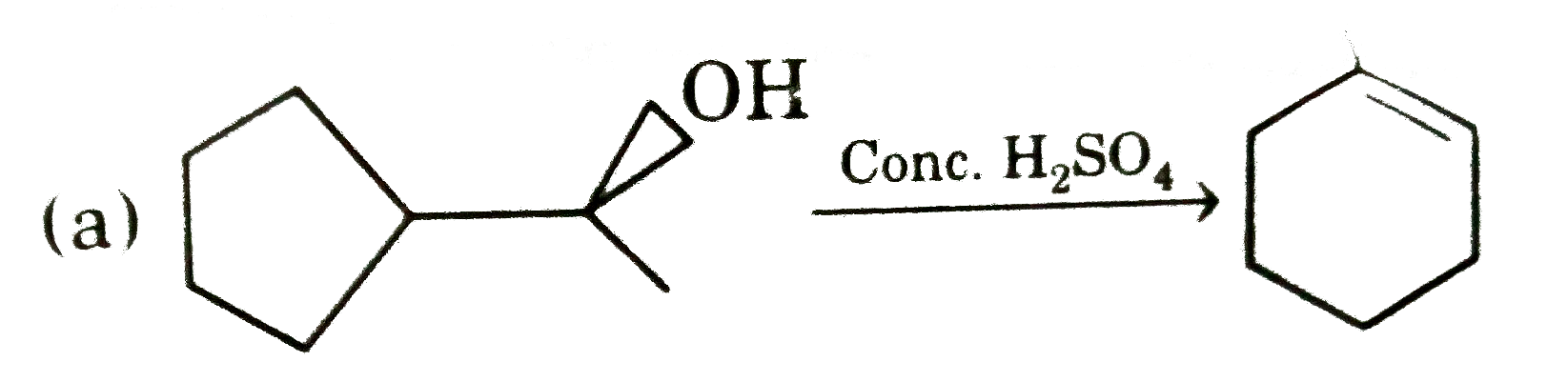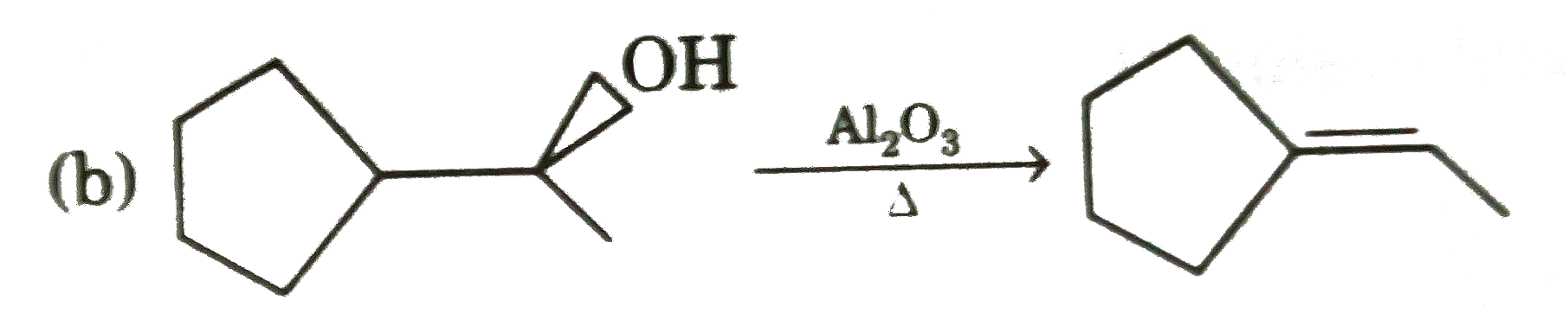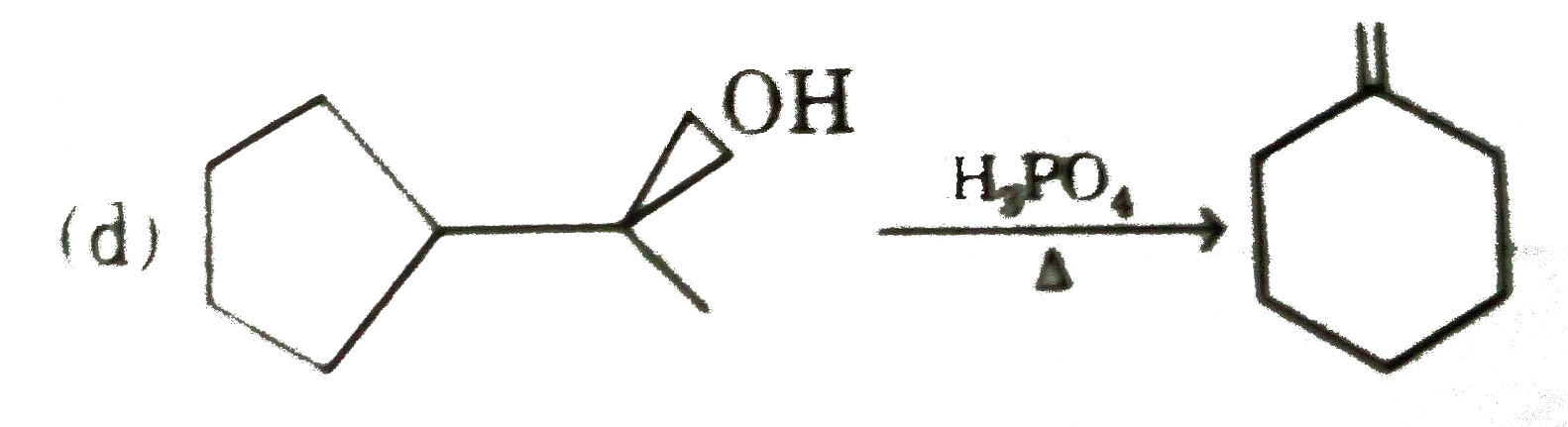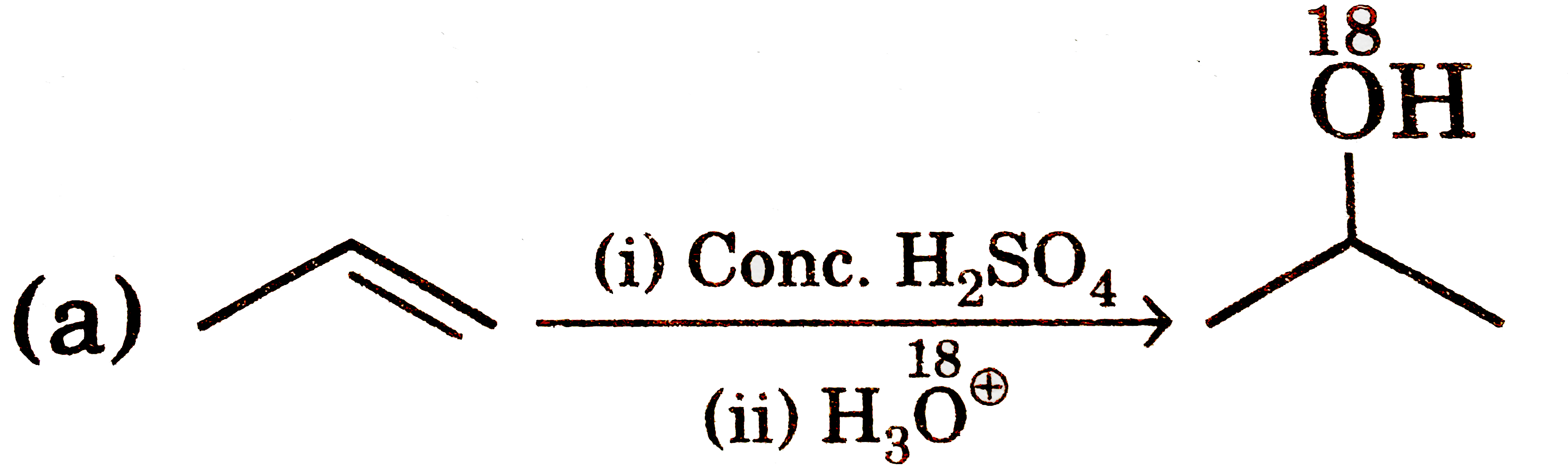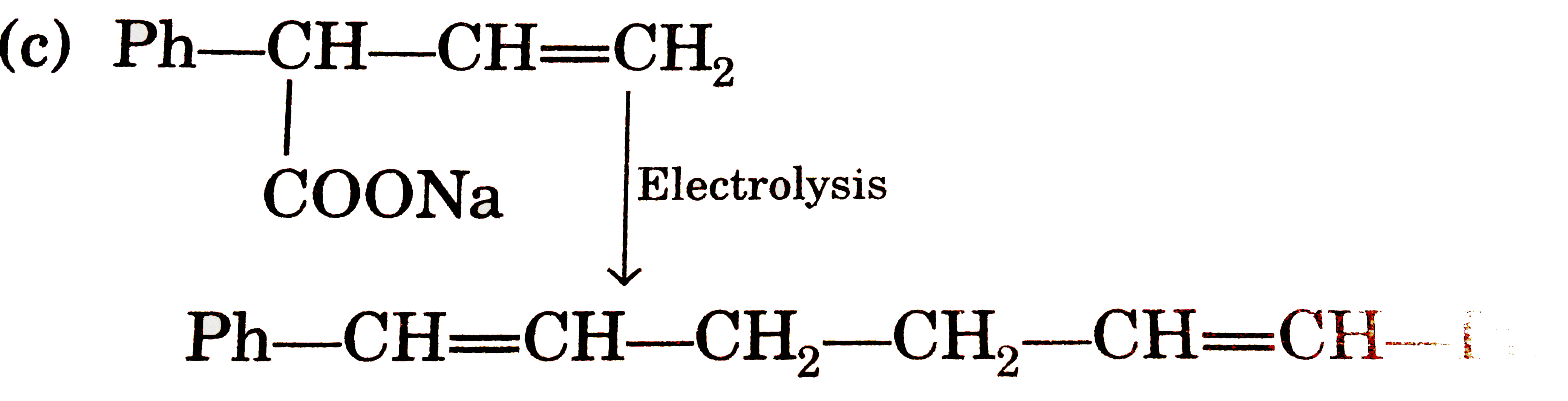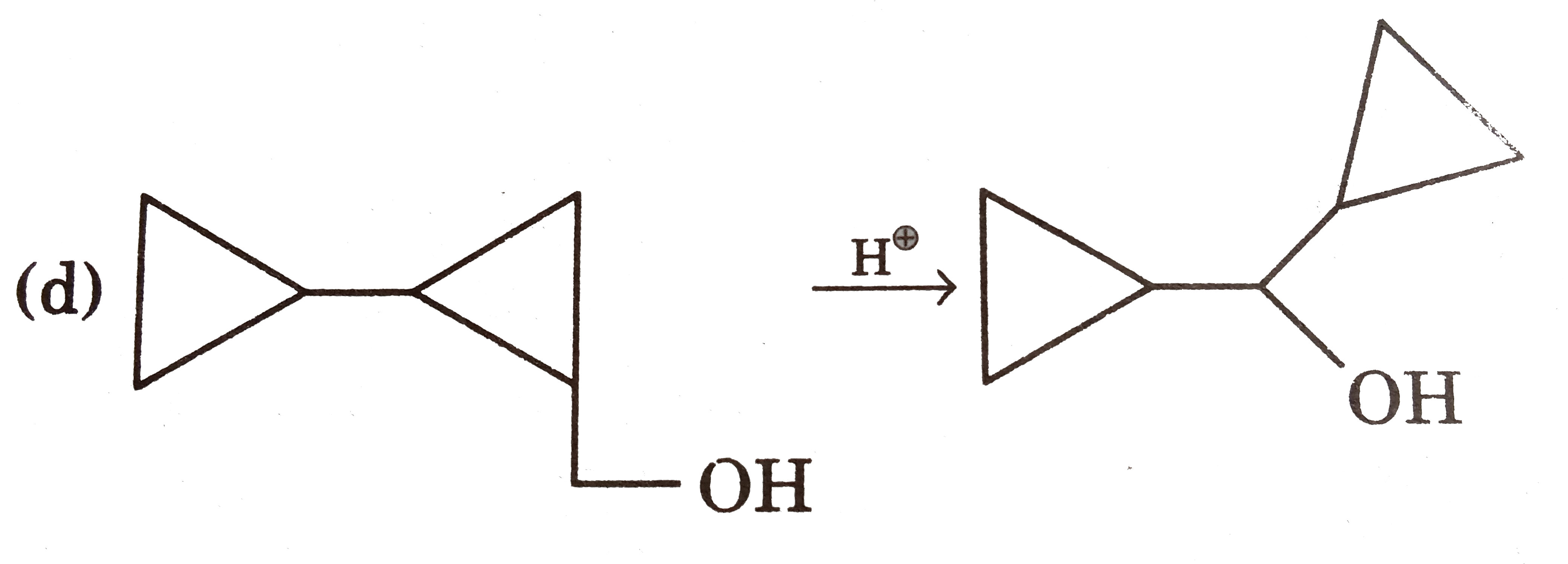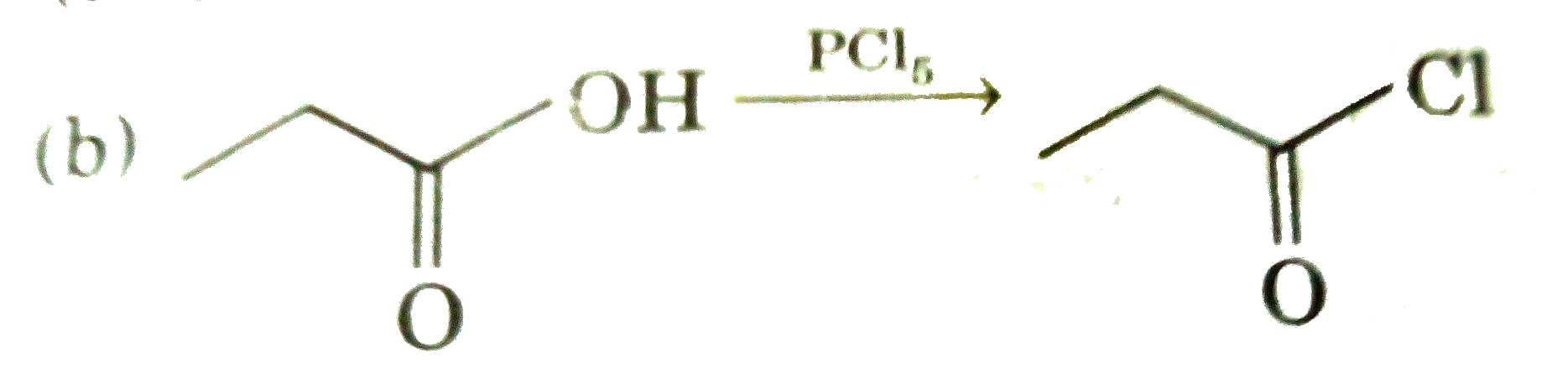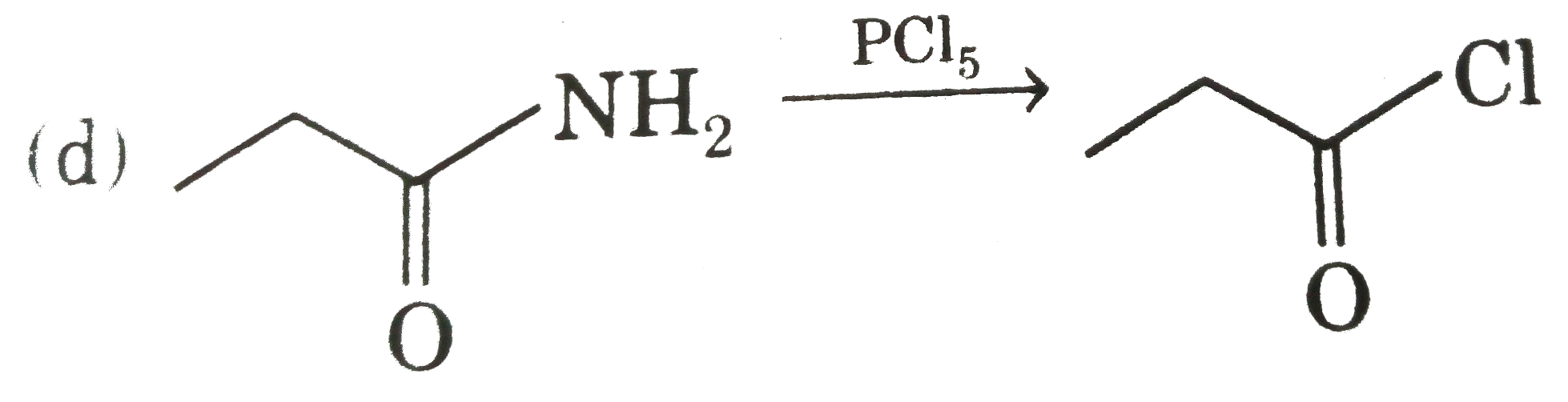Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Select incorrect option : |
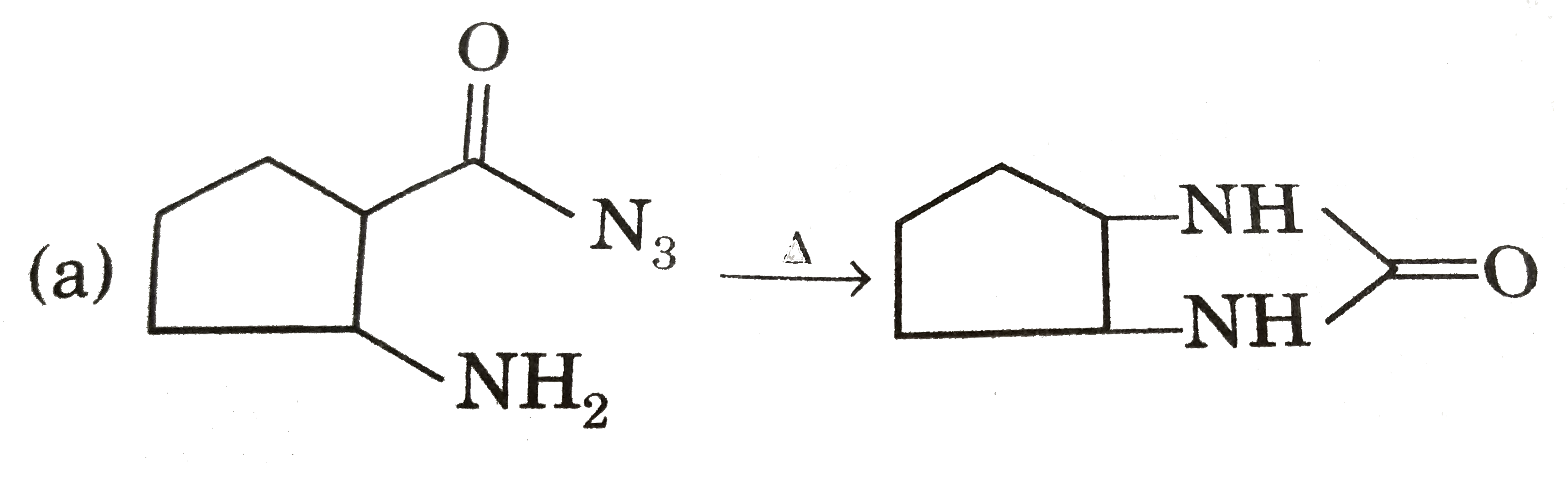
|
Answer» `EtOEt UNDERSET(/_\)OVERSET(NaOH)rarrEtONa+EtOH` (b) 
|
|
| 2. |
Select incorrect characteristics about [Zn(gly)_(2)]^(0) : |
|
Answer» It is diamagnetic in nature |
|
| 3. |
Select incorrect about complex [Fe(en)_(2)(bipy)]SO_(4): |
|
Answer»
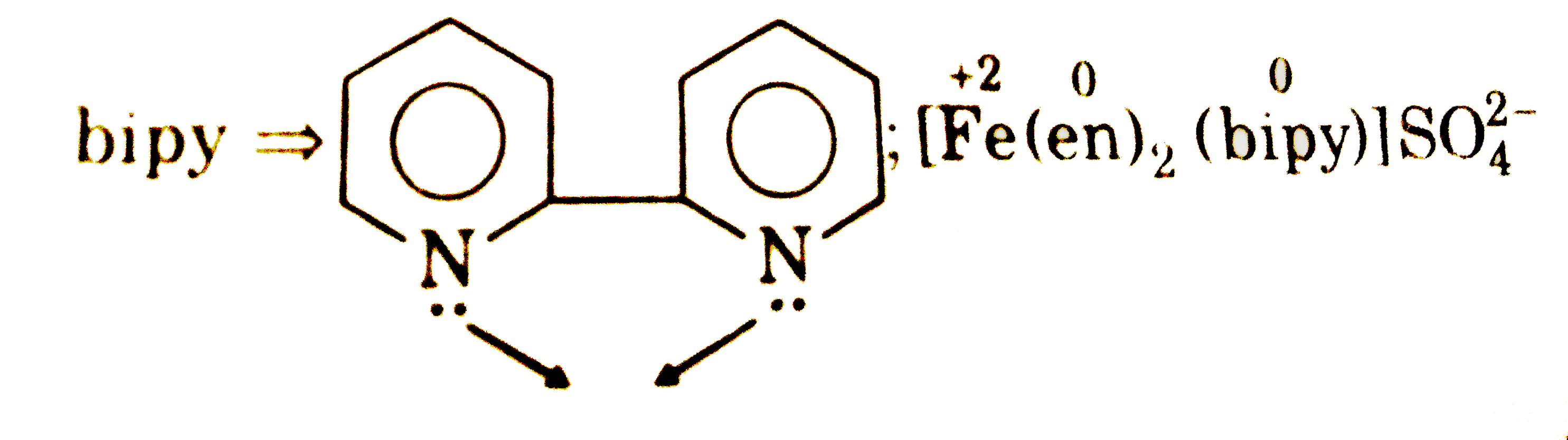
|
|
| 4. |
Select incorrect major product(s) : |
|
Answer»
|
|
| 5. |
Select in which both have sea-saw shape- |
|
Answer» `XeO_(2)F_(2), SiF_(4)` |
|
| 6. |
Select in correct statements : |
|
Answer» Gases which have high VALUE of van der Waals constant ‘a’ are EASILY liquefied |
|
| 7. |
