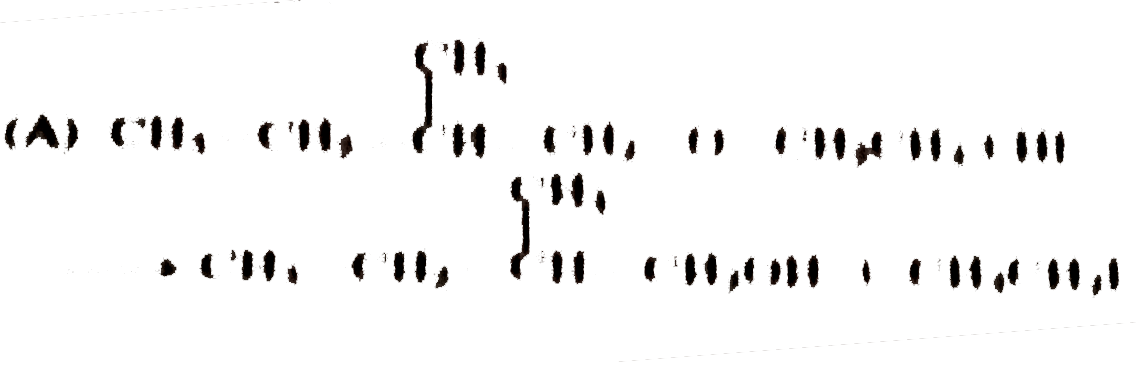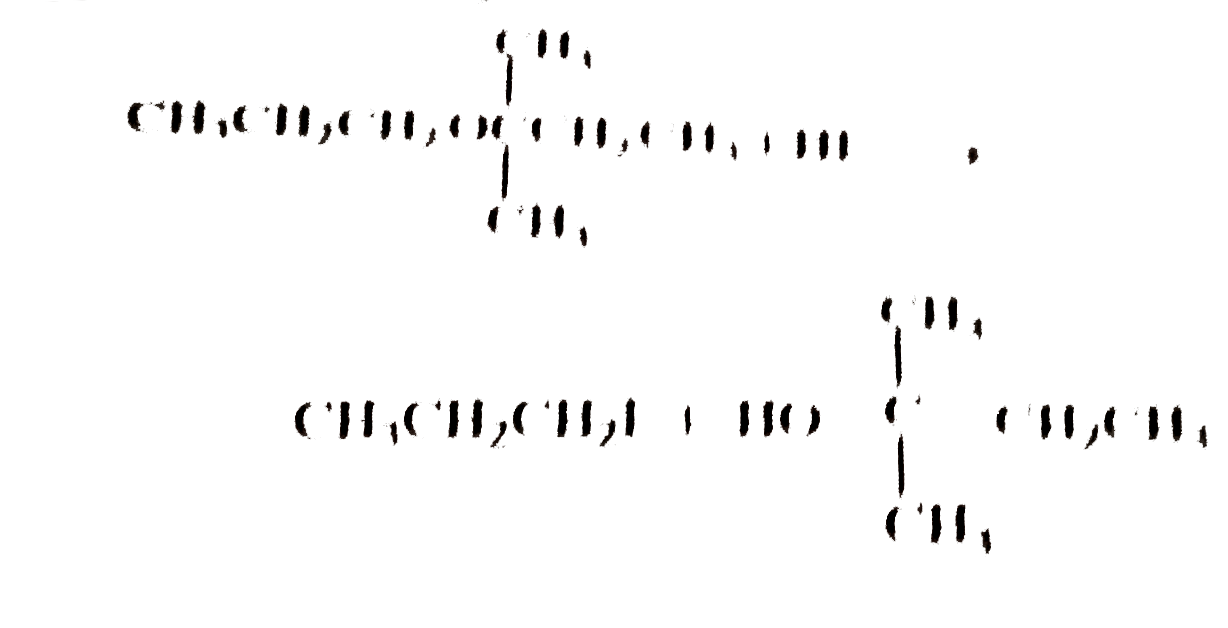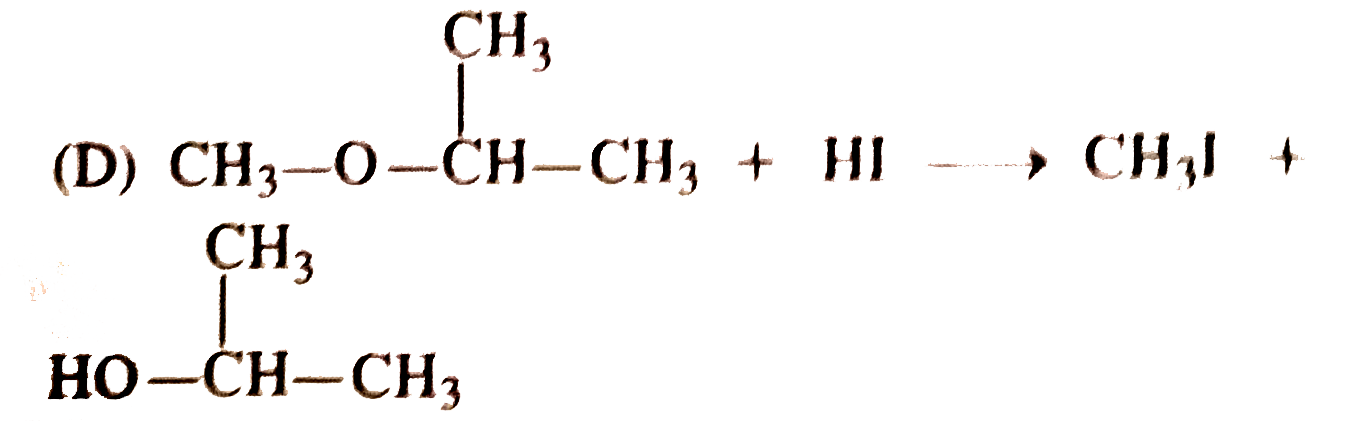Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Pick out the odd ones from the following sets:(i) Sulphur, Argon, Solid CO_2, Diamond ,. (ii) SiC, Quartz,BaO,Graphite |
|
Answer» SOLUTION :(i) Diamond because all others are molecular SOLIDS. (ii) BaO because all others are COVALENT solids. |
|
| 2. |
Pick out the one which does not belong to the family |
|
Answer» pepsin |
|
| 3. |
Pick out the odd ones from among the following compounds on the basis of their medicinal properties. (i) Luminal, seconal, phenacetin, equanil (ii) Chloroxylenol, phenol, chloramphenicol, bithional. |
| Answer» SOLUTION : (i) PHENACETIN is an ANTIPYRETIC while the remaining THREE are tranquillisers. (ii) Chloramphenicol is an antibiotic while the remaining three are ANTISEPTICS. | |
| 4. |
Pick out the odd one from amongst the following compounds on the basis of their medicinal properties mentioning the reason : luminal, seconal, phenacetin, equanil. |
| Answer» SOLUTION :PHENACETIN,-an ANTIPYRETIC while OTHERS are TRANQUILLIZERS. | |
| 5. |
Pick out the odd one amongston the basis of their medicinal properties. Give suitable reason : Sucralose, aspartame, alitame, sodium benzoate. |
| Answer» SOLUTION :SODIUM benzoate is a food preservative. Other THREE are artificialsweetners. | |
| 6. |
Pick out the odd one amongston the basis of their medicinal properties. Give suitable reason : Luminal, seconal, terfenadine, equanil. |
| Answer» SOLUTION :TERFENADINE is antihistamine other THREE are used as TRANQUILIZERS. | |
| 7. |
Pick out the odd one. |
|
Answer» `._(92)U^(238)RARR ._(90)Th^(234)+._(2)He^(4)` |
|
| 8. |
