's temperature is the temperature at which gas shows ideal behaviour over a pressure range in the low pressure region.Boyle's temperature (T_b)=a/(Rb).If a plot is obtained at temperature well below Boyle's temperature then the curve will show negative deviation, in low pressure region and positive deviation in the high pressure region. Near critical temperature the curve is more likely as CO_2 and the temperature well above critical temperature curve is more like H_2 at 0^@C as shown above.At high pressure suppose all the constant temperature curve varies linearly with pressure according to the following equation : Z=1+(Pb)/(RT) (R=2 cal "mol"^(-1) K^(-1)) In very high pressure region if Z v/s P is plotted at 1200 K for the above gas then it will have greatest slope. |
|
Answer» TRUE |
|
| 39. |
Size ratio is minimum in :- |
|
Answer» `(LI^(-))/(Li)` |
|
| 40. |
Sketch shows the plot of Z v/s P for a hypothetical gas for one mole at three distint temperature. Boyle's temperature is the temperature at which gas shows ideal behaviour over a pressure range in the low pressure region.Boyle's temperature (T_b)=a/(Rb).If a plot is obtained at temperature well below Boyle's temperature then the curve will show negative deviation, in low pressure region and positive deviation in the high pressure region. Near critical temperature the curve is more likely as CO_2 and the temperature well above critical temperature curve is more like H_2 at 0^@C as shown above.At high pressure suppose all the constant temperature curve varies linearly with pressure according to the following equation : Z=1+(Pb)/(RT) (R=2 cal "mol"^(-1) K^(-1)) Which of the following is correct : |
|
Answer» `a/b lt 0.4 k cal mol^(-1)` `implies 20lta/(Rb)lt1000implies 400 cal lt a/b lt 2000 cal` |
|
| 41. |
Sketch shows the plot of Z v/s P for a hypothetical gas for one mole at three distint temperature. Boyle's temperature is the temperature at which gas shows ideal behaviour over a pressure range in the low pressure region.Boyle's temperature (T_b)=a/(Rb).If a plot is obtained at temperature well below Boyle's temperature then the curve will show negative deviation, in low pressure region and positive deviation in the high pressure region. Near critical temperature the curve is more likely as CO_2 and the temperature well above critical temperature curve is more like H_2 at 0^@C as shown above.At high pressure suppose all the constant temperature curve varies linearly with pressure according to the following equation : Z=1+(Pb)/(RT) (R=2 cal "mol"^(-1) K^(-1)) For 500 K plot value of Z changes from 2 to 2.2 if pressure is varied from 1000 atm to 1200 atm (high pressure ) then the value of b/(RT) will be |
|
Answer» `10^(-3) "atm"^(-1)` SOLVE this equation for the two points and get result (VALUE of `b/(RT)`) |
|
| 43. |
Size order of octahedral, tetrahedral & cubic void is - |
|
Answer» CUBIC `GT` tetrahedral VOID `lt` OCTAHEDRAL void |
|
| 44. |
Size of colloidal particles may range from: |
|
Answer» `1` to `1000 nm` |
|
| 45. |
Sixteen aldohexosea are possible. How many of these do not undergo mutarotation or osazone formation ? |
|
Answer» |
|
| 46. |
Size of ionic species follows the order: |
|
Answer» `I^(-) LT I^+ lt I` |
|
| 47. |
Six volumes of oxygen, on complete ozonisation, form…..volumes of ozone. |
|
Answer» 2 |
|
| 48. |
Six grams of urea ( molar mass = 60 ) are dissolved in 90 g of water. The relative lowering of vapour pressure is equal to |
|
Answer» 0.0196 `x_(B)=((6)/(60))/(6/60 +90/18)=0.0196` |
|
| 49. |
Siulphide ores of metals are usually concentrated by froth floatation process. Which one of the following sulphide ores offfers an exception and is concentrated by leaching ? |
|
Answer» Argentitie |
|