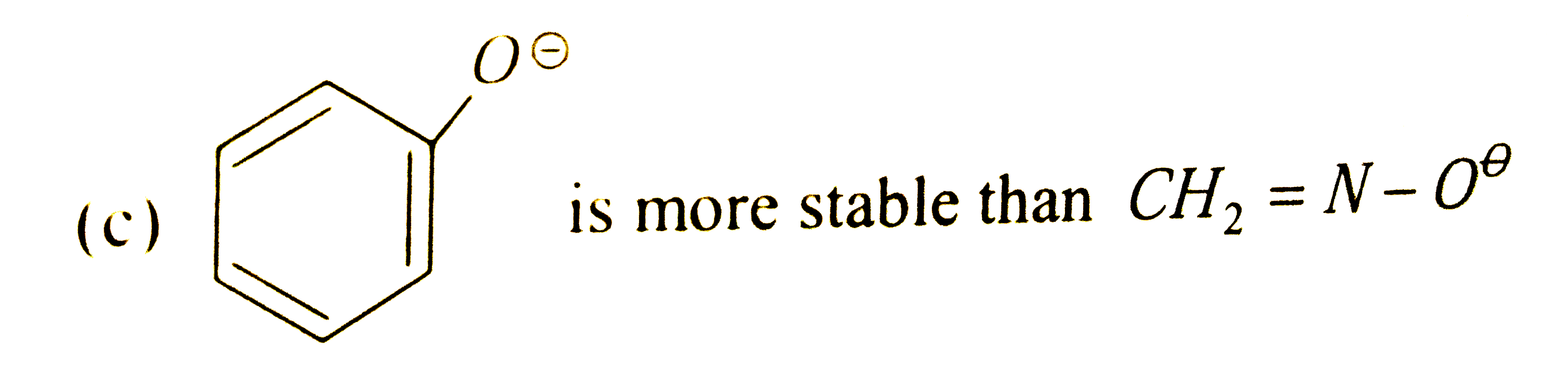Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Phenylmethanol can be prepared by reducing the benzaldehyde with : |
|
Answer» `CH_(3)Br` |
|
| 2. |
Phenylhydrazine can be used to test for |
|
Answer» CARBOXYL group. |
|
| 3. |
Phenyl methyl ether reacts with HI to give phenol and methyl iodide and not iodobenzene and methl alcohol. Why? |
Answer» Solution :Protonation of phenyl methyl ether (anisole) gives oxononium ion. In this ion, `O-CH_(3)` bond is weaker than `O-C_(6)H_(5)` bond which has some double bond character DUE to resonance between the lone pair of electrons on the oxygen atom and the `SP^(2)`-HYBRIDIZED carbon atom of the phenyl ring. therefore, ATTACK by `I^(-)` ion exclusively BREAKS the weaker `O-CH_(3)` bond forming methyl iodide and phenol. 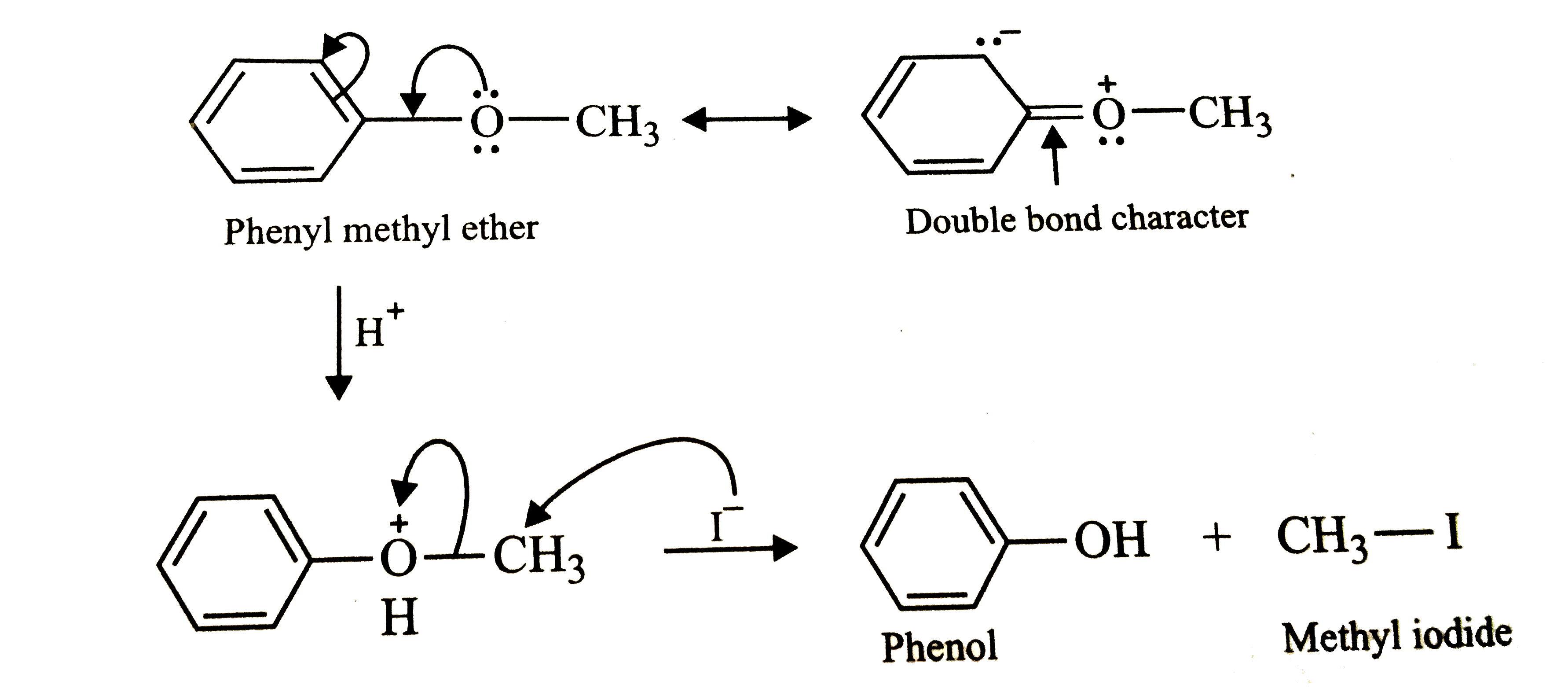
|
|
| 4. |
Phenylacetylene on treatment with HgSO_(4)//H_(2)SO_(4), H_(2)O produces |
|
Answer» acetophenone |
|
| 6. |
Phenyl methanal is reacted with concentrated NaOH to give two products X and Y. X reacts with metallic sodium to liberate hydrogen X and Y are |
|
Answer» sodiumbenzoate and phenol 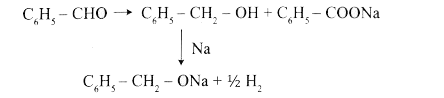
|
|
| 7. |
Phenyl isocyanides are prepared from which of the following reactions ? |
|
Answer» Rosenmund's REACTION 
|
|
| 8. |
Phenyl isocynanides are preapred from which of the following reactions |
|
Answer» ROSENMUND's REACTION 
|
|
| 9. |
Phenyl isocyanides are prepared from which of the following reaction? |
|
Answer» ROSENMUND's REACTION |
|
| 10. |
Phenyl isocynaide is prepared from aniline by: |
|
Answer» Rosenmud's REACTION |
|
| 11. |
Phenyl isocyanides are prepared from which of the following reaction ? |
|
Answer» Rosenmund's reaction `C_(6)H_(5)NH_(2)+CHCl_(3)+underset((alc.))(3KOH)to underset("PHENYL ISOCYANIDE")(C_(6)H_(5)NC)+3KCl+3H_(2)O` |
|
| 12. |
Phenyl isocyanides are prepared by |
|
