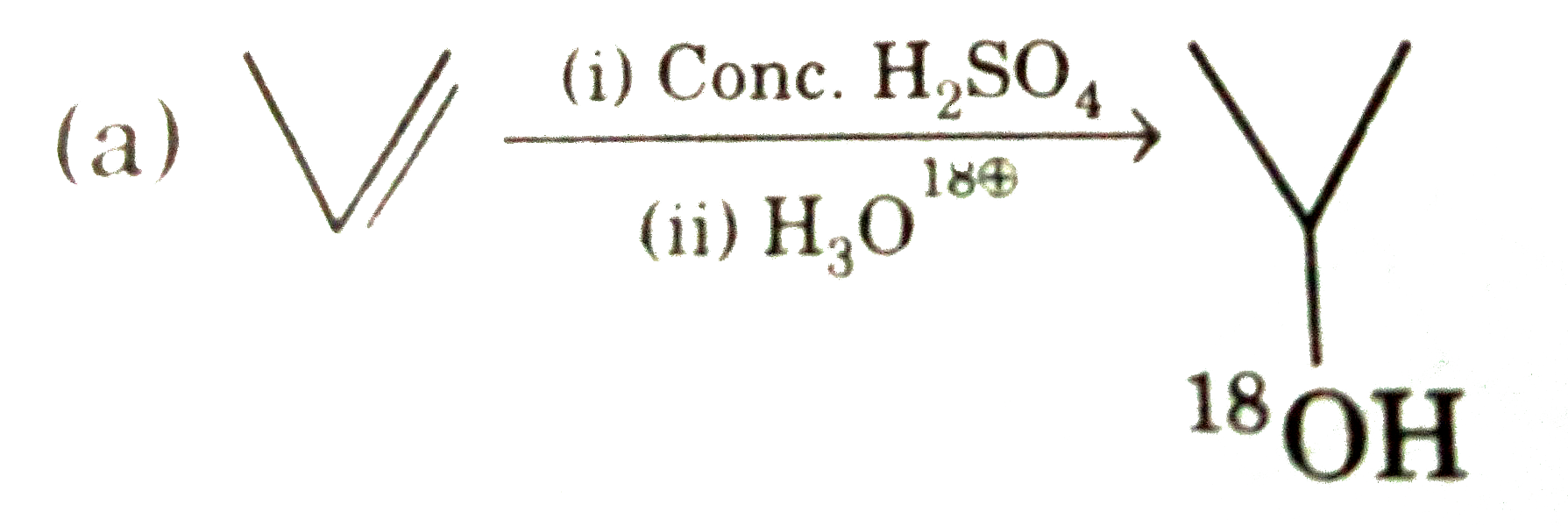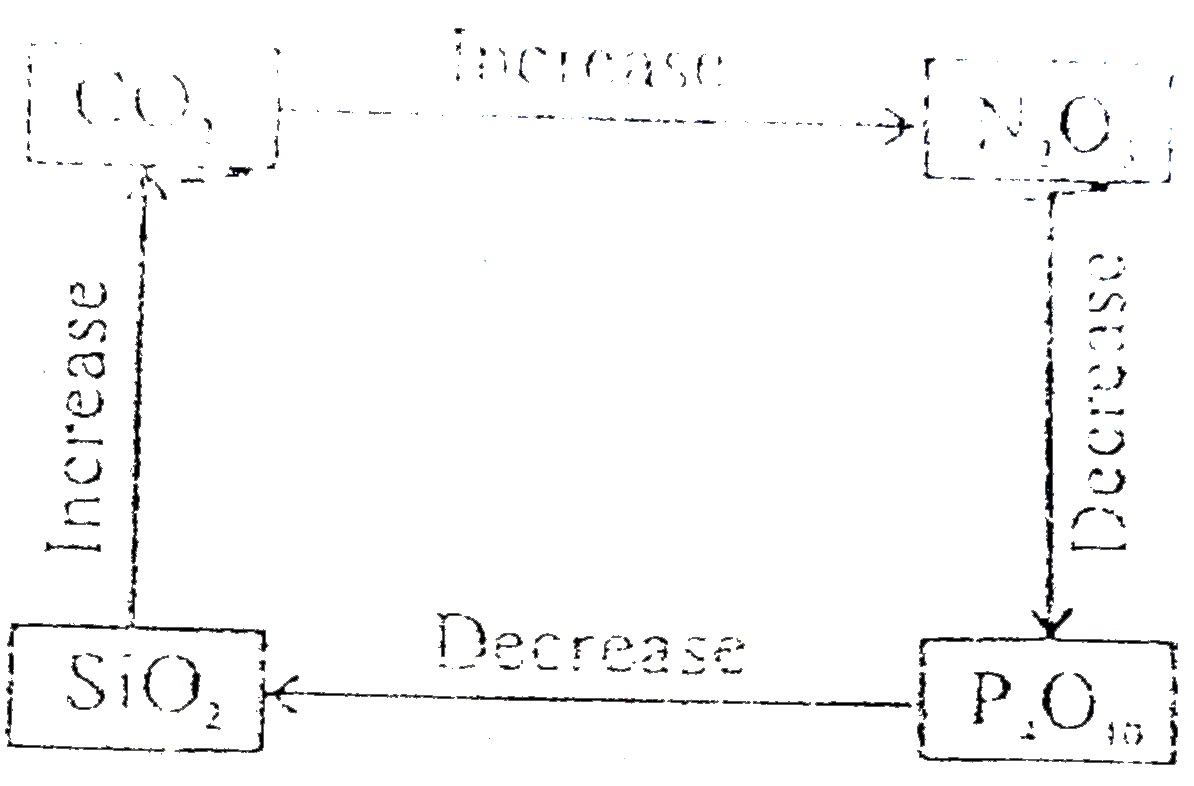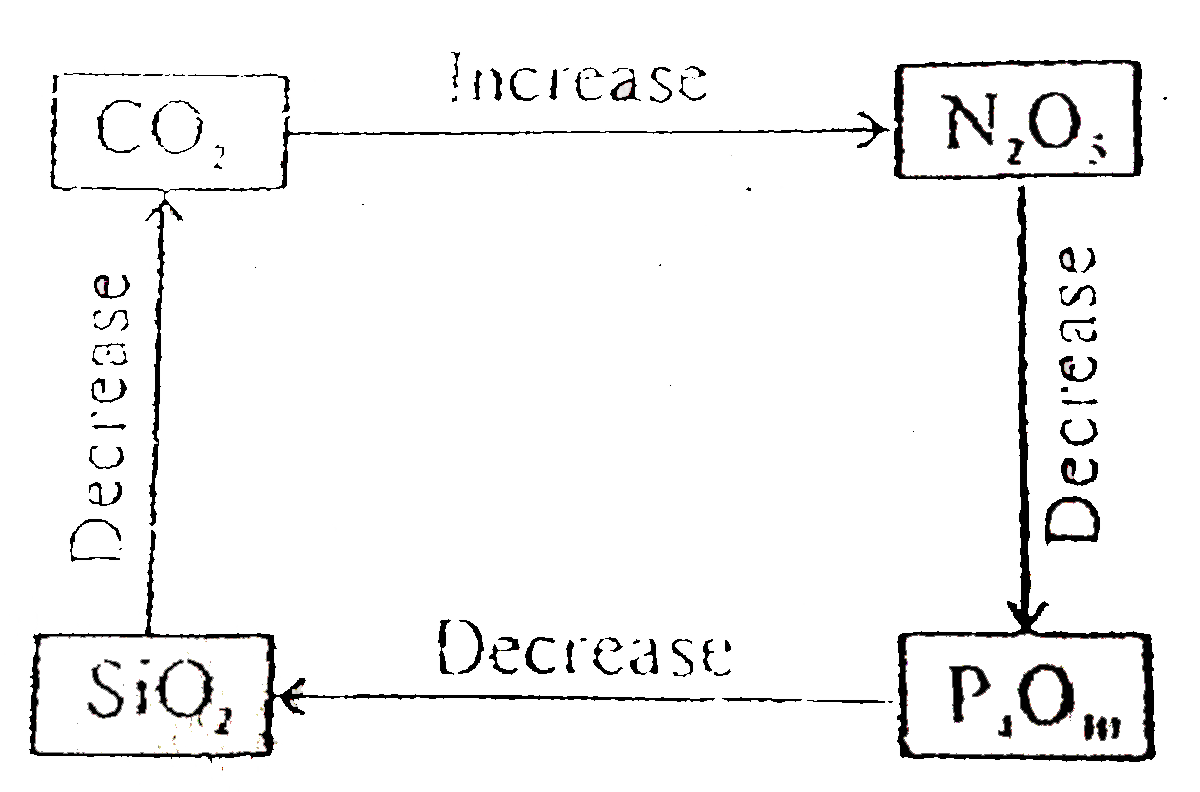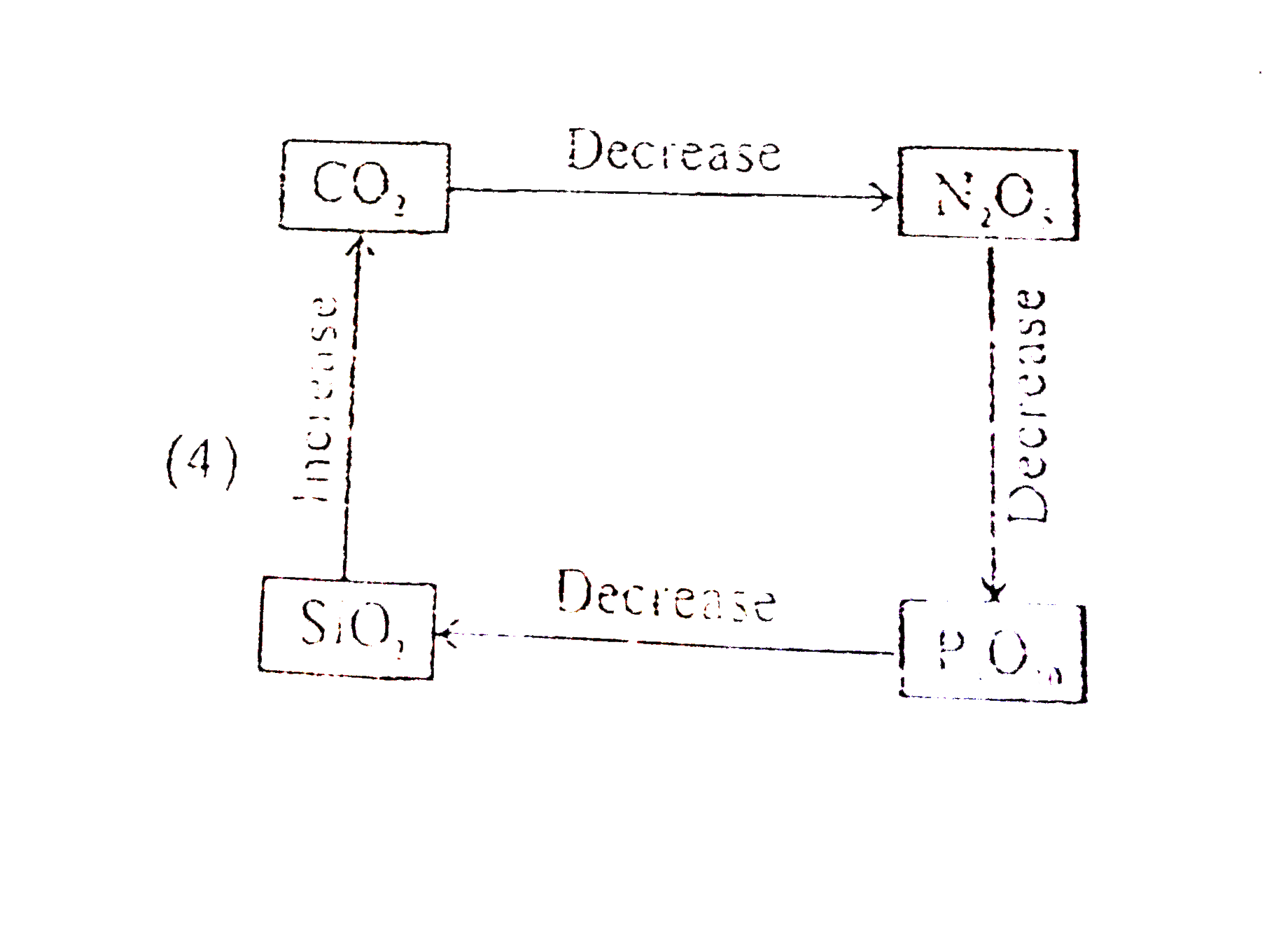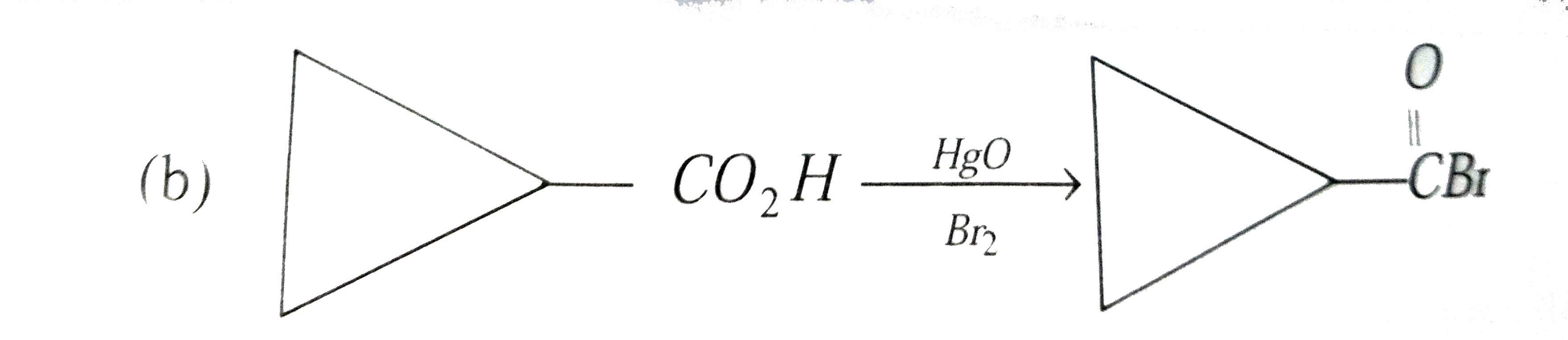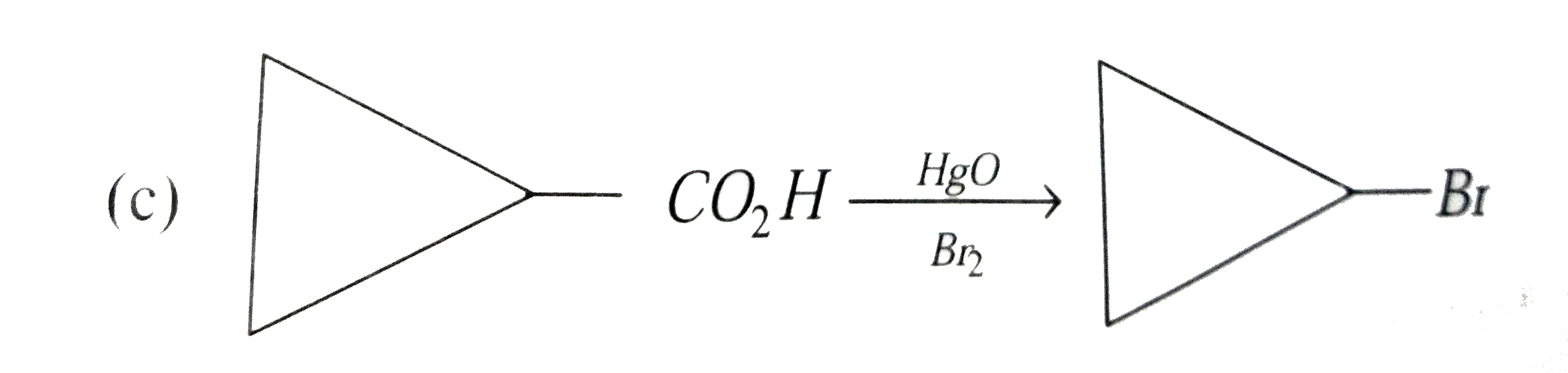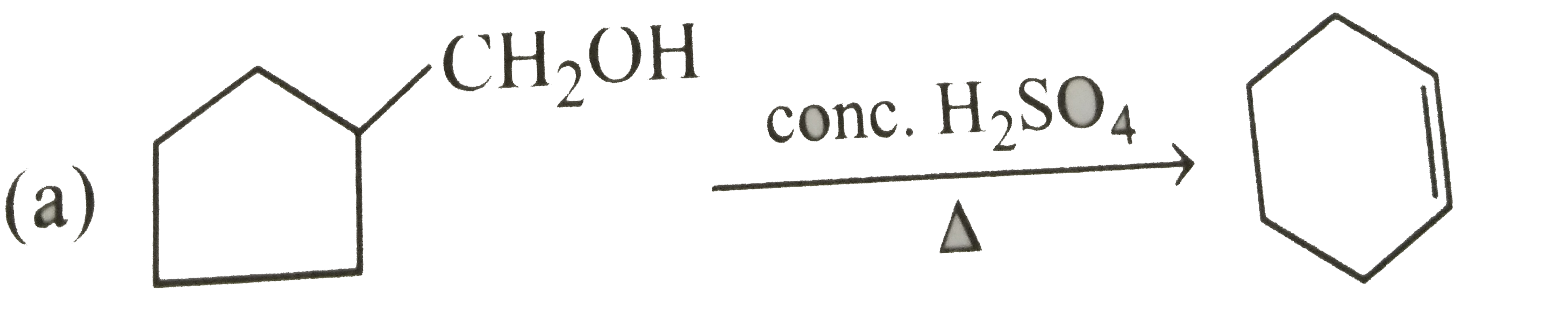Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Select correct order about bond strength |
|
Answer» `O_(2)gtO_(2)^(+1)gtO_2^(-)gtO_(2)^(2-)` |
|
| 2. |
Select correct optionhaving majoor product: |
|
Answer»
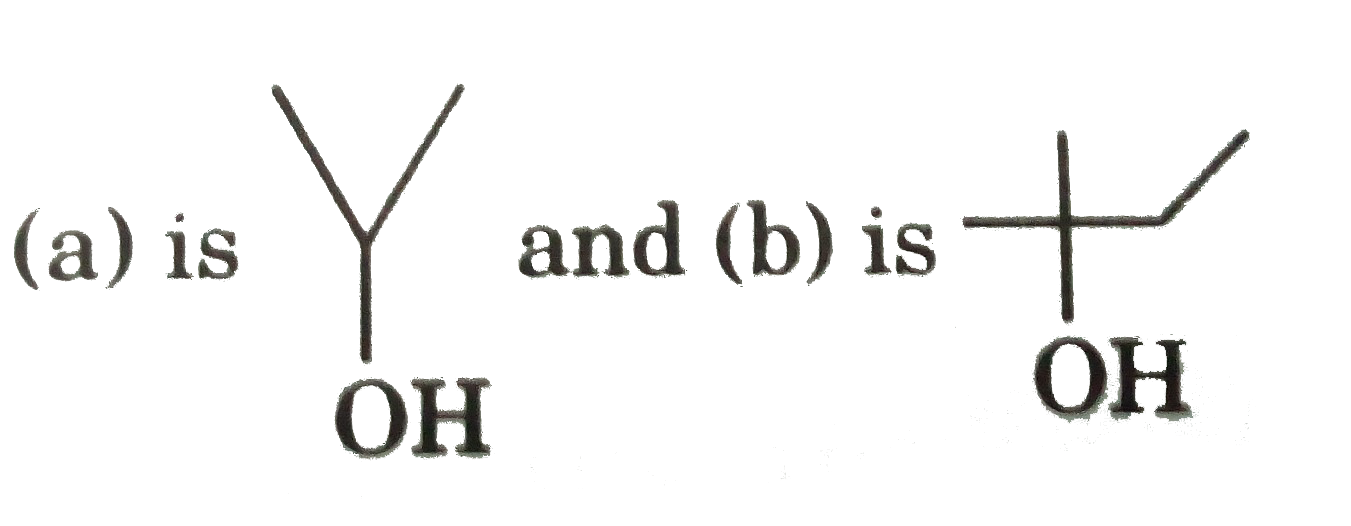
|
|
| 3. |
Select correct option for True (T) and False (F) (i) In the unit cell of NaCl there are 4Na^(+)Cl^(-) units present. (ii) At very high temperature paramagnetic substance changes to ferromagnetic substance. (iii) CrCO_2 is ferromagnetic and has conductivity as that of metal. (iv) Solid ammonia is molecular solid having low melting point. |
|
Answer» TFTT |
|
| 4. |
Select correct option for the statements given with reference to electrochemical cell (where T=true and F=false) (i) In external circuit e^(-) flow from cathode to anode (ii) In solution electricity conducted through ions (iii) In external circuit electric current flow from anode to cathode (iv) Anions move from anode to cathode through salt bridge |
|
Answer» TFTF |
|
| 5. |
