Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Phenol is an antiseptic because of its _____________ |
|
Answer» toxic EFFECT on micro-organisms |
|
| 2. |
Phenol is acidic because of resonance stabilization of its conjugate base, namely______. |
| Answer» SOLUTION :PHENOXIDE ION | |
| 3. |
Phenol is an acid but does not react with sodium bicarbonate solution. Explain. |
| Answer» SOLUTION :Phenol is a weaker acid than CARBONIC acid `(H_(2)CO_(3))` ANND hence does not liberate `CO_(2)` from SODIUM bicarbonate. | |
| 4. |
Phenol is also known as |
|
Answer» PHENOLIC acid |
|
| 5. |
Phenol is a very weak acid. What substitutions in the molecule can make it a stronger acid and a weaker acid and why? |
| Answer» Solution :Phenol is very weak acid. Electron withdrawing groups like `-NO_(2)` will INCREASE ACIDIC character due to stabilisation of PHENOXIDE ION whereas electron releasing groups like `-CH_(3)` will make it weaker acid because of destabilisationo f phenoxide ion. | |
| 7. |
Phenol is a bifunctionalcompound because ______. |
|
Answer» it GIVES reactions of HYDROXYL group (-OH) as well as of aromatic ring |
|
| 8. |
Phenol is.... |
|
Answer» STRONG BASE than ammonia |
|
| 9. |
Phenol is |
|
Answer» a weakerbase than `NH_3` . |
|
| 10. |
Phenol has __________ nature and is __________ in H_2O . |
|
Answer» hydroscopic, easily soluble |
|
| 11. |
Phenol is ……. |
|
Answer» ACIDIC |
|
| 12. |
Phenol gives which product of reaction with acid anhydried or acid chloride in alkali medium ? |
|
Answer» PHENYL ester |
|
| 14. |
Phenol gives violet colour with |
|
Answer» NEUTRAL `FeCl_3` |
|
| 15. |
Phenol gives tribromophenol when treated withbromine inaqueous solutionby only o- and p- bromophenolsinCCl_(4) solutionbecause |
|
Answer» in aqueous SOLUTION the brominein ionised |
|
| 17. |
Phenol gives characteristic colouration with |
|
Answer» IODINE solution |
|
| 18. |
