Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Pick out the correct set of statements for Hl. 1. It reduces iodic acid to I_(2) 2. It reduces H_(2)SO_(4) to SO_(2) 3. It decolourises acidified KMnO_(4) 4. It liberates I_(2) withCuSO_(4) solution |
|
Answer» 1, 2, 3 |
|
| 2. |
Pick out the correct statement 1. The C-Cl bond in Chlorobenzene is shorter than methyl chloride. 2. The C-Cl bond in chlorobenzene has some double bond character 3. The C-Cl bond in chlorobenzene has some double bond character |
|
Answer» only 1, 2 |
|
| 3. |
Pick out the compound/s that gives effervescence with aqueous NaHCO_(3) I. C_(6)H_(5)OH""II.C_(6)H_(5)NH_(2) III. (CH_(3)CO)_(2)O""IV.CH_(3)COOH |
|
Answer» I and II |
|
| 4. |
Pick out the complex compound in which the central metal atom obeys EAN rule strictly? |
|
Answer» `K_(4)[Fe(CN)_(6)]` EAN of `Fe=26-2+6xx2=36`. The above complex strictly obeys EAN RULE. |
|
| 5. |
Pick out the alloy that contains a non-metal as constituent in it |
|
Answer» Brass |
|
| 6. |
Pick out the combination which show positive deviations from Raoult.s law: |
|
Answer» `C_2H_5OH + C Cl_4` |
|
| 7. |
Pick out the alkane which differs from the other members of the group. |
|
Answer» 2,2-dimethyl propane 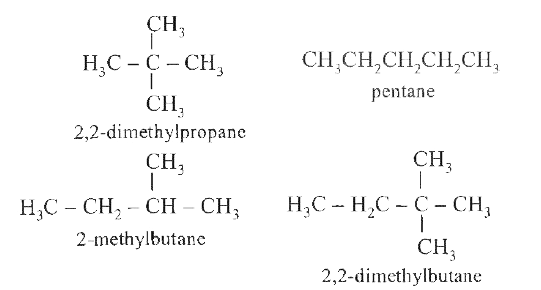
|
|
| 8. |
Pick out incorrect statemment about K_(2) Cr_(2)O_(7). |
|
Answer» It oxidazes KI to `I_(2)` |
|
| 9. |
Pick out incorrect statement with respect to methyl nitrite |
|
Answer» They are ALSO CALLED PSEUDO acid |
|
| 10. |
Pick out incorrect statement for H_2SO_3 |
|
Answer» But-1-ene when HYDRATED in presence of conc. `H_2SO_4`, form butan-2-ol `Cu+2H_(2)SO_(4) to CuSO_(4)+2H_(2)O+2SO_(2)` (a). `CH_(3)-CH_(2)-CH=CH_(2)+H_(2)SO_(4) to CH_(3)-CH_(2) underset(OSO_(3)H)underset(|)(CH)-CH_(3) overset(H_(2)O)to CH_(3)-CH_(2)-underset(OH)underset(|)(CH)-CH_(3)+H_(2)SO_(4)` (b). `C+underset(("Conc.""))(2H_(2)SO_(4))to 2H_(2)O+CO_(2)+2SO_(2)` `(b) S+2H_(2)SO_(4) to 2H_(2)O+3SO_(2)` `(d) underset(("98% )acid")) (Zn+H_(2)SO_(4)) to ZnSO_(4)+2H_(2)O+SO_(4)` |
|
| 11. |
Pick out incorrect statement regarding ethers |
|
Answer» Ethers have HIGHER boiling points than alkanes of COMPANIES mass. |