Select correct method to prepare Me_(3)C-COOCMe_(3) : |
|
Answer» `Me_(3)C-COOH+Me_(3)C-OHoverset(conc.H_(2)SO_(4))to` |
|
| 6. |
Select correct code about complex [Cr(NO_(2)(NH_(3))_(5)][ZnCl_(4)] (P) IUPAC name of the compound is pentaamminentrito-N-chromium (III) tetrachloridozincate (II) (Q) It shows geometrical isomerism (R ) It shows linkage isomerism (S) It shows co-ordination isomerism |
|
Answer» R, S only |
|
| 7. |
Select correct match: |
|
Answer» |
|
| 8. |
Select correct matching :- |
|
Answer» PYROMETALLURGY : EXTRACTION of Fe |
|
| 9. |
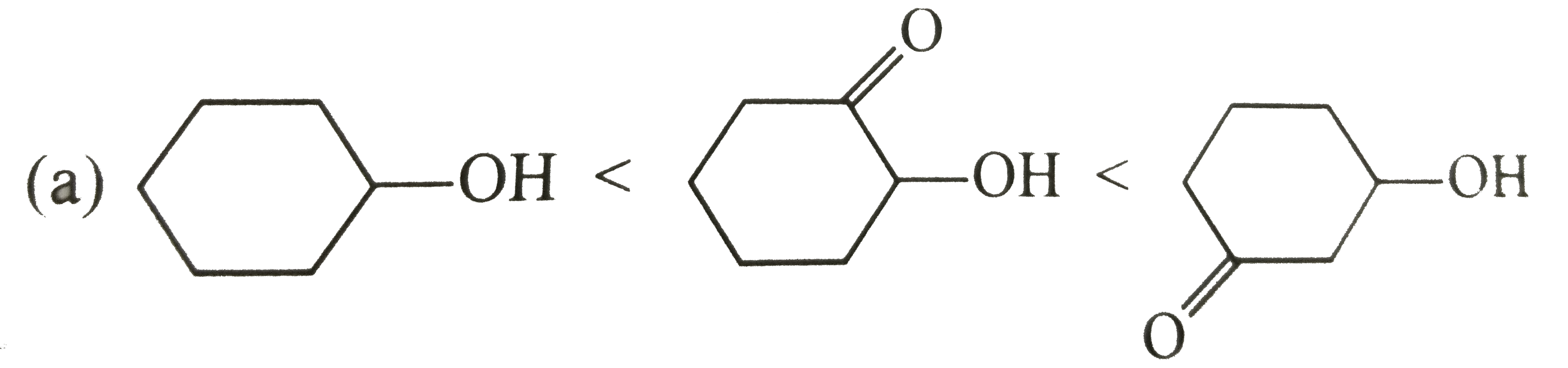
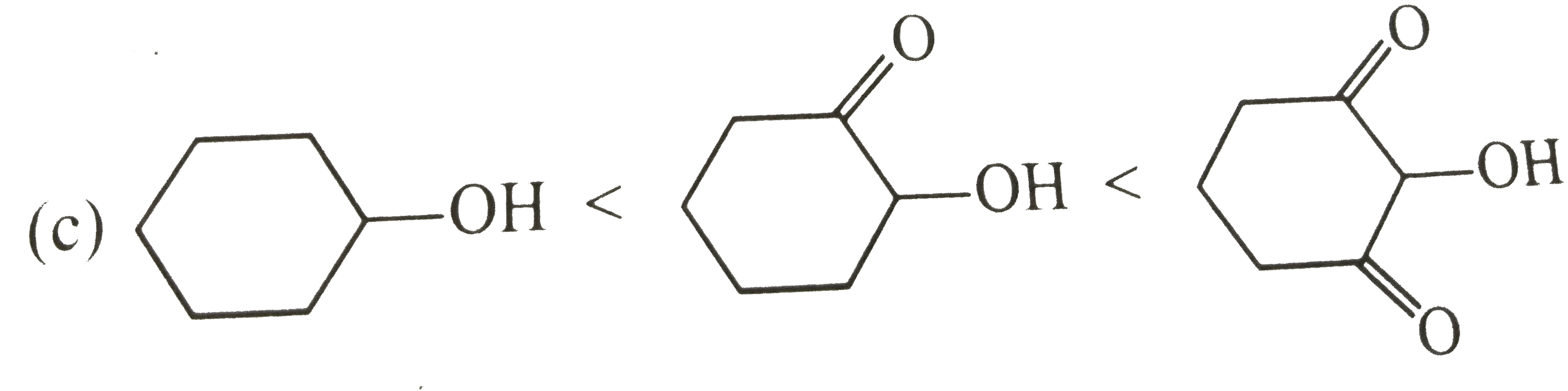
Select correct diagram about the acidic strength of oxides :- |
|
Answer»
|
|
| 11. |
Select compound having maximum solubility in water. |
|
Answer» `P=2P_(0)` 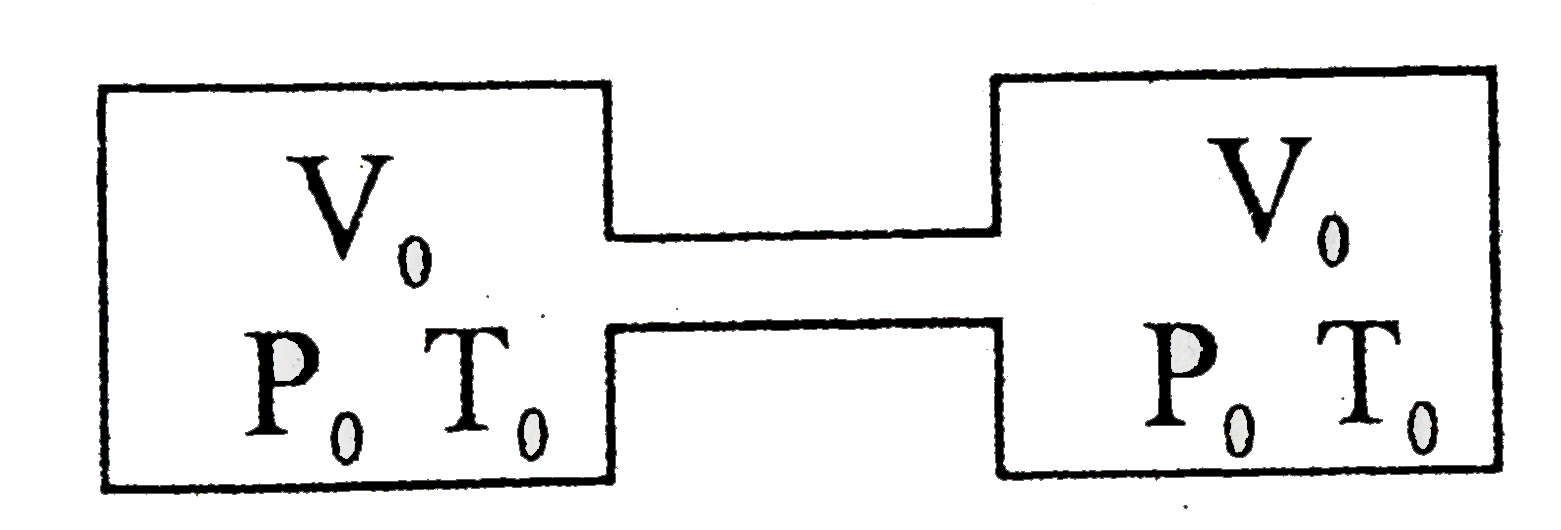 NUMBERS of moles =`(P_(0)V_(0))/(RT_(0)) =x` `(n_(i)R_(0)T_(0))/(V_(0))=(n_(2)R_(0)2T_(0))/(V_(0))` `n_(2)=1/3xx2x =1/3 xx2 xx(P_(0)V_(0))/(RT_(0))` `n_(2)=2/3 (P_(0)V_(0))/(RT_(0))` `P=n_(2)(R(2T_(0)))/(V_(0)) =(2R_(0)V_(0))/(3RT_(0)) xx(R(2T_(0)))/(V_(0))=(4P_(0))/3` |
|
| 12. |
Selectcorrect code:-(a) In (BeH_2)_n all bond's are 3c - 2e. (b) In (B_2H_6), all H-atom are present in a plane (c) In I_2Cl_6, I-Atom sp^3d^2 hybridised (d) In C_2H_6, C-atom used vacant orbital in a hybridisation |