Phenol gives salicylaldehyde onheating withCHCl_3 and NaOH. The reaction is called: |
|
Answer» CANNIZZARO's reaction |
|
| 21. |
Phenol do not react with |
|
Answer» POTASSIUM hydroxide |
|
| 22. |
Phenol does not react with sodium carbonate. Why? |
| Answer» SOLUTION :DUE to RESONANCE stablisation. | |
| 23. |
Phenol decolourises reddish brown colour of bromine, though it is not unsaturated compound. Explain. |
|
Answer» Solution :Phenol is highly REACTIVE with `Br_2-H_2O` and FORMS 2, 4, 6-tribromophenol. The REACTION is electrophelile substitution with liberation of HBR. HENCE, the colour of bromine disappears. |
|
| 24. |
Phenol (CHCl_(3)//NaOH) underset(H^(+)) to Salicyldehyde. The above reaction is known as |
|
Answer» Rimer-Tiemann REACTION. |
|
| 25. |
Phenol can be separated from Benzoic acid effectively by the use of |
|
Answer» `NaHCO_(3)` |
|
| 26. |
Phenol can be prepared from |
|
Answer» BENZENE DIAZONIUM chloride |
|
| 27. |
Phenol can be prepared from ________. |
|
Answer» SODIUM PHENOXIDE |
|
| 28. |
Phenol can be prepared by the reaction between |
|
Answer» Aniline and `HNO_3`at 373 K |
|
| 29. |
Phenol can be distinguished from ethyl alcohol by all reagents EXCEPT ________. |
|
Answer» `I_2+NaOH` |
|
| 30. |
Phenol can be distinguished from ethanol the reaction with …. |
|
Answer» `Br2//"water"` |
|
| 31. |
Phenol can be distinguished from ethanol by the reagent |
|
Answer» BROMINE water  ALCOHOL does not react with bromine water. |
|
| 32. |
Phenol can be distinguished from ethanol by the reaction with ____________. |
|
Answer» `Br_(2)//"WATER"` |
|
| 33. |
phenolcan be distinguishedfromethanolby thereactionwith_______. |
| Answer» Answer :D | |
| 34. |
Phenol can be distinguished from ethanol by thefollowing reagents except |
|
Answer» SODIUM<BR>`NaOH//I_(2)` 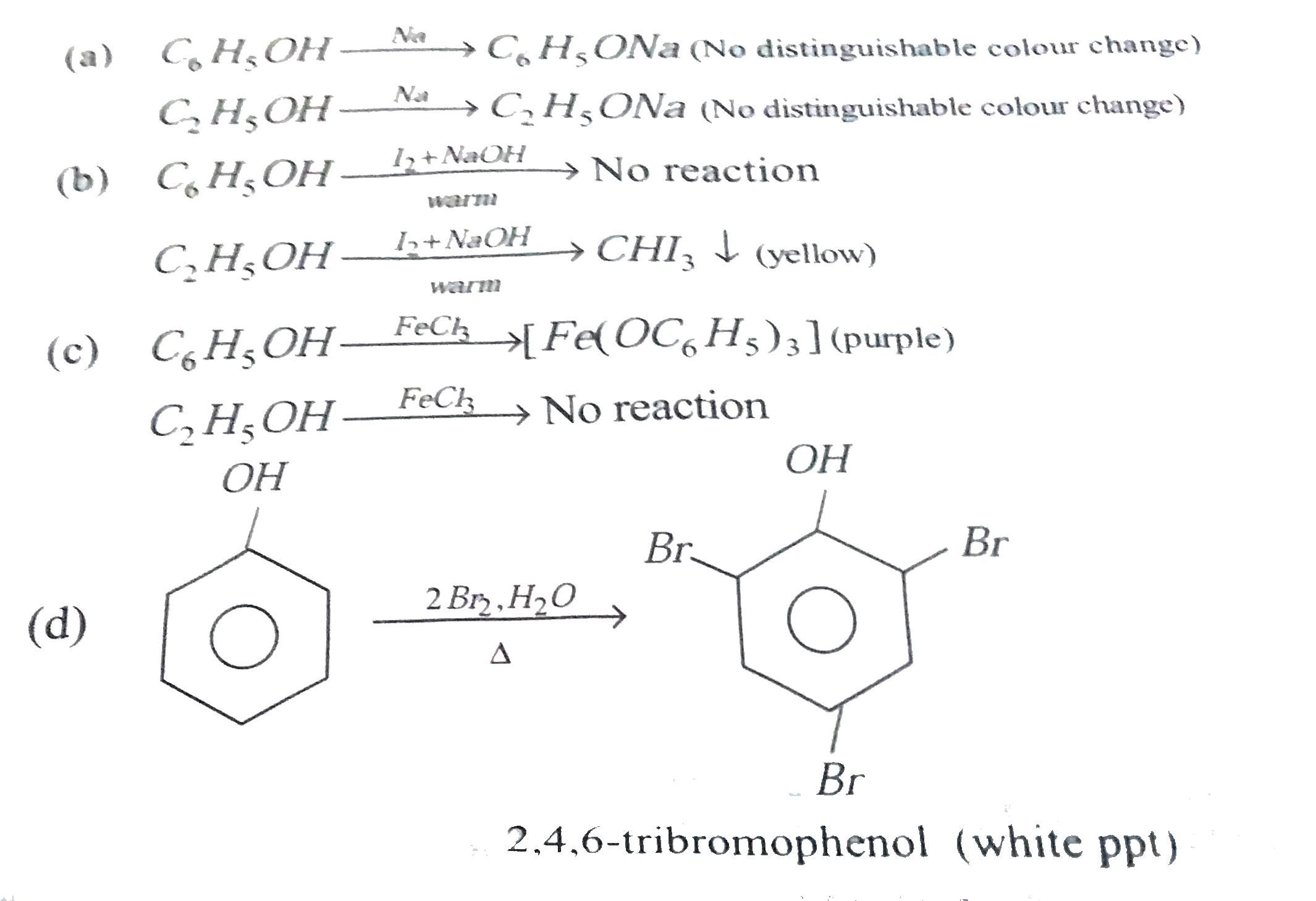 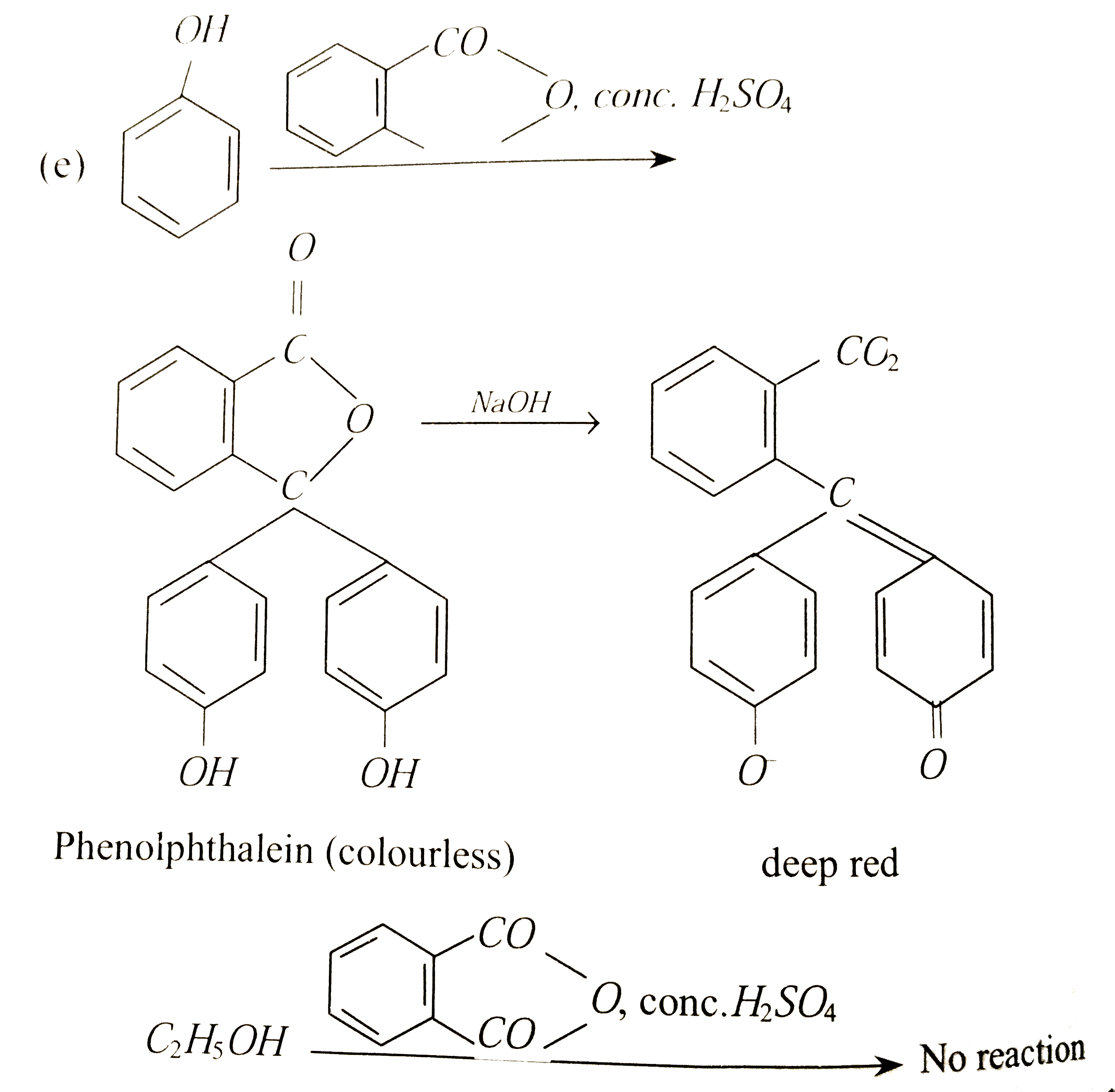
|
|
| 35. |
Phenol at25^(@)C is _____________. |
|
Answer» a colourless crystalline SOLID |