|
| 12. |
Pick out incorrect statement |
|
Answer» Colloidal sols are heterogeneous |
|
| 13. |
Pick out incorrect statement. |
|
Answer» `NF_3` molecules has trigonal pyramidal structure a) From the above figure, it is clear that `NF_(3)`, has a trigonal pyramidal structure. B). `N_(2)O_(3)(l)+H_(2)O(l) to underset("Acid")(2HNO_(2)(l))` 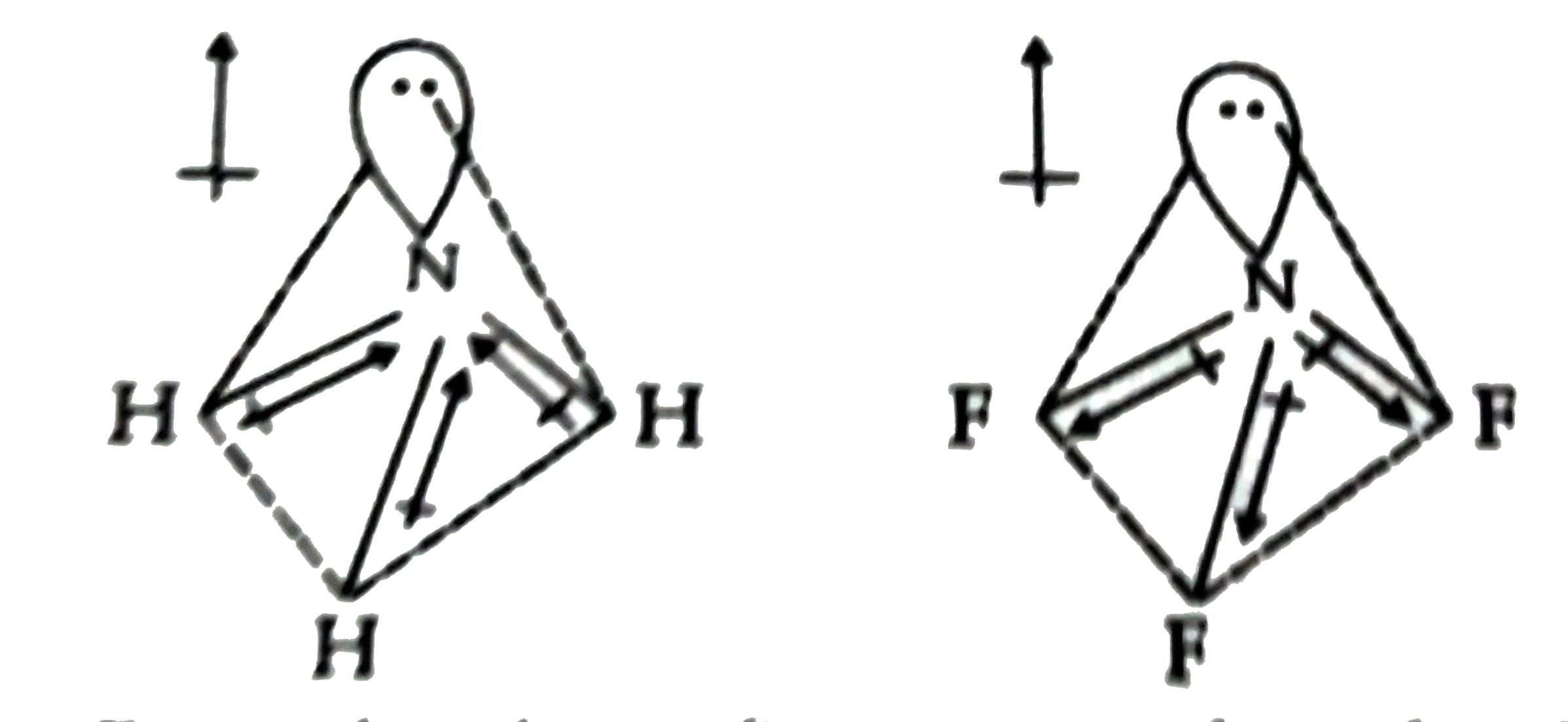
|
|
| 14. |
Pick out an incorrect statement from the following: |
|
Answer» Benzene cannot be iodinated with `l_(2)` directly |
|
| 15. |
Pick a poor electrolytic conductor complex in solution: |
|
Answer» `K_2[PtCI_6]` |
|
| 16. |
Pich out the statement which is not relevant in the discussion of colloids |
|
Answer» SODIUM aluminium silicate is used in the softening of hard water |
|
| 17. |
pi-bonding is not involved in : |
|
Answer» FERROCENE |
|
| 18. |
Phytol is |
|
Answer» oils |
|
| 19. |
Physisorption is reversible while chemisorption is irreversible. Why? |
| Answer» Solution : Physisorption INVOLVES weak forces between the ADSORBENT and the adsorbate while chemisorption involves the FORMATION of a chemical bond between the adsorbent and adsorbate. Once a chemical bond is formed, it is difficult to break. Therefore physisorption is REVERSIBLE but chemisorption is irreversible | |
| 20. |
Physisorption and chemisorption are two types of adsorption. What is the effect of temperature on physisorption and chemisorption? |
|
Answer» |
|
| 21. |
Physical properties of : |
|
Answer» ALCOHOLS LIE between ALKANES and `H_2O` |
|
| 22. |
Physical and chemical adsorption respond differently to rise in temperature ? What is this difference and why is it so ? |
| Answer» | |
| 23. |
Physical adsorption of a gasous species may change to chemical adsorption with ___. |
|
Answer» DECREASE in temperature |
|
| 24. |
Physical adsorption of a gaseous species may change to chemical adsorption with |
|
Answer» DECREASE in temperature |
|
| 25. |
Physical adsorption of a gaseous species may change to chemical adsorption with ________ . |
|
Answer» decrease in temperature |
|
| 26. |
Physical adsorption of a gaseous species may change to chemical adsorption with ....... |
