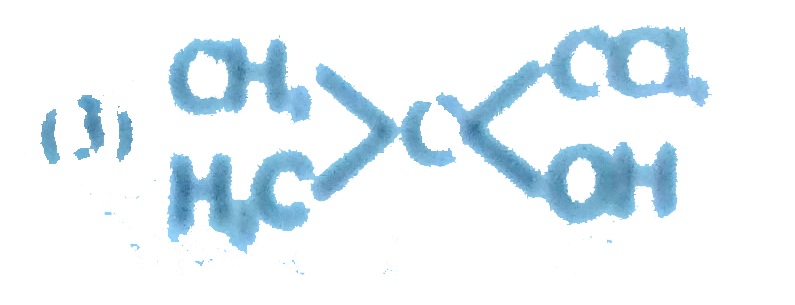Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Phenoland benzoicacid may bedistinguishedby theirreaction with . |
|
Answer» aqueousNaOH |
|
| 2. |
Phenol and benzoic can be separated by |
|
Answer» `NaHCO_(3)` (b) Both will react with NaOH, THEREFORE `NAOH` cannot be used for SEPARATION. (c )Phenol forms complex with `FeCl_(3)` from which phenol cannot be recovered, therefore cannot be used. (d ) is not possible, |
|
| 3. |
phenol and benzoic acid may be distinguished their reaction with |
|
Answer» aqueous NaOH |
|
| 4. |
Phenol and benzoic acid can be distinguished by |
|
Answer» Conc. `H_(2)SO_(4)` |
|
| 5. |
Phenoland benzoic acid is distinguished by |
| Answer» Answer :B | |
| 6. |
Phenetole is ….. |
| Answer» Solution :Ethoxy benzene | |
| 8. |
Phenetole react with cold HI gives |
|
Answer» `C_6H_5-I+C_2H_5-OH` |
|
| 9. |
Phenol and benzoic acid are separated by: |
|
Answer» Solution :`PhOh+NaHCO_(3)rarr"No reaction "` `PhCOOH+NaHCO_(3)rarrPhCOunderset(("salt"))overset(Θ)O Na^(o+)+H_(2)O+CO_(2)` |
|
| 10. |
Phenol. |
|
Answer» Solution :(1) Phenol is USED in the preparation of phenol-formaldehyde polymer which is used in a plastic BAKELITE. (2) It is used in the preparation of phenol-phthalein-an indicator and in certain dyes. (3) It is used in the preparation of drugs such as SALOL, aspirin, etc. (4) It is used in the preparation of DETTOL, which is an antiseptic. (5) It is used in the preparation of 2,4-dichlorophenoxy acetic acid which is used as selective weed killer. (6) It is used to prepare picric acid which is used as explosive. |
|
| 11. |
phenelzine is used in |
|
Answer» DEPRESSION |
|
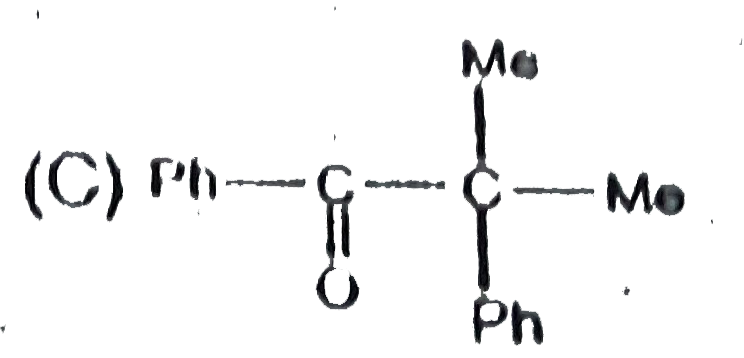
| 12. |
Phenelzin is used as an |
|
Answer» ANALGESIC |
|
| 13. |
PhCOOH, PhCH_3 can be separated by |
|
Answer» `KMnO_4` |
|
| 14. |
Phenacetin is used as |
|
Answer» ANTIPYRETIC |
|
| 15. |
Phenacetin is used as : |
|
Answer» Antipyretic |
|
| 16. |
PhCOOC_(2)H_(5)+C_(2)H_(5)-COOHC_(2)H_(5) overset(NaNH_(2))(to) Number of condensated products (including stereoisomer) |
Answer» 
|
|
| 17. |
PhCOCH_(2)CH_(2)NH_(2) and PhNHCH_(2)COCH_(3) can be distinguished by |
|
Answer» Carbylamine TEST |
|
| 18. |
PhCHO+(CH_3CO)_2Ooverset(CH_2COONa)toAoverset(HBr)toB The product B is |
|
Answer» `PhCH=CHCH_2Br` |
|
| 19. |