Pick out the odd one amongston the basis of their medicinal properties. Give suitable reason : Chloroxylenol, phenol, chloamphenicol, bithional. |
| Answer» Solution :Chloramphenicol is a BROAD spectrum antibiotic. Other THREE haveantiseptic PROPERTIES. | |
| 9. |
Pick out the odd one among the following: |
|
Answer» `CH_(3)CHOHCH_(3)` |
|
| 10. |
Pick out the odd from amongst the following on the basis of their medicinal properties mentioning the reason. Chloroxylenol, phenol, chloramphenicol, bithional. |
| Answer» SOLUTION :Chloramphenicol-an ANTIBIOTIC while OTHERS are ANTISEPTICS. | |
| 11. |
Pick out the more reactive metal |
|
Answer» Cu |
|
| 12. |
Pick out the mononuclear carbonyl among the following |
|
Answer» ` [Co_2(CO)_s]` |
|
| 13. |
Pick out the incorrect staterment. |
|
Answer» `I_(2)O_(5)` is formed by heating `HIO_(3)", to "170^@C` `2I_(2)O_(5) overset(300^(@)C)to 2I_(2)+5O_(2)` (a) `2HIO_(3) overset(170^(@)C)to I_(2)O_(5)+H_(2)O` (c) `I_(2)O_(5)` oxidizes CO to `CO_(2)` quantitatively at room TEMPERATURE, liberating `I_(2)`, which can be tirated with SODIUM thoiosulphate. `I_(2)O_(5)+5CO to 5CO_(2)+I_(2)` (d) `2I_(2)+3O_(3)to I_(4)O_(9)` `4I_(4)O_(9) overset( GT 75^(@)C)to 6I_(2)O_(5)+2I_(2)+3O_(2)` |
|
| 14. |
Pick out the incorrect statement regarding anisole. |
|
Answer» Anisole is not as reactive as phenol |
|
| 15. |
Pick out the incorrect statement with regard to catalyst |
|
Answer» Presence of catalyst does not change the nature of products |
|
| 16. |
Pick out the incorrect statement w.r.t. NH_3 |
|