Answer» ROSENMUND's REACTION |
|
| 13. |
Phenyl isocyanide on reduction with hydrogen and Raney nickel gives____ |
| Answer» SOLUTION :N-methylaniline (N-methylbenzenamine). | |
| 14. |
Phenyl isocyanide is prepared by heating CHCl_(3) with : |
|
Answer»
|
|
| 15. |
Phenyl ethane on oxidation produces |
|
Answer» <P>phthalic acid |
|
| 16. |
Phentole reacts with HI to give |
|
Answer» iodobezene and ethanol |
|
| 17. |
Phenoxide ion is morestable than phenol due to the _______. |
|
Answer» resonating structureof benzene RING |
|
| 18. |
Phenolunderset(dust)overset(Zn)toX underset("Anhyd. "AlCl_(3))overset(CH_(3)Cl)toY underset(KMnO_(4))overset(Alk.)toZ The product Z is |
|
Answer» benzaldehyde |
|
| 19. |
PhenolundersetDeltaoverset"Zn"to(A)underset(conc. H_2SO_4 "at" 27^@C)overset(conc. HNO_3)to (B)overset(Fe+HCl)to(C ) In above reaction ,compound A,B, C are |
|
Answer» BENZENE, benzene sulphoric ACID, aniline |
|
| 20. |
"Phenol"underset("dust")overset(Zn)to'X'underset("anhy"AlCl_(3))overset(CH_(3)Cl)toYunderset(KMnO_(4))overset("alk")toZ the product Z is _________. |
|
Answer» benzaldehyde |
|
| 21. |
Phenols turn blue litmus red |
|
Answer» |
|
| 22. |
Phenols on treatment with chloroform in the presence of aqueous sodium hydroxide or potassium hydroxide solution give hydroxy aldehydes. In this reaction, the formyl group is directed to ortho position. But if one of the ortho position is occupied then para product is formed. The reaction is electrophilic substitution reaction. What is the name and the structure of the final product formed. |
Answer» SOLUTION :
|
|
| 23. |
Phenols on treatment with chloroform in the presence of aqueous sodium hydroxide or potassium hydroxide solution give hydroxy aldehydes. In this reaction, the formyl group is directed to ortho position. But if one of the ortho position is occupied then para product is formed. The reaction is electrophilic substitution reaction. What is the intermediate of the reaction if we use carbon tetrachloride in place of chloroform? |