| Answer» Answer :C | |
| 13. |
Select correct about the complex salt formed by the combination of cation of tris(ethylenediamine)platinum(IV)chloride and Anion of sodium tetrachoridolplatinate(II). |
|
Answer» It is `[Pt(NH_(2)CH_(2)CH_(2)NH_(2))_(3)][PtCl_(4)]` |
|
| 14. |
Select correct about covelent character |
|
Answer» `SnCl_(4) gt SnCl_(2)` |
|
| 16. |
Select and write the most appropriate answer from the given alternatives for each sub-question : What is natural rubber? |
|
Answer» Cis-1, 4-polyisoprene |
|
| 17. |
Select and write the most appropriate answer from the given alternatives for each sub-question : Which among the following reducing agent is 'not' used to reduce acetaldehyde to ethyl alcohol? |
|
Answer» Na-Hg and water |
|
| 18. |
Select branched polymer of glucose : |
|
Answer» Amylopectine |
|
| 19. |
Select and write the most appropriate answer from the given alternatives for each sub-question : C_(2)H_(5)-Br+NaI overset("dry acetone")rarr C_(2)H_(5)-I+ NaBr The above reaction is : |
|
Answer» WURTZ reaction |
|
| 20. |
Select and write the most appropriate answer from the given alternatives for each sub-question : Acetaldehyde, when treated with which among the following reagents does 'not' undergo addition reaction? |