|
| 36. |
Phenol can be distinguished from ethanol by the reaction with ______. |
|
Answer» `Br_(2)`/water<BR>Na |
|
| 37. |
Phenol can be distinguished from ethanol by the following reagents except …………………. . |
|
Answer» SODIUM |
|
| 38. |
Phenol can be distinguished from ethanol by |
| Answer» Answer :C | |
| 39. |
Phenol, C_(6)H_(5)OH when it first reacts with concentrated sulphuric acid, forms Y. The compound, Y is reacted with concentrated nitric acid to form Z. Identify Y and Z and explain why phenol is not converted commercially to Z by reacting it with conc. HNO_(3). |
Answer» SOLUTION :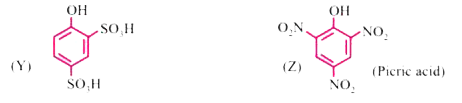 Phenol is not reacted directly with conc. `HNO_(3)` because the yield of PICRIC ACID is very poor. |
|
| 40. |
Phenols can bedistinguishedfromalcoholsby |
|
Answer» NEUTRAL `FeCl_3` |
|
| 41. |
Phenol (C_(6)H_(5)OH) gets associated in water to from double molecules. 0.6677 g of phenol dissolved in 35.5 g of water depresses the freezing point of water by 0.215. (K_(f) "for water"=1.86 K kg^(-1)mol^(-1)). Calculate Observed molar mass of phenol Degree of association of phenol. |
|
Answer» `M_(B)=(K_(f)xxW_(B))/(DeltaT_(f)xxW_(A))` `K_(f)=1.86" K kg mol"^(-1),W_(B)=0.6667 g, DeltaT_(f)=0.215 K, W_(A)=35.5=0.0355 kg.` `M_(B)=((186" K kg mol"^(-1))xx(0.6667g))/((0.215K)xx(0.0355 "kg"))=162.47" g mol"^(-1)` Step II. Calculation of Van't HOFF factor (i). `i=("Normall molar mass of phenol"(C_(6)H_(5)OH))/("OBSERVED molar mass of phenol")=((94"g mol"^(-1)))/((162.47" g mol"^(-1)))=0.579` Step III. Calculation of degree of association of phenol. `alpha=(i-1)/(1/n-1)=((0.579-1))/((0.5-1))=((-0.421))/((-0.5))=0.842.` |
|
| 42. |
Phenol associates in benzene to produce double molecules. To what degree phenol associates if van't Hoff factor is 0.54? |
|
Answer» 0.54 |
|
| 43. |