|
Answer» decrease in temperature |
|
| 27. |
Physical adsorption of a gaseous species may change to chemical adsorption with………… |
|
Answer» DECREASE in temperature |
|
| 28. |
Physical adsorption is inversly proportional to .... |
|
Answer» volume |
|
| 29. |
Physical adsorption of a gaseous speciesmay change to chemical adsorption with ……. . |
|
Answer» decrease in TEMPERATURE |
|
| 30. |
Physical adsorption of a gaseous species may change to chemical adsorption with ____________ |
|
Answer» decrease in temperature |
|
| 31. |
Physical adsorption is appreciable at |
|
Answer» Higher TEMPERATURE |
|
| 32. |
Physical adsorption is considerable at |
|
Answer» LOW TEMPERATURE |
|
| 33. |
Physical adsorption is: |
|
Answer» HIGHLY SPECIFIC |
|
| 34. |
Physical adsorption is.... |
|
Answer» INSTANTANEOUS and REVERSIBLE. |
|
| 35. |
Physical adsorption is : |
|
Answer» HIGHLY specific |
|
| 36. |
Physical adsorption increases when : |
|
Answer» TEMPERATURE increases |
|
| 37. |
Phthalic acid +NH_(3)toDoverset(Delta)toE |
Answer» 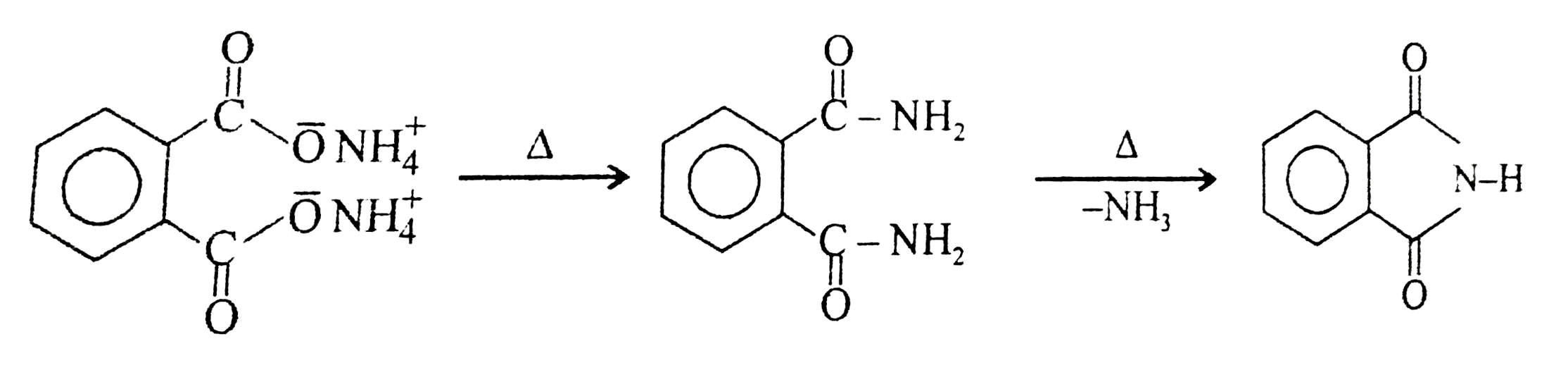
|
|
| 38. |
Physical adsorption become appreciable at : |
|
Answer» HIGH temperature |
|
| 39. |
Phthalic acid reacts with resorcinol in the presence of concentrated H_(2)SO_(4) to give: |
|
Answer» Phenolphthalein 
|
|
| 40. |
Physical adsorption: |
|
Answer» Is reversible |
|
| 41. |
Photosysthesis si a bio process by which plants make energy rich molecules from low energy molecules with the help of energy from sunlight . The photosysthesis of glucose can be represented as: 6CO_(2(g))+ 6H_(2)O_(g) + hv rarr C_(5)H_(12)O_(6(s)) + 6O_(2)(g)..........(i) The energy of one mole of a photon of wave lenght is known as one Einstein. A weight lifter lifts a weight of 160Kg through of 2.4 m . Assuming all the energy required for this task is obtained by the combustion of glucose , calculate the change in the current produced by a sample of blood of the weight lifter . Same volume of blood (5mL) is tested both before and after lifting the weight and the total volume of blood in his body is 5L. (1 g of glucose releases 15.58 KJ of energy ) |
|
Answer» |