Answer» SOLUTION :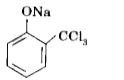
|
|
| 24. |
Phenols on treatment with chloroform in the presence of aqueous sodium hydroxide or potassium hydroxide solution give hydroxy aldehydes. In this reaction, the formyl group is directed to ortho position. But if one of the ortho position is occupied then para product is formed. The reaction is electrophilic substitution reaction. Write the reaction with p cresol. |
Answer» SOLUTION :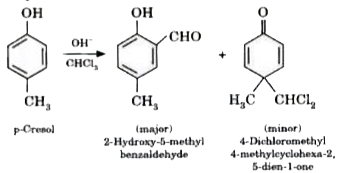
|
|
| 25. |
Phenols on treatment with chloroform in the presence of aqueous sodium hydroxide or potassium hydroxide solution give hydroxy aldehydes. In this reaction, the formyl group is directed to ortho position. But if one of the ortho position is occupied then para product is formed. The reaction is electrophilic substitution reaction. What is the electrophile in the reaction? |
| Answer» SOLUTION :DICHLOROCARBENE `C Cl_(2)` | |
| 26. |
Phenols on treatment with chloroform in the presence of aqueous sodium hydroxide or potassium hydroxide solution give hydroxy aldehydes. In this reaction, the formyl group is directed to ortho position. But if one of the ortho position is occupied then para product is formed. The reaction is electrophilic substitution reaction. What is the name of the reaction? |
| Answer» SOLUTION : Reimer-Tiemann REACTION. | |
| 27. |
Phenols on exposureto light and airgetsoxidised to ________. |
|
Answer» QUININES |
|
| 29. |
Phenols are more acidic than aliphatic alcohols because |
|
Answer» phenoxide ION is STABILISED by resonance |
|
| 30. |
Phenols does not protonation reactions readily-Give reason. |
| Answer» Solution :In Phenol, the lone PAIR of electron on the oxygen ATOM are delocalised due to RESONANCE, HENCE are not easily available for protonation. | |
| 31. |