| Answer» Solution :Ammonical silver nitrate | |
| 21. |
Select and write the most appropriate answer from the given alternatives for each sub-question: The hybridisation of phosphorus in phosphorus pentachloride is : |
|
Answer» `DSP^(3)` |
|
| 22. |
A polymer which containsester linkage is : |
|
Answer» Teflon |
|
| 23. |
Select and write the most appropriate answer from the given alternatives for each sub-question : |
|
Answer» `CrO_(2) Cl_(2)` |
|
| 24. |
Seiect the incorrect statement(s) about benzene amongst the following |
|
Answer» because of UNSATURATION BENZENE easily UNDERGOES addition |
|
| 25. |
Select a ferromagnetic material from the following |
|
Answer» Dioxygen According to MOT dioxygen `(O_(2))` contain ONE unpaired electron. Hence it is paramagnetic. Chromium (IV) oxide `(CuO_(2))` contain two unpaired electrons in d-orbitals. Hence it is ferromagnetic i.e. maximum paramagnetic. Dihydrogen monoxide `(H_(2)O)`: it is DIAMAGNETIC Benzene `(C_(6)H_(6))`: It is diamagnetic |
|
| 26. |
[Se(H_2 O)_6]^(3+) ion is |
|
Answer» colourless and DIAMAGNETIC Oxidation state of Sc in `[Sc(H_2 O)_6]^(3+)` is `Sc^(3+)` `Sc^(3+) = [Ar] 3d^0 , 4s^(0)` Thus `Sc^(3+)` does not have unpaired ELECTRON, hence it is diamagnetic and colourless. |
|
| 27. |