Select in correct statement. |
|
Answer» Solid `PCl_5` EXIST as `[PCl_4]^(+)[PCl_6]^(-)`<BR>Solid `PBr_5` exist as `[PBr_4]^(+)[Br]` |
|
| 8. |
Select from each of thefollowingsets the hydrocarbons havinglowestand higher boilingpoint : (a) n- butane ,n-hexane , n-pentane ( b) n- pentane ,n-hexane ,2,3-dimethylbutane ( C) 3,3- dimethylpentane ,2,3-methylhexane ,n- heptane ,n- heptane |
|
Answer»
(B) higher b.pt ., n-hexane , lowestb.pt ., n- pentane ( c)higherb.pt., n-heptane by HYDROLYSIS,b,pt.,3,3,-dimethypentane . |
|
| 9. |
Select equation having endothermic step: |
|
Answer» `S^(-)(g) to S^(2-)(g)` |
|
| 10. |
Select correctly matched with major product : |
|
Answer»
|
|
| 11. |
Select correctly matched name and structure at given pH : |
|
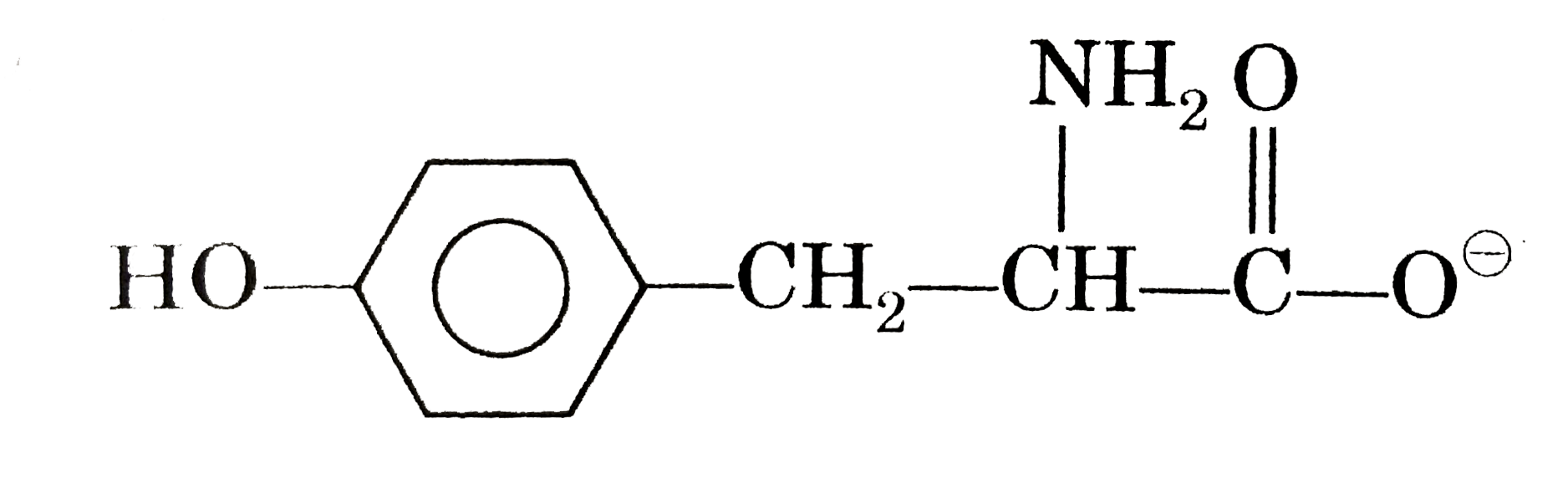
Answer» Alanine at pH = 4,`CH_(3)-overset(COOH)overset(|)(CH)-overset(o+)NH_(3)` (b) 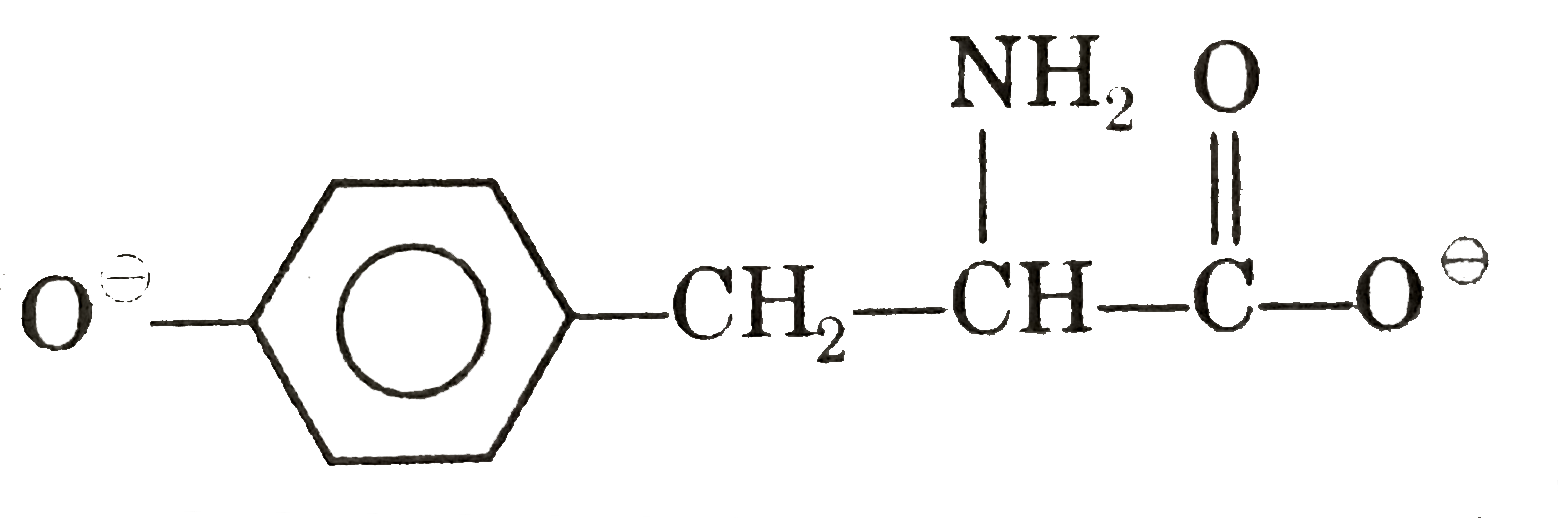
|
|
| 12. |
Select correctly matched major product: |
|
Answer»

|
|
| 13. |
Select correct statements(s): |
|