Answer» It contains a lone pair of ELECTRONS, which can bonds to a proton to form tetrahedra `NH_(4)^(+)` ions `4NH_3+3O_(2) to 2N_(2)+6H_(2)` a) NH, molecule is trigonal pyramidal is shape with a lone pair of electron in one of the four `sp^3` hybridized orbitals. When it is bonded to `H^+`, it forms tetrahedral `NH_(4^(+))` ions, because all the four sp hybrid orbitals possess four bonded pairs of electrons. b). `2NH_(3)+3CuO to N_(2)+3H_(2)O+Cu` d). `4NH_(3)+5O_(2) overset(Pt//Rh)to 4NO+6H_(2)O` |
|
| 17. |
Pick out the incorrect statement regarding hydrogen halides. |
|
Answer» Hydrogen chloride can be prepared by the reaction of NaCl with conc. `H_2SO_4` `2HX +H_(2)SO_(4)to SO_(2)+2H_(2)O+X_(2), X=Br,I` (a) (i) `NaCl+H_(2)SO_(4) to NaHSO_(4)+HCl("in the cold")` (ii) `NaHSO_(4)+NaCl to NaHSO_(4)+HCl("on heating")` (c) `PX_(3)+3H_(2)O to H_(2)PO_(3)+3HX` For example `PCl_(3)+3H_(2)O to H_(3)PO_(3)+3HCl` `underset(("Red"))(2P)+3Br_(2) to 2PBr_(3)` `underset(("Red"))(2P)+3I_(2) to 2PI_(3)` `PBr_(3)+3H_(2)O to H_(3)PO_(3)+3HBr_(2)` `and PI_(3)+3H_(2)O to H_(3)PO_(3)+3HI` |
|
| 18. |
Pick out the incorrect statement regarding ozene |
|
Answer» `O_3` is an emetable, dark-blue diamagnetic gas `BaO_(3) +O_(3) to BaO+2O_(2)` (a). `O_(3)` is an unstable, because its decomposition is exothermic. The blue colour is due to intenstion absorption of red light. It also absorbs strongly in UV region `(lambda=255mm)` b. In 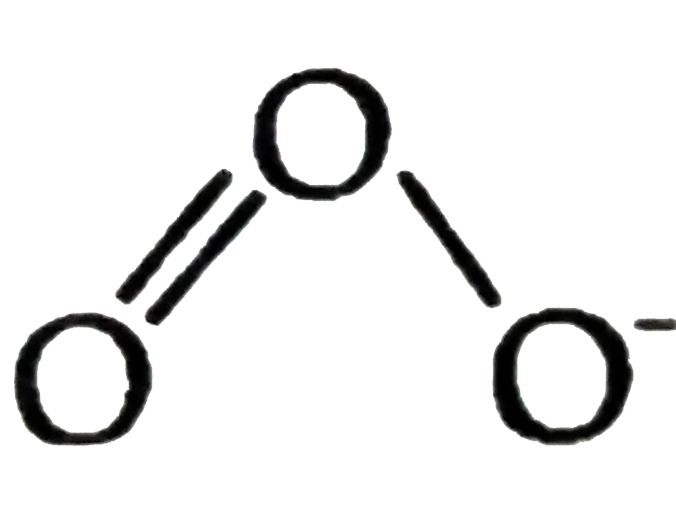 there are three electron pairs (all bonds pairs) AROUND central O atom. To accommodate three electron pairs in trigonal planar orientation (VSEPR THEORY), the central O atom is `sp^(2)-"hybridized"`. there are three electron pairs (all bonds pairs) AROUND central O atom. To accommodate three electron pairs in trigonal planar orientation (VSEPR THEORY), the central O atom is `sp^(2)-"hybridized"`. c. In presence of `O_(3)`, Hg is oxidized to a suboxide. If then, starts sticking to glass and loss mobality. This is TERMED as TAILING of mercury. |