PhCH_(2)CH_(2)COOH overset(Ag_(2)O)to PhCH_(2)CH_(2)COOAg overset(Br_(2))to PhCH_(2)CH_(2)Br+CO_(2)+AgBr The reaction is known as Hunsdiecker reaction. Choose incorrect statement(s) regarding the Hunsdiecker reaction. |
|
Answer» A more convenient WAY to preform the Hunsdiecker REACTION is the use of a mixture of acid and mercuric oxide. |
|
| 20. |
PhCHO +(CH_(3)CO)_(2)O overset((1)CH_(3)COONa)underset((2)"hydrolysis", Delta)toA overset(HBr)toB B The product B is |
|
Answer» `PhCH=CHCH_(2)Br` `underset("(A)")(Ph-CH=CH-COOH) overset(HBR)to Ph-underset(Br)underset(|)(C)H-CH_(2)-COOH` |
|
| 21. |
Ph(CH_2)_3 -oversetoverset(O)(||)C-Cloverset(AlCl_3)to(X)overset(Na_2Cr_2O_7. H^(+)"heat")to(Y)overset("heat")to(Z) |
|
Answer»
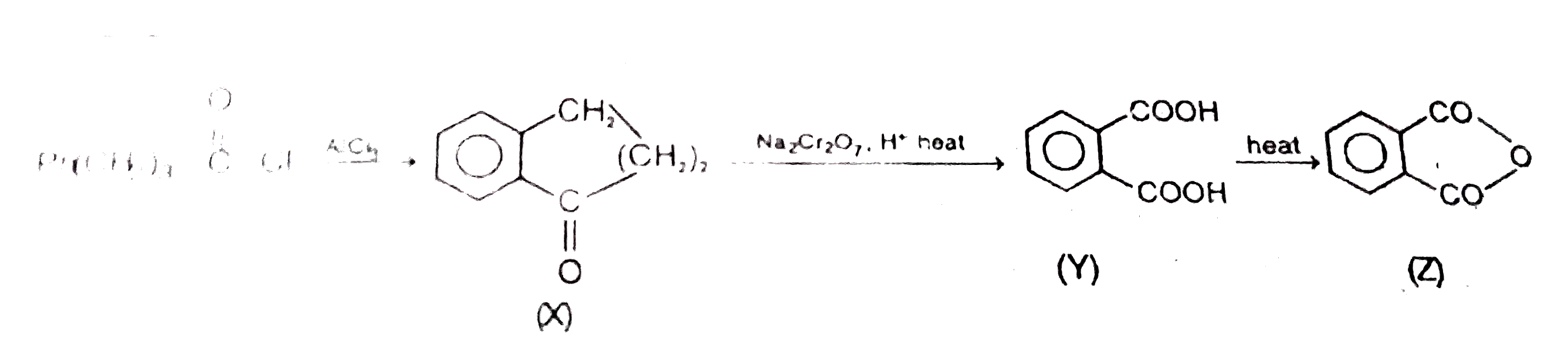
|
|
| 22. |
PHBV is prepared from |
|
Answer» glycolic ACID |
|
| 23. |
PhC-=CPh overset(Hg^(2+)+H^(o+))to(A). The compound (A) is The compound (A) is: |
|
Answer» a.  HENCE the ANSWER is `(a)`. |
|
| 24. |
PHBV is of which class polymer ? |
| Answer» Solution :Polyester | |
| 25. |
PHBV is a biodegradable polymer and is obtained by copolymerization of ....................with ................... |
|
Answer» |
|
| 26. |
Phase transitions are ubiquitous in nature. We are all familiar with the different phase of water (vapour, liquid and ice) and with the change from one to another, the change of phase are called phase transitions. There are six ways a substance can change between these three phase, melting, freezing, evaporating, condensing sublimation and decomposition. At 1 atm pressure vaporisation of 1 mole of water from liquid (75^(@)C) to vapour (120^(@)C). C_(v)(H_(2)O,l)=75 J "mole"^(-1)K^(-1), C_(p)(H_(2)O,g)=33.3J"mole"^(-1)K^(_1) Delta H_(vap) at 100^(@)C=40.7KJ//"mole" Calculate change in internal energy when Water vapour at 100^(@)C to 120^(@)C ? |
|
Answer» `666 J` |
|
| 27. |
Phase transitions are ubiquitous in nature. We are all familiar with the different phase of water (vapour, liquid and ice) and with the change from one to another, the change of phase are called phase transitions. There are six ways a substance can change between these three phase, melting, freezing, evaporating, condensing sublimation and decomposition. At 1 atm pressure vaporisation of 1 mole of water from liquid (75^(@)C) to vapour (120^(@)C). C_(v)(H_(2)O,l)=75 J "mole"^(-1)K^(-1), C_(p)(H_(2)O,g)=33.3J"mole"^(-1)K^(_1) Delta H_(vap) at 100^(@)C=40.7KJ//"mole" Calculate change in internal energy when Water liquid at 100^(@)C to vapour at 100^(@)C ? |