Answer» We can condense vapours simply by applying pressure |
|
| 14. |
Select correct statements (s) : |
|
Answer» Acidic strength of `HBr gt HCL` but reverse is true for their REDUCING property |
|
| 15. |
Select correct statement(s) regarding sigma and pi bonds : |
|
Answer» `SIGMA` - bond lies on the line joining the nuclei of bonded atoms |
|
| 16. |
Select correct statement(s) regarding [Ni(DMG)_(2)] complex compound: |
|
Answer» it acts as oxidinsing agnet because `NI^(2+)` cation is having E.A.N. 34. |
|
| 17. |
Select correct statement(s) regarding octahedron complex having CFSE=-1.2Delta_(0). |
|
Answer» Compound is neither LOW spin nor high spin complex |
|
| 18. |
Select correct statement(s) regarding given complexes: |
|
Answer» `[Fe(CO)_(5)]`, the orbitals used for HYBRIDISATION in Fe atom are s, `p_(x),p_(y),p_(z),d_(z^(2))` and it is high spin complex |
|
| 19. |
Select correct statements out of following. |
|
Answer» `H_(3)PO_(4)`is conjugate ACID of `HPO_(4)^(-2)` `rarr H_(2)PO_(3)^(-1) " isC.Aof "HPO_(3)^(-2)` `rarr H_(2)PO_(2)^(-1)` can.t behave as an acid `rarr H_(2)PO_(4)^(-1)` is C.B of `H_(3)PO_(4)` |
|
| 20. |
Select correct statement(s) is /are : |
|
Answer» In `AsH_(3)` molecule lone pair at central atom is PRESENT in almost pure s-orbital (b) Number of `p pi- d pi ` bond(s) in `SO_(3)` is TWO and in `SO_(2)` is one (c ) Lewis BASIC order : `NCl_(3) gt NF_(3)` |
|
| 21. |
Select correct statement(s): (I) When excess FeCl_(3) solution is added to K_(4)[Fe(CN)_(6)] solution, in addition to Fe^(III)[Fe^(II)(CN)_(6)]^(-),Fe^(II)[Fe^(III)(CN)_(6)]^(-) is also formed due to side redox reaction (II) When FeCl_(2) is added to K_(3)[Fe(CN)_(6)] solution, in addition to Fe^(II)[Fe^(III)(CN)_(6)]^(-),Fe^(III)[Fe^(II)(CN)_(6)]^(-) is also formed due to side redox reaction. (III) Fe^(III)[Fe^(II)(CN)_(6)]^(-) is paramagnetic while Fe^(II)[Fe^(III)(CN)_(6)]^(-) is diamagnetic. (IV) Fe^(III)[Fe^(II)(CN)_(6)]^(-) is diamagnetic while Fe^(II)[Fe^(III)(CN)_(6)]^(-) is paramagnetic |
| Answer» Answer :A | |
| 22. |