|
| 42. |
Phthaldehyde has |
|
Answer» no CARBOXYLIC group |
|
| 43. |
Photosynthesis in plants is brought about by chlorophyll. It involves : |
|
Answer» CONVERSION of CHEMICAL energy into radiant energy |
|
| 44. |
Photon having energy equivalent to the binding energy of 4th state of He^(+) atom is used to eject an electron from the metal surface of work function 1.4 eV. If electrons are further accelerated through the potential difference of 4V then the minimum value of De-brogile wavelength associated with the electron is: |
|
Answer» `:. 2pir=5lambda …(1)` However, path length in a sate `n` is `n` times the de-Broglie wavelength. `:. 2pir=nlambda …(2)` From (1) & (2), principal quantum number (n) of the excited state `=5`. PHOTON having `2^(nd)` highest energy corresponds to back transition of electron from `n=4` to `n=1` This photon will cause an already excited `Li^(2+)` electron to go some higher state. Let the initial excited state of `Li^(2+)` ION be `n_(1)` and final excited state of `Li^(2+)` ion be `n_(2)` `:. (13.6(1)^(2)[1/1^(2)-1/4^(2)])/(("Photon having" 2^(nd) "highest energy"),("corresponding to transition n=4 to n=1 in H-atom"))=(13.6(3)^(2)[1/n_(1)^(2)-1/n_(2)^(2)])/(("Energy ABSORBED by" Li^(+) "ion to make"),("a transition from" n_(1) "to" n_(2)))` `:. 13.6[1/1^(2)-1/4^(2)]=13.6[3^(2)/n_(1)^(2)-3^(2)/n_(2)^(2)]` or `13.6[1/1^(2)-1/4^(2)]=13.6[1/((n_(1)//3)^(2))-1/((n_(2)//3)^(2))]` On cmparing both sides, `n_(1)/3=1` & `n_(2)/3=4rArr n_(1)=3` & `n_(2)=12` Thus, the final excited state of `Li^(2+)` ion electron is `n=12` Ans. |
|
| 45. |
Photographic plates and films are prepared by coating an emulsion of light sensitive substance |
|
Answer» POTASSIUM BROMIDE. |
|
| 46. |
Photographic plates and films have an essential ingredient of |
|
Answer» silver nitrate |
|
| 47. |
Photographic paper is developed with alkaline hydroquinone. selectthe correctstatements |
|
Answer» Hydroquinone is the OXIDANT. |
|
| 48. |
Photographic films or plates have …… as an essential ingredient. |
|
Answer» SILVER Oxide |
|
| 49. |
Photographic films or plates have ……… as an essential ingredient: |
|
Answer» SILVER bromide |
|