Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Phenolphthalein does not act as indicator for the titration between : |
|
Answer» KOH and `H_2SO_4` |
|
| 2. |
Phenolphthalein is a: |
|
Answer» WEAK acid |
|
| 3. |
Phenolphthaleis not a good indicator for titrating : |
|
Answer» NaOH against oxalic acid |
|
| 4. |
Phenolphthalein does not act as an indicator for the titration between |
|
Answer» NAOH and `CH_(3)COOH` |
|
| 5. |
Phenolic antibacterial used in body deodorants is |
|
Answer» 2,4-dichlorophenol |
|
| 6. |
Phenolic antibacterial used in body deodrants is |
|
Answer» 2, 4-Dichlorophenol |
|
| 7. |
Phenols are more acidic than alcohols. What is the product obtained when phenol is treated with conc.HNO_3? |
|
Answer» o-Nitrophenol |
|
| 8. |
Phenol, when it first reacts with concentrated sulphuric acid and then with concentrated nitric acid, gives |
|
Answer» 2,4,6-trinitrobenzene. |
|
| 9. |
Phenol , when first reacts with concentrated sulphuric acid and then when concentrated nitric acid, gives |
|
Answer» o-nitrophenol |
|
| 10. |
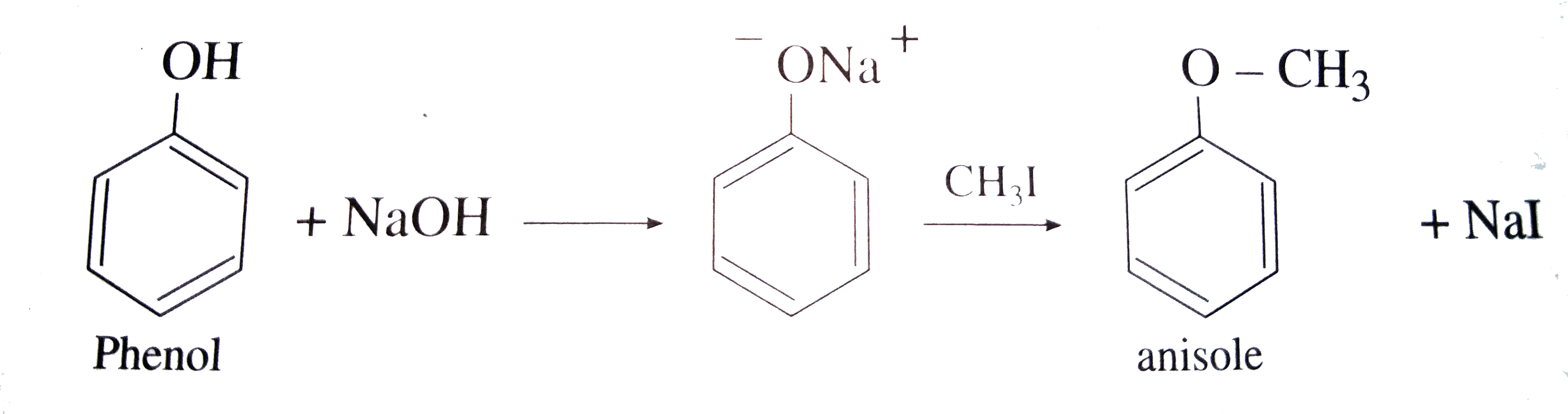
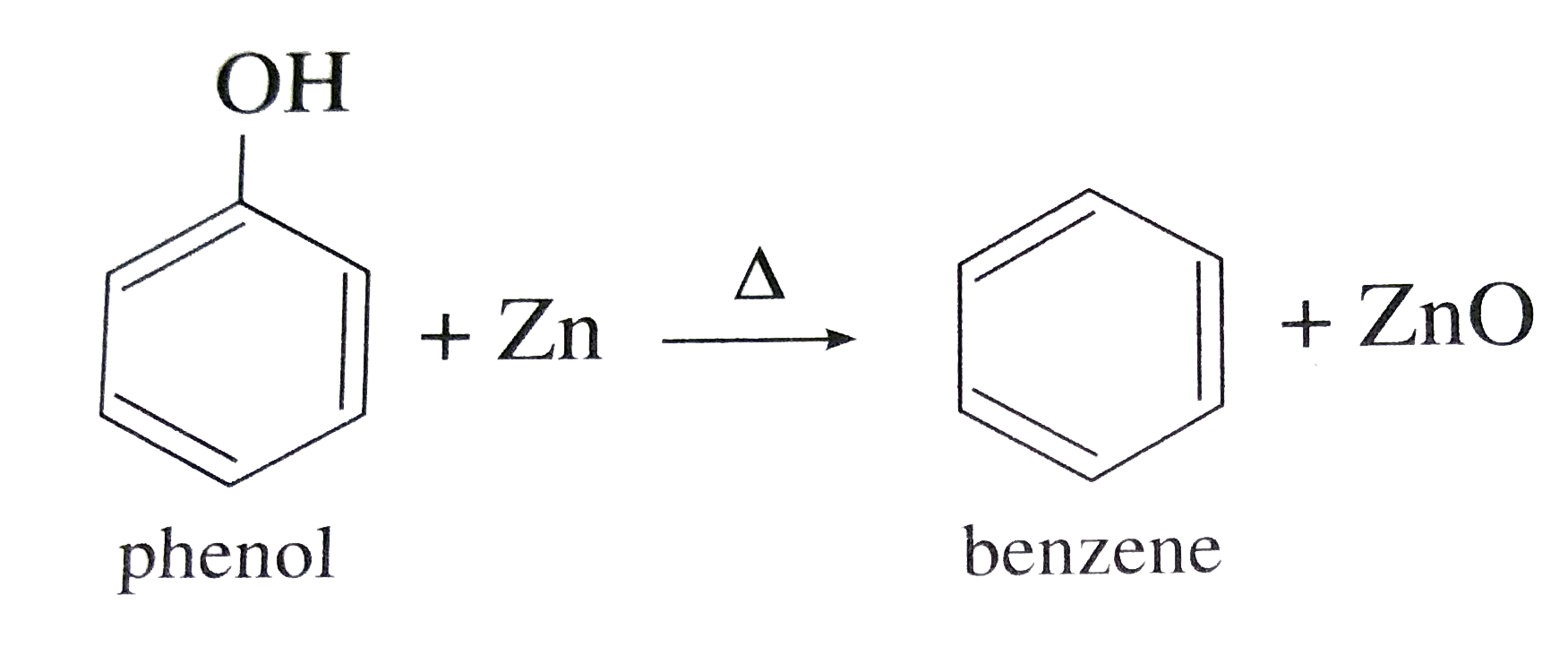
Write the following reactions : Phenol with zinc dust. |
|
Answer» Benzene |
|
| 11. |
"Phenol" underset("Powder")overset(Zn)to X underset("Anhy." AlCl_(3))to Y underset(KMnO_(4))overset("Alkaline")to Z What is Z? |
|
Answer» Benzaldehyde 
|
|
| 12. |
Phenol underset(Delta)overset(Zn)toX.The compound X on acylation gives aliphatic aromatic ketone. The reaction is |
|
Answer» Gattermann REACTION 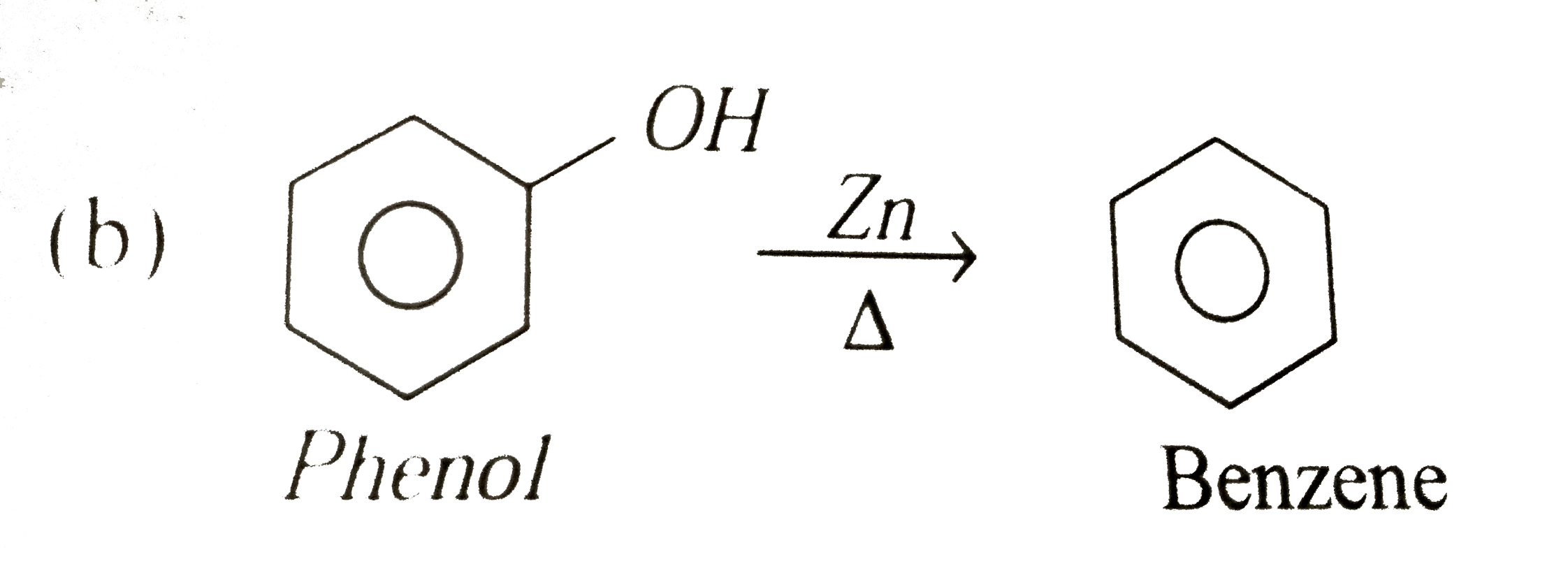 In Friedal craft's reaction ACETYL chloride reacts with benzene in the presence of ANHYDROUS aluminium trichloride to form acetophenone. 
|
|
| 13. |
Phenol underset("distillation")overset(Zn)to A underset(Conc. HNO_(3))overset(Conc. H_(2)SO_(4))to B underset(NaOH)overset(Zn)toC In the above reaction A, B and C are the following compounds |