Sea weeds are important source of |
|
Answer» Iron |
|
| 28. |
Secretion of HCl in the stomach is stimulated by |
|
Answer» Histamine |
|
| 29. |
Secondary structure of proteins refers to |
|
Answer» mainly denatured proteins and the structure of the PROSTHETIC group |
|
| 30. |
Secondary structure of proteins refers to: |
|
Answer» Mainly denatured proteins and structure of prosthetic group |
|
| 31. |
Secondary structure of protein is mainly governed by |
|
Answer» HYDROGEN BONDS |
|
| 32. |
Secondary structure of proteins is due to : |
| Answer» Answer :A | |
| 33. |
Secondary structure of protein refers to |
|
Answer» Mainly denatured proteins and structure of prosthetic groups |
|
| 34. |
Secondary pollutant which stops Hill reaction is |
|
Answer» Aldehydes |
|
| 35. |
Secondary nitroalkanesreact with nitrious acid to form |
|
Answer» RED solution |
|
| 36. |
Secondary nitro alkanes react with nitrous acid to form…………….. |
|
Answer» RED solution |
|
| 37. |
Secondary nitro compounds on hydrolysis in acidic medium give |
|
Answer» ketones |
|
| 38. |
Secondary and tertiary alcohols always give E_(1) reaction indehydration. Primary alcohols whose beta- carbon is 3^(@)or 4^(@) also give E_(1) reaction. However, the primary alcohols whose beta-carbon is 1^(@) or 2^(@) give