|
| 19. |
Pick out the incorrect statement regarding H_2SO_4. |
|
Answer» When treated with `H_2SO_(4), HCOOH" form "CO and H_2O` `8HI+H_(2)SO_(4) to 4H_(2)O+H_(2)S+4I_(2)` Thus, `H_(2)SO_(4)` oxidises both HBr and HI. (a). `HCOOH underset(-H_(2)O)overset(H_(2)SO_(4))to CO+H_(2)O` `(b). C_(6)H_(12)O_(6) underset(-6H_(2)O)overset(H_(2)SO_(4))to 6C+6H_(2)O` `(d) (a) NaNO_(3)+H_(2)SO_(4) overset("Warming")to NaHSO_(4)+HNO_(3)` |
|
| 20. |
Pick out the incorrect statement about CO . |
|
Answer» In laboratory it is prepared by dehydrating HCOOH with conc. `H_2SO_4` |
|
| 21. |
Pick out the incorrect statement for SO_2. |
|
Answer» It TURNS filter paper moistened with acidified `K_2Cr_2O_7` SOLUTION green |
|
| 22. |
Pick out the incorrect statement for CIO_(3) |
|
Answer» `CIO_(2)` is a powerful oxidizing and chlorinating agent and is prepared in the laboratory from `NaCIO_(3)` and oxalic acid. `2NaClO_(3)+2(COOH)_(2) overset(H_(2), 90^(@)C)to 2ClO_(2)+2CO_(2)+(COONa)_(2)+2H_(2)O` `(b) 2ClO_(2)+2NaOH+H_(2)O_(3) to 2NaClO_(2)O_(3)+2H_(2)O` (c) `2ClO_(3)+2O_(3) to Cl_(2)O_(6)+2O_(3)` . |
|
| 23. |
Pick out the incorrect statement for CIF_(3)^(-) |
|
Answer» It has trigonal planar geometry 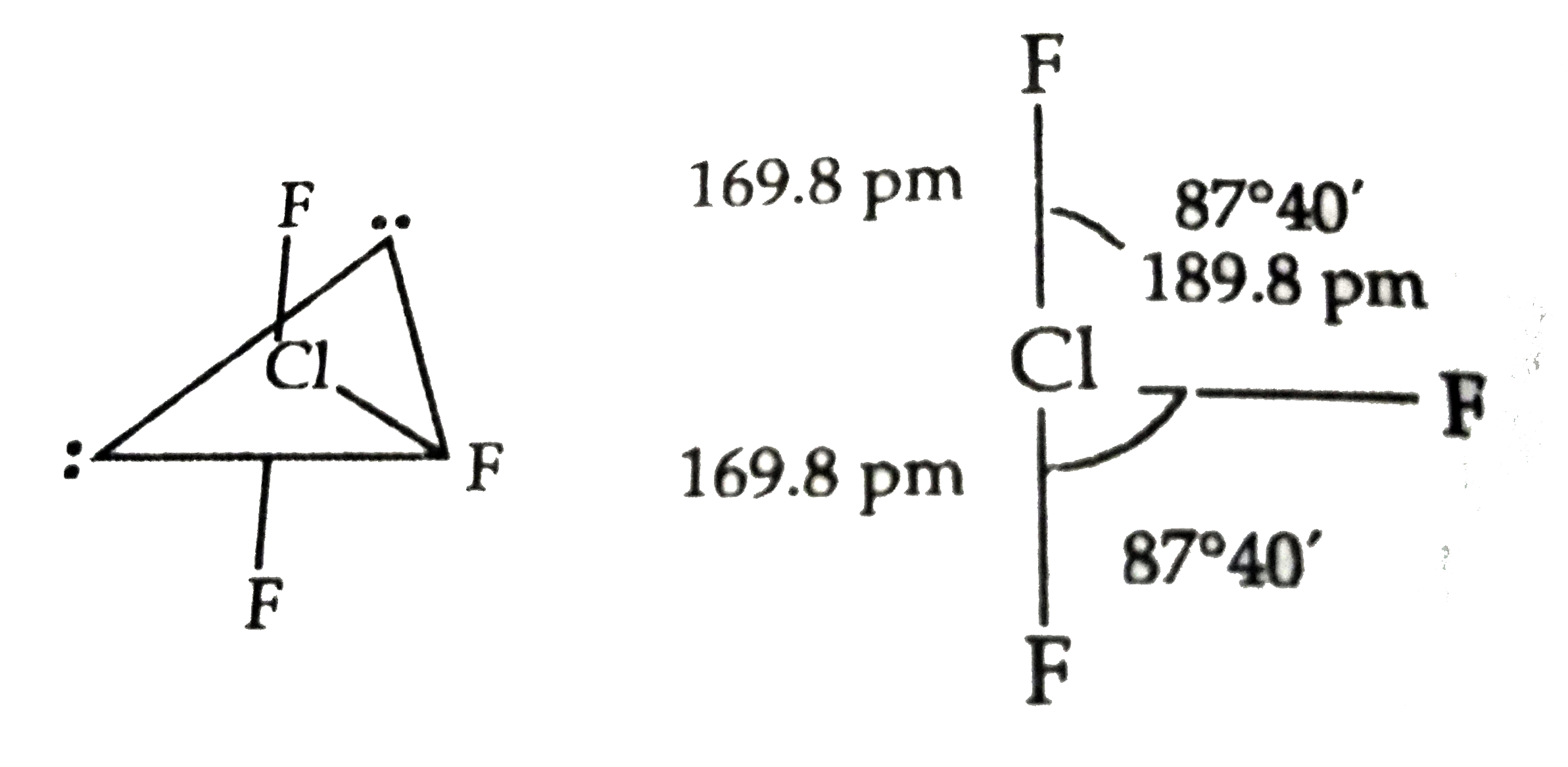 `(B) 2ClF_(3)+U to UF_(6)+Cl_(2)` (c) `4CIF_(3)+2Al_(2)O_(3) to 4AlF_(3)+2Cl_(2)+3O_(2)` `(b) 2CIF_(3)+2NH_(3) to 6HF+Cl_(2)+N_(2)` (d) `4ClF_(3)+3NH_(2)NH_(2) to 12HF+3N_(2)+2Cl_(2)` |
|
| 24. |
Pick out the incorrect statement about glycolysis |
|
Answer» It is anaerobic respiration |
|
| 25. |
Pick out the incorrect statement about ATP. |
|
Answer» It is a nucleotide |
|
| 26. |