|
Answer» `C_(6)H_(6), C_(6)H_(5)NO_(2)` and ANILINE 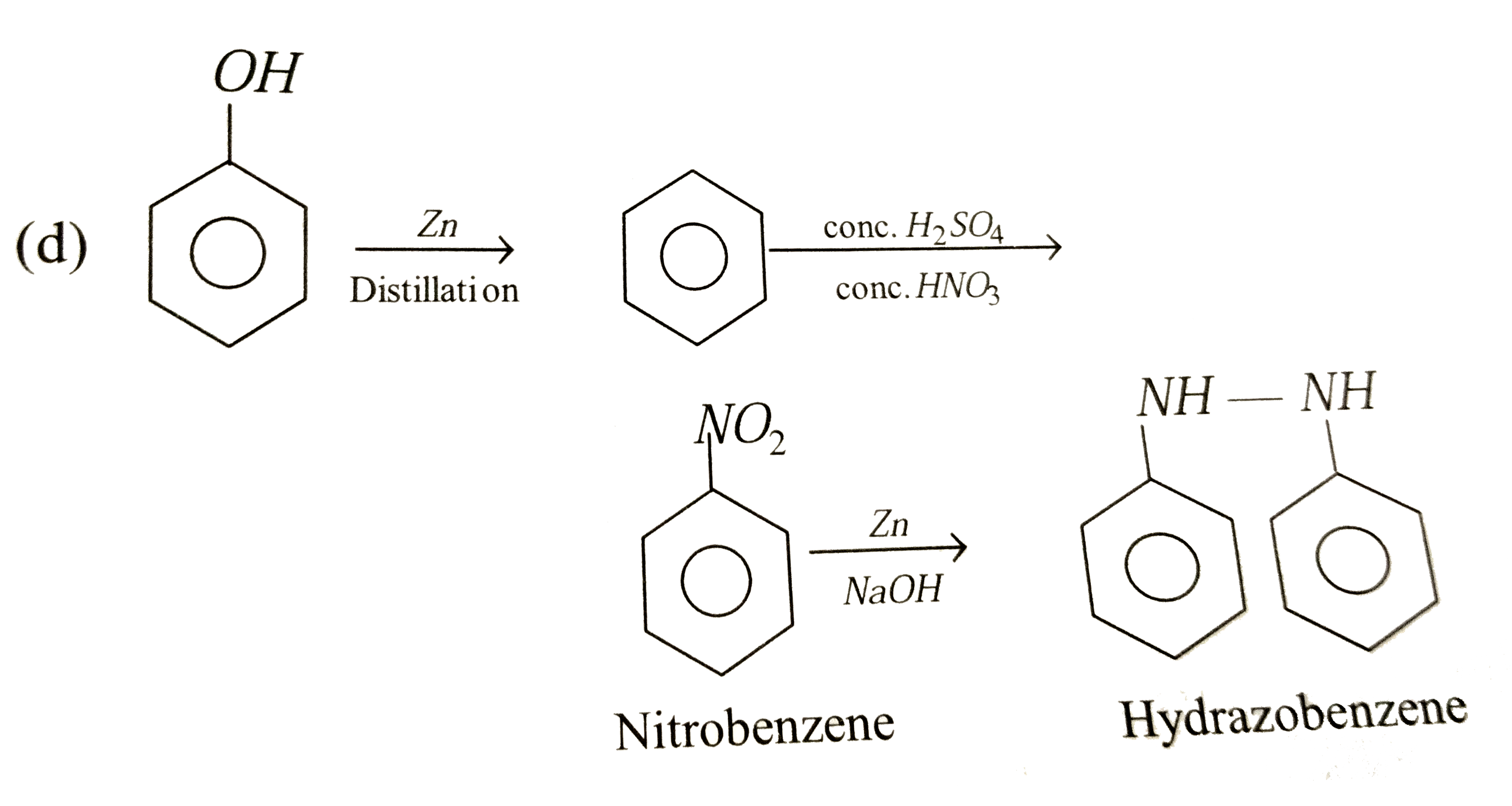
|
|
| 14. |
Phenol treated with excess of bromine water gives _____. |
|
Answer» |
|
| 15. |
Benzene underset("anhydrous" AICI_(3))overset(CH_(3)CI)(to) X overset("alkaline" KMnO_(4))(to) Y overset("alkaline" KMnO_(4))(to) Z, The product Z is |
|
Answer» Benzaldehyde |
|
| 17. |
How would you convert the following : (i)Phenol to benzoquinone (ii) Propanone to 2-methylpropan -2 ol (iii) Propene to propan -2 - ol |
Answer» SOLUTION :PHENOL to BENZOQUINONE : 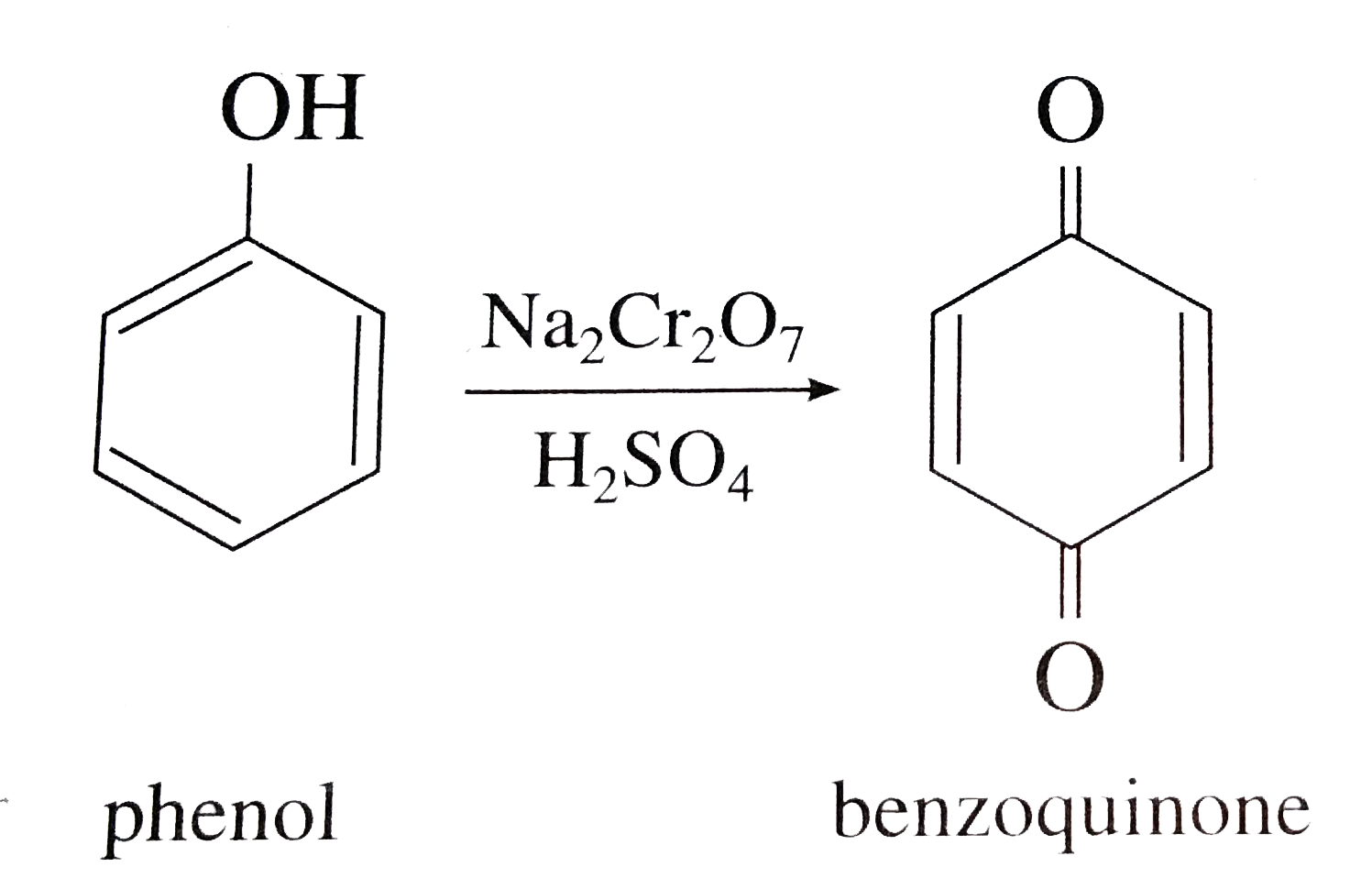
|
|
| 18. |
How will you convert : (i) Chlorobenzene to phenol (ii) Phenol to salicylic acid ? |
Answer» SOLUTION :PHENOL to SALICYLIC ACID : 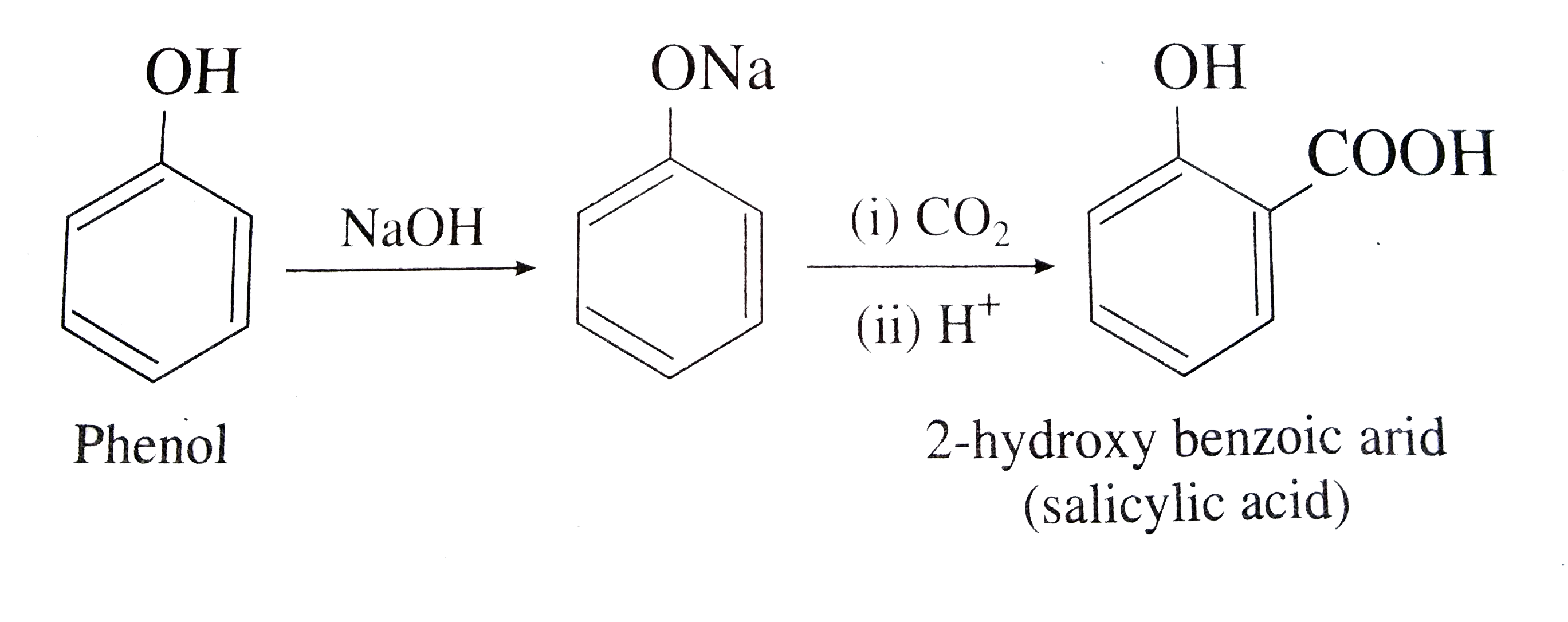
|
|
| 21. |