Phenols are more acidic than alcohols. Write the structure and IUPAC name of the above product. |
| Answer» SOLUTION :2,4,6-Trinitrophenol | |
| 32. |
Phenols are more acidic than alcohols. Name the product obtained when phenol is treated with chloroform in the presence of NaOH. |
| Answer» SOLUTION :SALICYLALDEHYDE | |
| 33. |
Phenols are more acidic than alcohols. Name the above reaction. |
| Answer» SOLUTION :Reimer-Teimann REACTION | |
| 34. |
Phenols are more acidic than alcohols. Ethanol and propane have comparable molecular masses but boiling points differ widely. Which of them has highes boiling point? Substantiate your answer. |
| Answer» Solution :Ethanol has HIGHER boiling point because its molecules are associated by INTERMOLECULAR HYDROGEN BONDING. | |
| 35. |
Phenols are more acidic than alcohols due to (a)In phenols,- OH is attached to sp^(2)hybridised carbon but in alcohols , -OH is attached to sp^(3)hybridised carbon (b) Phenoxide ion is more stable than alkoxide due to resonance (c)Phenoxide ion is more stable than phenol |
|
Answer» only a |
|
| 36. |
Phenols are converted into their salts by aqueous NaOH but not by aqueous bicarbonates. The salts are converted into the free phenols by aqueous mineral acids, carboxylic acid or carbonic acids. Most phenols have K_(a) value of about 10^(-10), and are tremondously more acidic than alcohols. The difference in acidity are due to difference in stablities of reactants and products. Phenol and phenoxide ions contain benzene ring and therefore must be hybrid of Kekuley structures Being basic, oxygen can share more than a pair of electron with the ring. Since energy must be supplied to separate opposite charge, the structure of phenols should contain more energy. The net effect of reasonance is therefore to stablise the phenoxideion to a greater extent than the phenol and thus to shift the equilibrium towards ionization and make K_(a) larger than for an alcohol. Choose the correct statement: |