Select correct statement(s) about following reaction : 2R-OH overset(H^+)to R-O-R + H_2O |
|
Answer» It is an example of `S_N` REACTION in which protonated ALCOHOL is the substrate and second molecule of alcohol is the nucleophile. |
|
| 23. |
Select correct statements : |
|
Answer» Gases which have high value of Van der Waals constant `'a'` are EASILY liquefied |
|
| 24. |
Select correct statement(s) : |
|
Answer» hydrophilic COLLOID is a colloid in which there is a strong attraction between the dispersed phase and water |
|
| 25. |
Select correct statement (s), about the following reaction [(CH_(3))_(3)N^(+)CH_(2)CH_(3)]OH^(-) overset(Delta)(rarr) (CH_(3))_(3)N+CH_(2)=CH_(2)+H_(2)O |
|
Answer» This reaction is called Hofmann ELIMINATION |
|
| 26. |
Select correct statement (s) about following reaction : C_(2) H_(5) O^(-) + CH_(3) CH_(2) overset(Br) overset(|) CHCH_(3) overset(C_(2)H_(5)OH) to |
|
Answer» It is termed as `BETA`- elimination reaction `2CH_(3)CHClCH_(3) +2Na overset("dry ether") to (CH_(3))_(2)CH-CH(CH_(3))_(2)` |