Phenol reacts with which one of the following reagents gives a conjugate diketone will be formed ? |
|
Answer» `Na_(2)Cr_(2)O_(7)` |
|
| 22. |
Phenol reacts with methyl chloroform in the presence of NaOH to form product A. A reacts with Br_(2) to form product B . A and B are respectively- |
|
Answer»
|
|
| 23. |
Phenol reacts with NH_(3) to give |
|
Answer» benzamide 
|
|
| 24. |
Phenol reacts with methyl chloroformate in the presence of NaOH to form product A. A reacts with Br_(2) to form product B. A and B are respectively |
|
Answer»
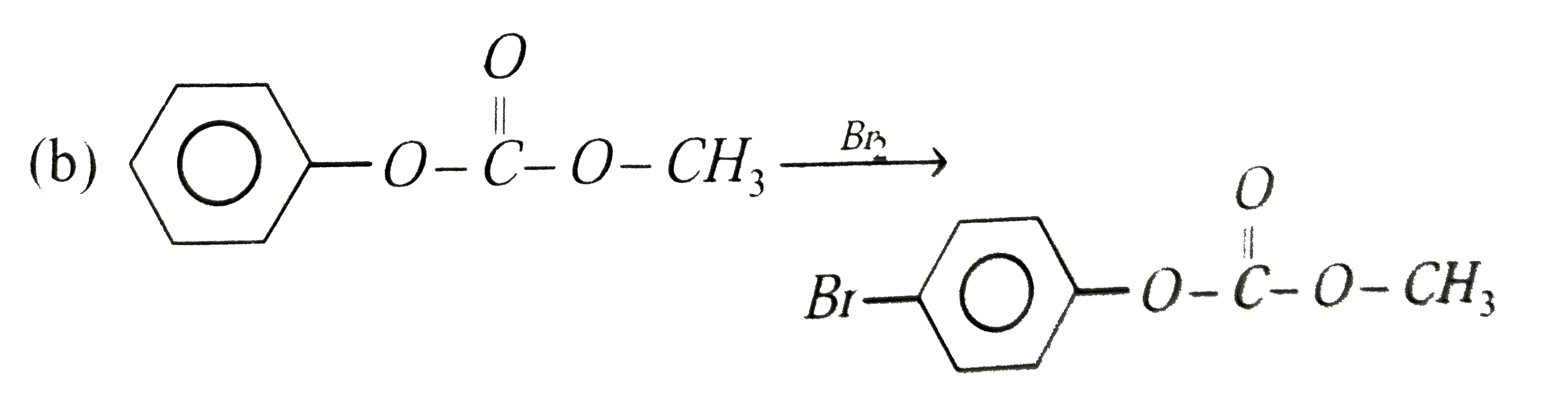
|
|
| 25. |
Phenol reacts with mercuric acetate to form _____and_____ when heated in presence of _____. |
|
Answer» <P> |
|
| 27. |
Phenol reacts with excess bromine water to give |
|
Answer» m bromophemophenol |
|
| 28. |
How does phenol react with: HNO_3 |
|
Answer» o-Nitrophenol |
|
| 29. |
Phenol reacts with bromine water to give a ………………… precipitate of ……………………… . |