|
Answer» Phenol GIVES effervesence with `NaHCO_(3)` |
|
| 37. |
Phenols are converted into their salts by aqueous NaOH but not by aqueous bicarbonates. The salts are converted into the free phenols by aqueous mineral acids, carboxylic acid or carbonic acids. Most phenols have K_(a) value of about 10^(-10), and are tremondously more acidic than alcohols. The difference in acidity are due to difference in stablities of reactants and products. Phenol and phenoxide ions contain benzene ring and therefore must be hybrid of Kekuley structures Being basic, oxygen can share more than a pair of electron with the ring. Since energy must be supplied to separate opposite charge, the structure of phenols should contain more energy. The net effect of reasonance is therefore to stablise the phenoxideion to a greater extent than the phenol and thus to shift the equilibrium towards ionization and make K_(a) larger than for an alcohol. Which of the following is more stable: |
|
Answer»
|
|
| 38. |
Phenols are converted into their salts by aqueous NaOH but not by aqueous bicarbonates. The salts are converted into the free phenols by aqueous mineral acids, carboxylic acid or carbonic acids. Most phenols have K_(a) value of about 10^(-10), and are tremondously more acidic than alcohols. The difference in acidity are due to difference in stablities of reactants and products. Phenol and phenoxide ions contain benzene ring and therefore must be hybrid of Kekuley structures Being basic, oxygen can share more than a pair of electron with the ring. Since energy must be supplied to separate opposite charge, the structure of phenols should contain more energy. The net effect of reasonance is therefore to stablise the phenoxideion to a greater extent than the phenol and thus to shift the equilibrium towards ionization and make K_(a) larger than for an alcohol. Consider the following curves: |