E_(2) reaction. Dehydrating agents like conc. H_(2)SO_(4), Al_(2)O_(3), anhydrous ZnCl_(2) are used. The reactivity of alcohols for elimination reaction lies in following sequence : Tertiary alcohol gt secondary alcohol gt primary alcohol Electron attracting group present in alcohols increase the reactivity for dehydration. Greater is the -I effect of the group present in alcohol, more will be its reactivity. Both E_(1) and E_(2) mechanism give the product according to Satzeff's rule, i.e., major product is the most substituted alkene. underset("3-Methylbutan-2-ol")(CH_(3)-underset(CH_(3))underset(|)(C)H-overset(OH)overset(|)(C)H-CH_(3)underset("above 413K")overset("conc." H_(2)SO_(4))(rarr)) underset("2-Methylbut-2-ene(major product)")(CH_(3)-underset(CH_(3))underset(|)(C)=CH-CH_(3))+underset("3-Methylbut-1-ene(minor product)")(CH_(3)-underset(CH_(3))underset(|)(C)H-CH=CH_(2)) Which of the following dehydration products is incorrect ? |
|
Answer»
|
|
| 39. |
Secondary and tertiary alcohols always give E_(1) reaction indehydration. Primary alcohols whose beta- carbon is 3^(@)or 4^(@) also give E_(1) reaction. However, the primary alcohols whose beta-carbon is 1^(@) or 2^(@) give E_(2) reaction. Dehydrating agents like conc. H_(2)SO_(4), Al_(2)O_(3), anhydrous ZnCl_(2) are used. The