| Answer» SOLUTION :WHITE, 2,4, 6-tribromo PHENOL | |
| 30. |
Phenol reacts with bromine is CS_(2) to give : |
|
Answer» <P>o - bromophenol |
|
| 31. |
Phenol reacts with bromine in CS_(2) to give |
|
Answer» o-bromophenol |
|
| 32. |
Phenol reacts with bromine in CS_2 at low temperature to give |
|
Answer» m-bromophenol |
|
| 33. |
Phenol reacts with bromine in CS_2, to give: |
|
Answer» o-Bromophenol |
|
| 34. |
Phenol reacts with Br_2 in C Cl_4 at low temperature to give |
|
Answer» <P>o- and p- bromophenol |
|
| 35. |
Phenol reacts with benzoyl chloride in presence of NaOH. What is the product formed? What is the name of this reaction? |
| Answer» SOLUTION :PHENYL BENZOATE. Schotten-Baumann REACTION. | |
| 36. |
"Phenol" overset("Znpower")to X underset("Anhy." AICl_(3))to Y underset(KmnO_(4))overset("Alkaline")to Z what is Z? |
| Answer» Solution : Benzoic acid | |
| 37. |
Phenol reacts with aqueous bromine to give : |
|
Answer» <P>o - bromophenol |
|
| 38. |
Phenol reacts with Br_2 water to give |
|
Answer» o-Bromophenol |
|
| 39. |
"Phenol" overset(x)toTribromo derivative. What is X? |
|
Answer» BROMINE + Benzene |
|
| 40. |
Phenol overset(NaNO_(2)//H_(2)SO_(4))to B overset(H_(2)O)to C overset(NaOH)toD name of the reaction is |
|
Answer» Liebermann's REACTION 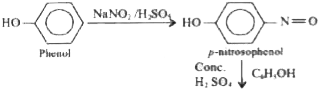 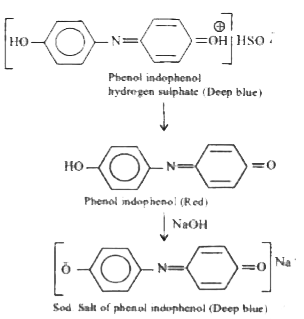
|
|
| 41. |
Phenol overset(X)rarr forms a tribromo derivative. X is |
|
Answer» BROMINE in benzene 
|
|
| 42. |
Phenol overset(X)(to) forms a tribromo derivation. ''X'' is |
|
Answer» BROMINE in BENZENE 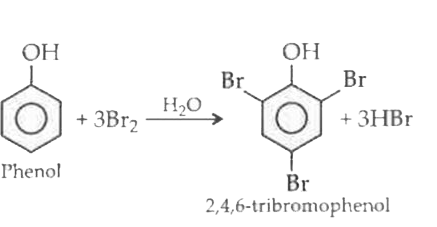
|
|
| 43. |
Phenoloverset((CH_3CO) _2O,H^(+)) to A overset(AlCl_3.Delta )to B+CIf 'B' is steam volatile, incorrect statement among the following is |
|
Answer» Second step is CALLED Fries rerrangement |
|
| 44. |
Phenol overset(NaNO_(2)//H_(2)SO_(4))to B overset(H_(2)O)to C overset(NaOH)to D Name the above reaction is |
|
Answer» Liebermann REACTION |
|
| 45. |
Phenol overset( NaOH) to A underset((2)H^(+)) overset((i)CO_2) to B overset((CH_3CO) _2 O, H^(+))to CIncorrect statement among the following is |
|
Answer» Preparation of 'B' from phenol is CALLED Kolbe s' REACTION. |
|
| 46. |
Phenol overset( conc. H_2SO_4 )toA overset( Conc .HNO_3) to BHereA and B are respectively. |
|
Answer» <P>P- Hydroxy benzenesulphonic acid, P- NITROPHENOL |
|
| 47. |
Phenol on treatment with conc. HNO_(3) produces |
|
Answer» 2-nitrophenol. |
|
| 48. |
Phenol on treatment with diethyl sulphate in presence of NaOH gives |
|
Answer» PHENETOLE `2C_6H_5OH-underset("Phenetole")(O-CH_2CH_3+Na_2SO_4` |
|
| 49. |
Phenol on treatment with CO_(2)//NaOH gives salicylic acid. This reaction is known as ___________. |
| Answer» SOLUTION :Kolbe.s REACTION. | |