Pick out the incorrect statement: |
|
Answer» `MnO_(4)^(2-)` is quite strongly oxidizing and stable only in very strong alkalies, In dilute ALKALI, neutral solutions, it disproportionates |
|
| 27. |
Pick out the incorrect statement. |
|
Answer» Orthophosphorus acid can be obtained by reacting `P_O_6" wIth "H_O` `O-underset(OH)underset(|)overset(OH)overset(|)(P)-OH+H" "O+underset(OH)underset(|)overset(OH)overset(|)(P)=O to O-underset(OH)underset(|)overset(OH)overset(|)(P)-O-underset(OH)underset(|)overset(OH)overset(|)(P)=O+H_(2)O` Although pyrophosphoric is tetrabais acid, yet only two sodium salts are known `Na_(2)P_(2)O_(7) and Na_2H_(2)P_(2)O_(7)`. `P_(4)O_(6)+6H_(2)O to 4H_(3)PO_(3) ` It structure is `O=underset(OH)underset(|)overset(H)overset(|)P-OH` Therefore, it is diabastic (b) `P_(4)O_(10)+6H_(2)O to 4 [O=underset(OH)underset(|)overset(OH)overset(|)P-OH]` This acid is tribasic. (d) `H_(3)PO_(4) overset(316^(@)C)to HPO_(3)+H_(2)O` The metaphosphate IONS up FORM `PO_(4)` units and are cyclic. The trimetaphosphate ion `(P_(3)O_(9)^(3-))` is shown below: 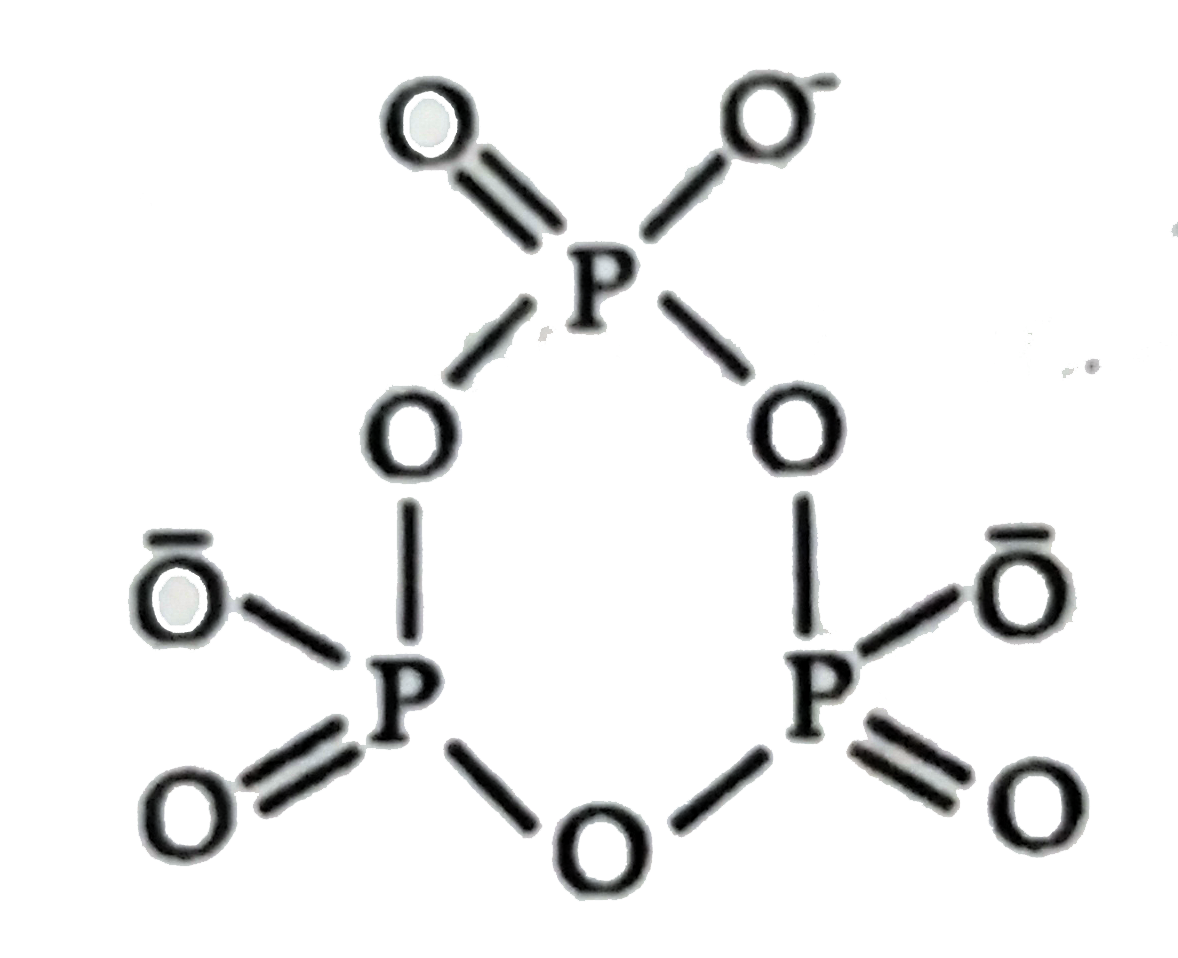
|
|
| 28. |
Pick out the incorrect match of intermolecular attraction between molecule/ion in the following pairs |
|
Answer» HBr and `H_(2)S`–DIPOLE-dipole attraction |
|
| 29. |
Pick out the incorrect statement : |
|
Answer» `MnO_4^(2-)` is quite STRONGLY oxidizing and stable only in very strong alkalies. In DILUTE ALKALI, neutral solutions, it disproportionates. |
|
| 30. |
Pick out the ideal conditions needed for the manufactor of H_(2)SO_(4) by contact process. |
|
Answer» Low temperature, high PRESSURE anid high CONCENTRATION of reactants |
|
| 31. |
Pick out the ideal condition for H_(2)SO_(4) manufactured by Contact process |