reactivity of alcohols for elimination reaction lies in following sequence : Tertiary alcohol gt secondary alcohol gt primary alcohol Electron attracting group present in alcohols increase the reactivity for dehydration. Greater is the -I effect of the group present in alcohol, more will be its reactivity. Both E_(1) and E_(2) mechanism give the product according to Satzeff's rule, i.e., major product is the most substituted alkene. underset("3-Methylbutan-2-ol")(CH_(3)-underset(CH_(3))underset(|)(C)H-overset(OH)overset(|)(C)H-CH_(3)underset("above 413K")overset("conc." H_(2)SO_(4))(rarr)) underset("2-Methylbut-2-ene(major product)")(CH_(3)-underset(CH_(3))underset(|)(C)=CH-CH_(3))+underset("3-Methylbut-1-ene(minor product)")(CH_(3)-underset(CH_(3))underset(|)(C)H-CH=CH_(2)) Which among the following sequences of reactivity for dehydration is incorrect ? |
|
Answer»
|
|
| 40. |
Secondary butyl alcohol is dehydrated according to |
|
Answer» SAYTZEFF RULE |
|
| 42. |
Secondary amines on oxidation with KMnO_(4) gives: |
|
Answer» DIALKYL hydroxylamine |
|
| 43. |
Secondary amines on oxidation with Caro's acid gives: |
|
Answer» DIALKYL hydroxylamine |
|
| 44. |
Secondary amines react with aldehydes and ketones containing alpha-hydrogens to form____ |
| Answer» SOLUTION :ENAMINES | |