|
Answer» CURVE A represents the ionisation of ALCOHOL |
|
| 39. |
Phenols are converted into their salts by aqueous NaOH but not by aqueous bicarbonates. The salts are converted into the free phenols by aqueous mineral acids, carboxylic acid or carbonic acids. Most phenols have K_(a) value of about 10^(-10), and are tremondously more acidic than alcohols. The difference in acidity are due to difference in stablities of reactants and products. Phenol and phenoxide ions contain benzene ring and therefore must be hybrid of Kekuley structures Being basic, oxygen can share more than a pair of electron with the ring. Since energy must be supplied to separate opposite charge, the structure of phenols should contain more energy. The net effect of reasonance is therefore to stablise the phenoxideion to a greater extent than the phenol and thus to shift the equilibrium towards ionization and make K_(a) larger than for an alcohol. Which of the following is strongest acid? |
|
Answer» `H_(2)CO_(3)` |
|
| 40. |
Phenols are more acidic than alcohols and CH_(3)NO_(2) because |
|
Answer»
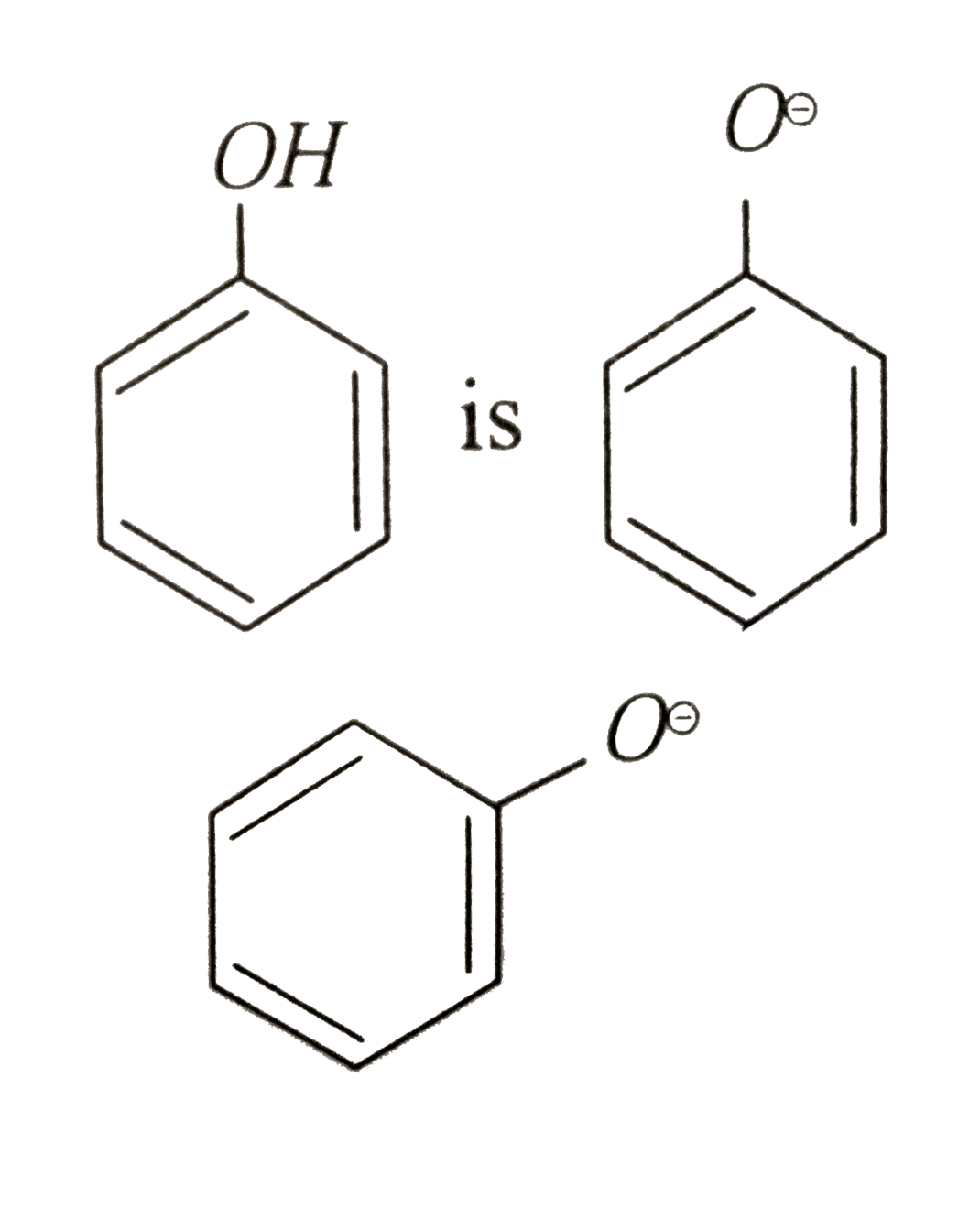 isresonance stabilized , while in `R-O^(Ө)` DUE to +I effect the `R-O^(Ө)` is destabilized . The conjugate base of `CH_(3)NO_(2)` is `overset(Ө)CH_(2)-NO_(2)` which is RESONANCE sbabilized 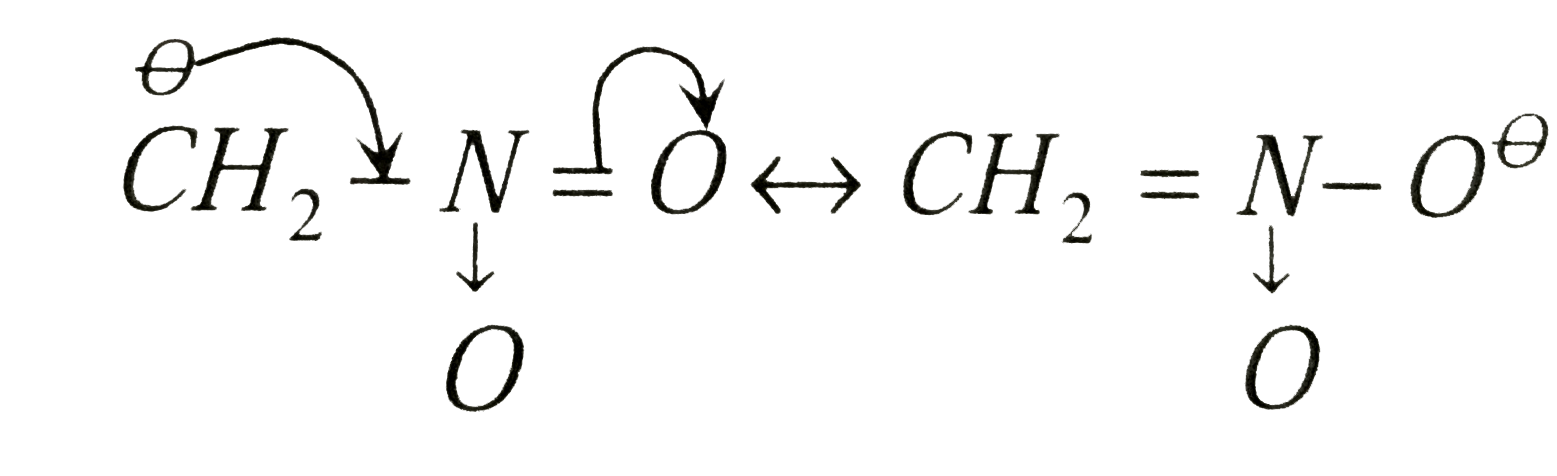 out of `-underset(..)overset(..)O-H` group whose conjugate base is `-O^(Ө)` and `-underset(|)overset(|)C^(Ө)` because the charge density is less in `-O^(Ө)` than in`-underset(|)overset(|)C^(Ө)` due to greater surface volume available on `-O^(Ө)` than in `-underset(|)overset(|)C^(Ө)` 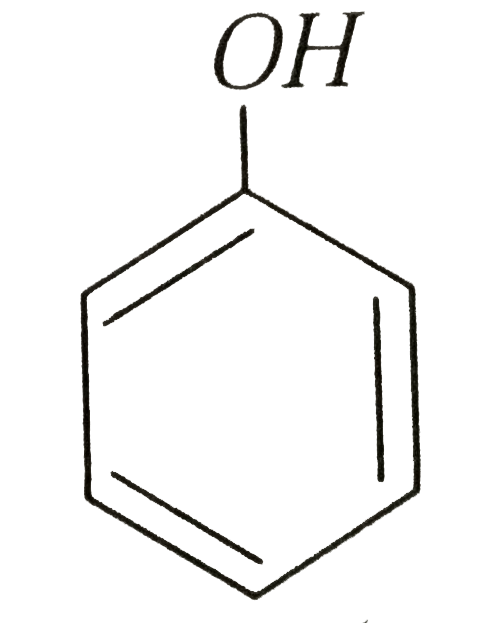 is more acidic than ROH and `CH_(3)NO_(2)` . Hence choices (a),(b) and (d) are correct while (c) is incorrect. is more acidic than ROH and `CH_(3)NO_(2)` . Hence choices (a),(b) and (d) are correct while (c) is incorrect.