|
Answer» Low TEMPERATURE, HIGH pressure and high CONCENTRATION of REACTANTS |
|
| 32. |
Pick out the false statement. |
|
Answer» Vapour pressure of a liquids is the MEASURE of the strength of intermolecular ATTRACTIVE forces |
|
| 33. |
Pick out the equation in which the products formed is not correct ? |
|
Answer»
`CH_3CH_2CH_2overset(CH_3)overset(|)UNDERSET(CH_3)underset(|)(OC) CH_2CH_3+HIto` `CH_3CH_2CH_2OH+I-overset(CH_3)overset(|)underset(CH_3)underset(|)(C)- CH_2CH_3` This is because the ether contains a `3^(@)` group and in such a CASE `3^(@)` alkyl iodide is the major PRODUCT along with the `1^(@)` alcohol. |
|
| 34. |
Pick out the correct statement(s) of the following. |
|
Answer» All atoms with an odd atomic number are NECESSARILY paramagnetic |
|
| 35. |
Pick out the effect of catalyst on activation energy ? |
|
Answer» Catalyst lowers the activation energy |
|
| 36. |
Pick out the correct statements from the following. (I) Cobalt (III) is more stable in octahedral complexes. (II) Zinc froms coloured ion or complexes. (III) Most of the d-block elements and their compounds are ferromagnetic. (IV) Osmium show (VIII) oxidation sate. |
|
Answer» I and II (II) Zn exhibits only `+2` oxidation STATE.So, `Zn^(2+)=[Ar]3d^(10),4s^(0)` Since, iy does not contain any unpaired electron, its compounds are colourless. (III) d-block elements are generally paramagnetic and sometime DIAMAGNETIC , but not ferromagnetic. (IV) OSMIUM and ruthenium are VIII group elements, so can exibit the highest oxidation state`+8` in their oxides, HENCE, statements I and IV are correct. |
|
| 37. |