|
Answer» `40700 J` |
|
| 28. |
Phase transitions are ubiquitous in nature. We are all familiar with the different phase of water (vapour, liquid and ice) and with the change from one to another, the change of phase are called phase transitions. There are six ways a substance can change between these three phase, melting, freezing, evaporating, condensing sublimation and decomposition. At 1 atm pressure vaporisation of 1 mole of water from liquid (75^(@)C) to vapour (120^(@)C). C_(v)(H_(2)O,l)=75 J "mole"^(-1)K^(-1), C_(p)(H_(2)O,g)=33.3J"mole"^(-1)K^(_1) Delta H_(vap) at 100^(@)C=40.7KJ//"mole" Calculate change in internal energy when Water liquid at 75^(@)C to 100^(@)C ? |
|
Answer» `1875 J` |
|
| 29. |
PHBV is |
|
Answer» polyester POLYMER |
|
| 30. |
PH_(4)I +KOH to underset(("gas"))(A) +underset(("salt"))(B) +H_(2)O Incorrect statement :- |
|
Answer» .A. is INFLAMMABLE gas |
|
| 31. |
Pharmaceutical products are highly effective in which of the following form ? |
|
Answer» EMULSION |
|
| 32. |
PH_4I + NaOH forms |
|
Answer» `PH_3` |
|
| 33. |
[(Ph_3P)_3RhCl]is a familiar catalyst used in |
|
Answer» HYDROGENATION of oils |
|
| 34. |
(Ph_3P)_3RhCl is |
|
Answer» ZIEGLAR nutta catalyst |
|
| 35. |
[(Ph_(3)P)_(3)RhCI], a rhodium complex is used for the hydrogenation of alkenes. |
|
Answer» |
|
| 36. |
PH_3 produces smoky rings when it comes in contact with air this is because: |
|
Answer» It is inflammable |
|
| 37. |
PH_3 has lower boiling point than NH_3. Why ? |
| Answer» SOLUTION :Unlike `NH_3, PH_3` molecules are not ASSOCIATED through HYDROGEN bonding in liquid STATE. That is why the BOILING point of `PH_3` is lower than `NH_3`. | |
| 38. |
PH_3 forms bubbles when passed slowly in water but NH_3 dissolves. Explain why ? |
| Answer» Solution :`NH_3` forms hydrogen bonds with WATER because of high electronegativity of nitrogen hence it gets soluble in water. PHOSPHORUS, HOWEVER cannot form hydrogen bonds hence it forms BUBBLES in water. | |
| 39. |
PH_3 forms bubbles when passed slowly in water but NH_3 dissolves. Explainwhy ? |