|
| 27. |
Select correct statement (s) about monochloroination of methycyclohexane: |
|
Answer» Total 10 monochloro products are obtained. |
|
| 28. |
Select correct statement (s). |
|
Answer» Borax is used as a buffer |
|
| 29. |
Select correct statement (s) : |
|
Answer» Only one`1^(@)` MONOCHLORO product is obtained. |
|
| 30. |
Select correct statement regarding [Ni(DMG)_(2)] complex compound. |
|
Answer» It acts as oxidising agent because `NI^(2+)` CATION is having EAN 34. |
|
| 31. |
Select correct statement regarding silver extraction process . |
|
Answer» When the LEAD-silver alloy is RICH in silver, lead is removed by the curpellation process |
|
| 32. |
Select correct statement (s) : |
|
Answer» strongest force of attraction in alkyl halide is the LONDON force |
|
| 33. |
Select correct statement for Cr. 6NH_3.Cl_3 and Cr.5NH_2.Cl_3 |
|
Answer» In both COMPLEX COMPOUNDS SECONDARY valency is satisfied by only `NH_3` |
|
| 34. |
Select correct statement about valence - bond approach : |
|
Answer» Each bond is formed by maximum OVERLAP for its maximum stability |
|
| 35. |
Select correct statement about the reaction : NH_(4)NO_(3) to N_(2) + 2H_(2)O |
|
Answer» |
|
| 36. |
Select correct statement among following |
|
Answer» Compound 'C' on TREATING with conc. NaOH showns disprotionation reaction. |
|
| 37. |
Select correct statement about product p_(1) ? |
|
Answer» It gives yellow ppt. with `NaOH//I_(2)` |
|
| 38. |
Select correct statement. |
|
Answer» CANE SUGAR is laevorotatory. |
|
| 39. |
Select correct statement. |
|
Answer» Lassaigen's extrect made by E gives prussian BLUE colour with `FeSo_(4)+FeCl_(3)`. |
|
| 40. |
Select correct statement - |
|
Answer» b.p. of 1 molal NaCl solution is TWICE that of 1 molal SUCROSE solution |
|
| 41. |
Select correct statement : |
|
Answer» Product mixture is having no optical activity if ALCOHOL is FULLY consumed. |
|
| 42. |
Selectcorrect statement : |
|
Answer» Geometricalisomersof COMPLEXES may differin dipole MOMENT and visible / UV SPECTRA |
|
| 43. |
Select correct statement : |