Pick out the correct statements from the following : 1. Cobalt (III) is more stable in octahedralcomplexes. 2. Zinc forms coloured ions or complexes. 3.Most of the d-block elements and their compounds are ferromagnetic . 4. Osmium shows (VIII)oxidation state. 5.Cobalt (II) is more stablein otahedral complexes. |
|
Answer» 1 and2 |
|
| 38. |
Pick out the correct statements from the following 1. Cobalt (III) is more stable in octahedral complexes 2. Zinc forms coloured complexes 3. Most of the d-block elements and their compounds are ferromagnetic 4. Osmium show (VIII) oxidation state 5. Cobalt (II) is more stable in octahedral complexes |
|
Answer» 1 and 2 |
|
| 39. |
Pick out the correct statements among the following from inspection of standard reduction potentials (Assume standard state conditions). Cl_2(aq.)+2e |
|
Answer» `Cl_2` can oxidise `SO_4^(2-)` from SOLUTION<BR>`Cl_2` can oxidise `Br^-` and `I^-` from aqueous solution |
|
| 40. |
Pick out the correct statement(s): |
|
Answer» `MnO_(2)` dissolves in conc.HCl but does not form `Mn^(4+)` ions (b)Decomposition of acidic `KMnO_(4)` is catalyst by sunlight `MnO_(4)^(-)+4H^(+) to 4MnO_(4)+2H_(2)O+3O_(2)` (c)`underset("green")(3K_(2)MnO_(4))+2H_(2)O to underset("purple")(2KMnO_(4)^(-)+underset("dark brown")(MnO_(2))(darr)+4KOH` (d) `KMnO_(4)` also acts as an OXIDIZING agent in alkaline medium. `MnO_(4)^(-)+2H_(2)O +3E^(-) to MnO_(2)+4OH^(-), E^(@)=+1.23` volt |
|
| 41. |
Pick out the correct Statements |
|
Answer» ACTIVATED complexis an unstable intermediate with MAXIMUM energy and normal bonds |
|
| 42. |
Pick out the correct statement(s) |
|
Answer» `MnO_(2)` dissolves in conc. `HCl`, but does not form `Mn^(4+)` ions (b) Decomposition of acidic `KMnO_(4)` is catalysed by sunlight. `4MnO_(4)^(-)+4H^(+)rarr 4MnO_(2)+2H_(2)O+3O_(2)` (c) `underset("green")(3K_(2)MnO_(4))+2H_(2)Orarrunderset("purple")(2KMnO_(4)^(-))+underset("dark brown")(MnO_(2)DARR)+4KOH` (d) `KMnO_(4)` also acts as an oxidizing agent in alkaline medium. `MnO_(4)^(-)+2H_(2)O+3e^(-)rarrMnO_(2)+4OH^(-),E^(@)=+1.23" volt"` |