Phenol associates in benzene to a certain extent to form dimer. A solution containing 2.0xx10^(-2)kg of phenol in 1.0 kg of benzene has its freezing point decreased by 0.69 K. Calculate the degree of association of phenol (K_(f)" for benzene = 5.12 K kg mol"^(-1)). |
|
Answer» `M_(2)"(calculated) for "C_(6)H_(5)OH="94 g mol"^(-1)""therefore""i=(M_(2)"(calculated)")/(M_(2)"(observed)")=(94)/(148.4)=0.633` `{:(,2C_(6)H_(5)OHhArr(C_(6)H_(5)OH)_(2)),("Initial","1mol"),("After asso."," "1-alpha""alpha//2"Total "=1-alpha//2):}` `i=(1-alpha//2)/(1)"or"alpha=2(1-i)=2(1-0.633)=0.734.` |
|
| 44. |
Phenol associates in benzene to a certain extent to form a dimmer. A solution containing20 g of phenol in 1.0 kg of benzene has its freezing point lowered by 0.69 K. Calculate thefraction of phenol that has dimerised. [Given K_f for benzene = 5.1 K m^(-1) |
|
Answer» SOLUTION :`M_2` (OBSERVED) ` = (1000 K_f w_2)/(w_1 Delta T_f)` SUBSTITUTING the values , we have `M_2 = (1000 XX 5.12 xx 2 xx 10^(-2) )/(1.0 xx 0.69) = 148.g "mol"^(-1)` `M_2` (calculated ) = 94 g/mol`i=(M_2("calculated"))/(M_2("observed") = (94)/(148.4) = 0.633` The association can be EXPRESSED as under 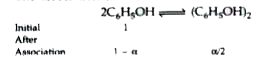 where `alpha ` is the degree of association `i= (1 - alpha//2)/(1) " or" alpha = 2(1 - i) = 2(1-0.633)= 0.734` |
|
| 45. |
Phenol associaes in bensen to a certain extent to from a dimer. A solution containing 2.0xx10^(-2) kg of phenol in 1.0 kg of benzene has a greezing point decreased by 0.69 K. What is the degree of association of phenol ? (K_(f) for benzene=5.12 K kg mol^(-1)). |
|
Answer» `M_(B)=(K_(f)xxW_(B))/(W_(A)xxDeltaT_(f))=((5.12" K kg mol"^(-1))xx(2.0xx10^(-2)kg))/((1.0kg)xx0.69K)` `=14.84xx10^(-2)" kg mol"^(-1)=148.4" G mol"^(-1)` Step II. Calculation of Van't Hoff factor `"Van't Hoff factor (i)"=("Normal molar mass of phenol")/("Obsrved molar mass of phenol")` `=((94" g mol"^(-1)))/((148.4" g mol"^(-1)))=0.633` Step III. `"Calculation of DEGREE of association of phenol "(alpha)` `2C_(6)H_(5)OHhArr(C_(6)H_(5)OH)_(2)` `alpha=(i-1)/(1/n-1)=((0.633-1))/((1/2-1))=((-0.367))/((-0.500))=0.734 or 73.4`% |