|
Answer» The `d_(x^(2)-y^(2))` and `d_(z^(2))` orbitals are called `e_(g)` orbitals for octahedral splitting |
|
| 44. |
Select correct set of species which can't react with water burt react with NaOH, (i) NO_(2) (ii) P_(4) (iii) Al (iv) I_(2) |
|
Answer» Only (IV) 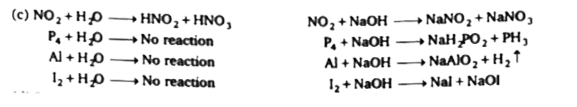
|
|
| 45. |
Select correct sreaction : |
|
Answer»
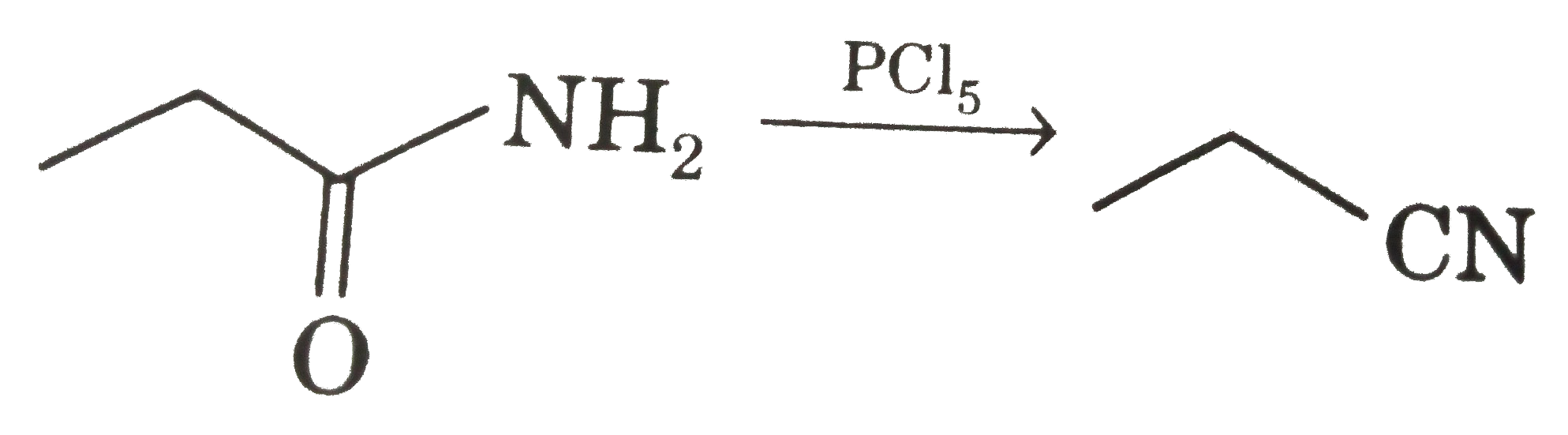
|
|
| 46. |
Select correct properties of emulsions |
|
Answer» emulsions EXHIBIT TYNDALL effect oil emulsions are more viscous than the aqueous emulsions for AQ emulsions electrical conductance more emulsions exhibit brownian moment |
|
| 47. |
Select correct order of stability of different forms of 1-Chloro-4-iodo cyclohexane. |
|
Answer» `IV GT III gt I gt II` |
|
| 48. |
Select correct order(s) of electronegativity of element is/are: |
|
Answer» Paulling scale (E.N. of F-atom)gtMulliken scale (E.N. of F-atom) |
|
| 49. |
Select correct order of dipole moment? |
|
Answer» `B_(3)N_(3)H_(6)gtC_(6)H_(6)` (c) 
|
|