|
| 43. |
Pick out the correct statement with respect to [Mn(CN)_6]^(3-) |
|
Answer» It is `dsp^2` hybridised and SQUARE planar |
|
| 44. |
Pick out the correct statement with respect to [Mn(CN)_(6)]^(3-) |
|
Answer» It is `sp^(3)d^(2)`-hybridized and OCTAHEDRAL  In presence of strong field LIGAND, `CN^(-)`, `[Mn(CN)_(6)]^(3-)` =  ltbgt Thus, the COMPLEX is `d^(2)sp^(3)` hybridized having octahedral shape. ltbgt Thus, the COMPLEX is `d^(2)sp^(3)` hybridized having octahedral shape.
|
|
| 45. |
Pick out the correct statement with respect of lyophillic sol |
|
Answer» They are less stable |
|
| 46. |
Pick out the correct statement set for transition metals 1. They form interstitial compounds 2. 5d- elements have higher energies than 3d 4d - elements |
|
Answer» 1, 2 |
|
| 47. |
Pickout the correct statement (s): |
|
Answer» Goldenyellow `PbI_(2)` DISSOLVES in hot water to give is COLOURLESS solution |
|
| 48. |
Pick out the correct statement from the following and choose the correct answer from the codes given below (1) Hex-1,5-diene is a conjugated dience (2) Prop-1,2-diene is a conjugated dience (3)Hexa-1,3-diene is a conjugated dience (4) Buta-1,3, diene is an isolated dience (5) Prop-1,2-diene is a cummulative diene |
|
Answer» 1,2 |
|
| 49. |
Pick out the correct statement regarding resistance of an electrolytic solution |
|
Answer» It is inversely PROPORTIONAL to the length (L) |
|
| 50. |
Pick out the correct statement. |
|
Answer» Both glycine and p - aminobenzoic ACID form DIPOLAR IONS |
|