|
|
| 41. |
Phenols and alcohols have both the same functional group (-OH) but phenols are stronger acids than alcohols. This is mainly due to the reason that the phenoxide ion (C_(6)H_(5)O^(Ɵ)) left after the release of H^(+) ion in phenol is resonance stabilised while the alkoxide ion (RO^(Ɵ)) in alcohols does not show similar characteristics. The electron withdrawing groups tend to increase the acidic strengths of phenols while electron releasing groups tend to decrease it. the effect of both types of groups is more pronounced when present at the para position than when these are present at the ortho position in the ring. However, their effect at the meta position is relatively very small. Which of the following is the strongest acid ? |
|
Answer» `FCH_(2)CH_(2)CH_(2)CH_(2)OH` |
|
| 42. |
Phenols and alcohols have both the same functional group (-OH) but phenols are stronger acids than alcohols. This is mainly due to the reason that the phenoxide ion (C_(6)H_(5)O^(Ɵ)) left after the release of H^(+) ion in phenol is resonance stabilised while the alkoxide ion (RO^(Ɵ)) in alcohols does not show similar characteristics. The electron withdrawing groups tend to increase the acidic strengths of phenols while electron releasing groups tend to decrease it. the effect of both types of groups is more pronounced when present at the para position than when these are present at the ortho position in the ring. However, their effect at the meta position is relatively very small. The stronger acid among the following is |
|
Answer» o-methoxyphenol |
|
| 43. |
Phenols and alcohols have both the same functional group (-OH) but phenols are stronger acids than alcohols. This is mainly due to the reason that the phenoxide ion (C_(6)H_(5)O^(Ɵ)) left after the release of H^(+) ion in phenol is resonance stabilised while the alkoxide ion (RO^(Ɵ)) in alcohols does not show similar characteristics. The electron withdrawing groups tend to increase the acidic strengths of phenols while electron releasing groups tend to decrease it. the effect of both types of groups is more pronounced when present at the para position than when these are present at the ortho position in the ring. However, their effect at the meta position is relatively very small. The acidic strength increases in the order |