|
| 46. |
Phenol associates in benzene to a certain extent to form a dimer. A solution containing 20 g of phenol in 1.0 kg of benzene has its freezing point lowered by 0.69 K. Calculate the fraction of phenol that has dimerised. [Given : K_(f) for benzene = 5.1 Km^(-1)] |
|
Answer» Solution :Association of phenol `C_(6)H_(5)OH (M_(B) = 94)` `W_(B)=20` g ( Phenol ) `DeltaT_(F) `=0.69 K `W_(A) =1` kg (Benzene fraction) `K_(f) =5.1 km^(-1)` Phenol dimerised `DeltaT_(f)= iK_(f)m` `0.69=ixx5.1xx(20)/(94xx1)` `i =(94xx0.69)/(20xx5.1)` `=(64.86)/(102.51)` =0.63 `alpha=(i-1)/((1)/(n)-1)` (Association) `=(0.63-1)/((1)/(2)-1)=(0.37)/(-0.5)` `alpha=0.74` or `= 74%` |
|
| 47. |
Phenol and formaldehyde undergo condensation to give a polymer (A) which on heating with formaldehyde gives a thermo- setting polymer (B). Name the polymers. Write the reactions involved in the formation of (A). What is the structural difference between two polymers ? |
|
Answer» Solution :Phenol and formaldehyde undergo CONDENSATION to give a polymer NOVOLAC (A) Which arc heating with formaldehyde GIVES BAKELITE (B) as a thermosetting polymer. Sequences of the reaction can be WRITTEN as  Structural differences in between Novolac and bakelite is that nonolac is a lnear polymer while bakelite is a cross linked polymer. 
|
|
| 48. |
Phenol and succinic acid |
|
Answer» PHENOL and PHTHALIC ANHYDRIDE |
|
| 49. |
Phenol and ethanol are distinguished chemically by …… |
|
Answer» NEUTRAL `FeCl_(3)` |
|
| 50. |
Phenol and formaldehyde undergo condensation to give a polyer (A) which on heating with formaldehyde gives a thermosetting polymer (B). Name the poylemers. Write the reactions involved in the formation of (A). What is the structural difference between two polymers ? |
| Answer» SOLUTION :A' is novolac and 'B' is bakelite. For REACTIONS involved ANS structural DIFFERENCES. | |