| 45. |
Secondary amine forms yellow oily liquid with nitrous acid. which on warming with phenol and conc. H_(2)SO_(4) given a brown or red colour and which at once change into blue-green. This reaction is called as: |
|
Answer» CARBYLAMINE REACTION |
|
| 46. |
Secondaryamineformsyellowoilyliquidnitronsacid, whichonwarmingwithphenoland conc. H_2 SO_4givesa brown or redcolourandwhichat oncechangesintoblue= green . Thisreactionis calledas : |
|
Answer» CARBYLAMINEREACTION |
|
| 48. |
Secondary amine could be separated by |
|
Answer» reduction of nitriles |
|
| 49. |
Second series of transition elements starts with: |
|
Answer» YITRIUM |
|
| 50. |
Second law of thermodynamics is not a restriction in the working of a fuel cell. Explain. |
|
Answer» Solution :Convential method of converting chemical energy into ELECTRICAL energy INVOLVES three steps Step I: Chemical energy `rarr` Heat energy Step II : Heat energy `rarr` Mechanical energy Step III : Mechanical energy `rarr` Electrical energy The extent of heat CONVERTED into mechanical energy (step II) is limited by the second law of thermodynamics . Hence, the power plants are with only 40% efficiency. But in a fuel CELL, chemical energy is directly converted into electrical energy, with a theoretical 100% efficiency. |
|