|
Answer» p-Nitrophenol `lt` m-Nitrophenol `lt` o-Nitophenol |
|
| 44. |
Phenols and alcohols have both the same functional group (-OH) but phenols are stronger acids than alcohols. This is mainly due to the reason that the phenoxide ion (C_(6)H_(5)O^(Ɵ)) left after the release of H^(+) ion in phenol is resonance stabilised while the alkoxide ion (RO^(Ɵ)) in alcohols does not show similar characteristics. The electron withdrawing groups tend to increase the acidic strengths of phenols while electron releasing groups tend to decrease it. the effect of both types of groups is more pronounced when present at the para position than when these are present at the ortho position in the ring. However, their effect at the meta position is relatively very small. The increasing order of reactivity of 1^(@), 2^(@) and 3^(@) alcohols towards sodium metal is |
|
Answer» `1^(@) lt 2^(@) lt 3^(@)` |
|
| 45. |
Phenols and alcohols are respectively ________ |
|
Answer» NEUTRAL and BASIC |
|
| 46. |
Phenolphthalein shows ____ in acid medium: |
| Answer» Answer :D | |
| 47. |
Phenolphthalein is not suitable for the titration of |
|
Answer» `NaOH` vs `(COOH)_(2)` |
|
| 48. |
Phenolphthalein is most suitable indicator for the titration of |
|
Answer» `CH_(3)COOH and NH_(4)OH` |
|
| 49. |
Phenolphthalein is not a good indicator for titrating: |
|
Answer» NAOH against OXALIC ACID |
|
| 50. |
Phenol upon distiliation with zinc dust gives : |
|
Answer» Benzene |
|




_S01_401_O04.png)
_S01_399_O01.png)
_S01_399_O02.png)
_S01_397_O02.png)
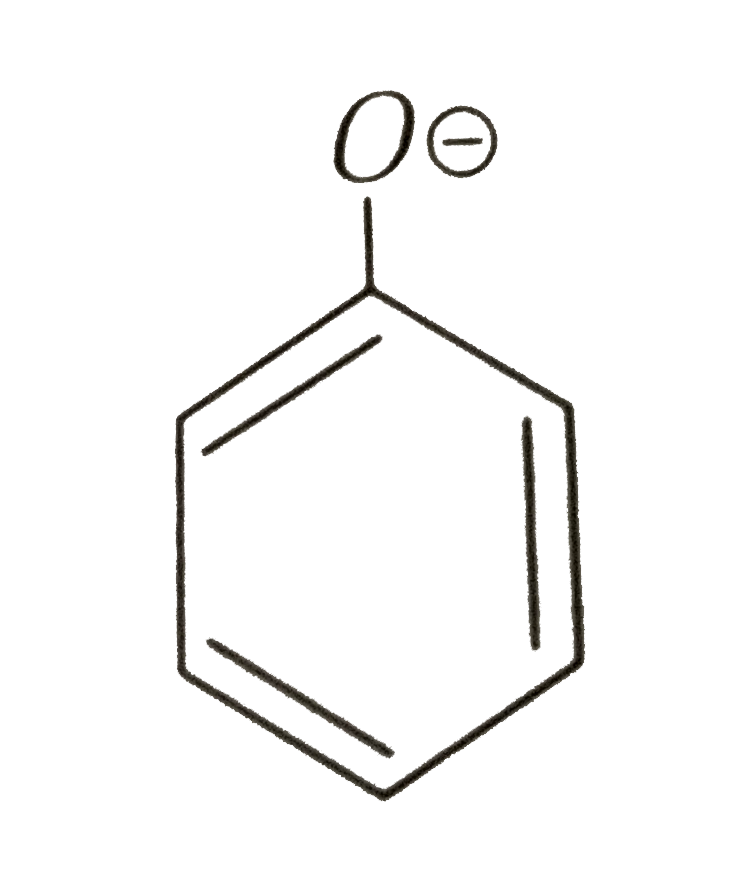
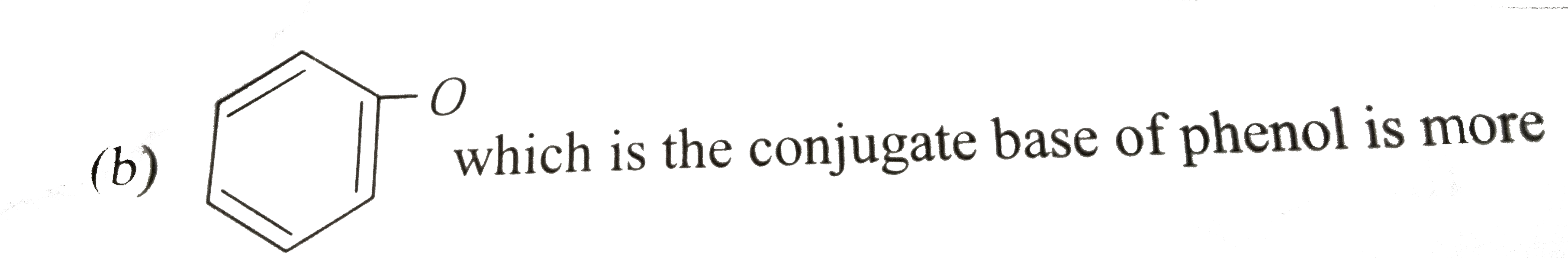 which is the conjugate base of phenol is more stable than `Coverset(Ө)H_(2)-NO_(2)`
which is the conjugate base of phenol is more stable than `Coverset(Ө)H_(2)-NO_(2)`