| Answer» Solution :` NH_3` forms HYDROGEN bonds with water. THEREFORE it is soluble in water but `PH_3` cannot FORM hydrogen BOND with water. Therefore it ESCAPES as gas. | |
| 40. |
PH_3 can be obtained by heating |
|
Answer» White phosphorus with not concentrated alkali b) `2H_(3)PO_(2)overset(DELTA)rarr3H_(3)PO_(4)+PH_3` C) `4H_(3)PO_(3)overset(Delta)rarr3H_(3)PO_(4)+PH_3` |
|
| 41. |
PH_(3) can be obtained by : |
|
Answer» heating hypophosphorus acid While heating oxyacids YIELDS oxides and water. |
|
| 42. |
pH values of HCl and NaOH solutions each of strength (N)/(100) will be respectively |
|
Answer» 2 and 3 `[OH] = 10^(-2) M` for NAOH `pH + pOH = 14, pH =14-2, pH = 12`. |
|
| 43. |
Ph_(2)CH-overset(O)overset("||")C-OHoverset(1.SOCl_(2))underset(2.Et_(3)N,Delta)rarr |
| Answer» Answer :A | |
| 44. |
pH value of which one of the following is not equal to one |
|
Answer» `0.1 M HNO_(3)` (a) value of pH `= 0.1 M HNO_(3) = -log[0.1] = 1` (b) Value of pH `= 0.05 H_(2)SO_(4) = -log [2 xx 0.05] = 1` (c) Value of pH `= 0.1 M CH_(3)COOH != 1` as it is a weak acid so, will not undergo complete ionization. (d) Milli-equivalent of acid `= 50 xx 0.4 = 20` Milli-equivalent of BASE `= 50 xx 0.2 = 10` Remaining acid `= 20 -10 = 10` milli-eq. `[H^(+)] = (10)/(50+50) = (10)/(100) = 0.1` `pH = -log[H^(+)] = 1`. |
|
| 45. |
pH value of which one of the following is NOT equal to one ? |
|
Answer» `0.1" M HNO"_(3)` (a) VALUE of `pH=0.1" M HNO"_(3)=-log[0.1]=1` (b) Value of `pH=0.05H_(2)SO_(4)=-log[2xx0.05]=1` (c) Value of `pH=0.1"M "CH_(3)COOH!=1` as it is a weak acid so, will not undergo complete ionization. (d) Milli - equivalent of acid `=50xx0.4=20` Milli-equivalent of base `=50xx0.2=10` Remaining acid `=20-10=10` milli-eq. `[H^(+)]=(10)/(50+50)=(10)/(100)=0.1` `pH=-log[H^(+)]=1`. |
|
| 46. |
Ph-underset(O)underset(||)(C)-NH_(2) overset(POCl_(3))to products is |
|
Answer» Benzonitrile |
|
| 47. |
Ph-undersetunderset(OH)(|)oversetoverset(Me)(|)C-undersetunderset(OH)(|)oversetoverset(Me)(|)C-Et overset(conc.H_2SO_4,Delta)toProducts How many number of different type of carbonylproducts (only structural isomers ) can be formed (major or minor ) in this reaction, (considering all types of possible migrations) |
|
Answer» |
|
| 48. |
Ph-undersetunderset(Me)(|)oversetoverset(OH)(|)C-undersetunderset(I)(|)oversetoverset(Me)(|)C-Phunderset(Delta)overset(AgNO_3)to? Major product is : |
|
Answer»
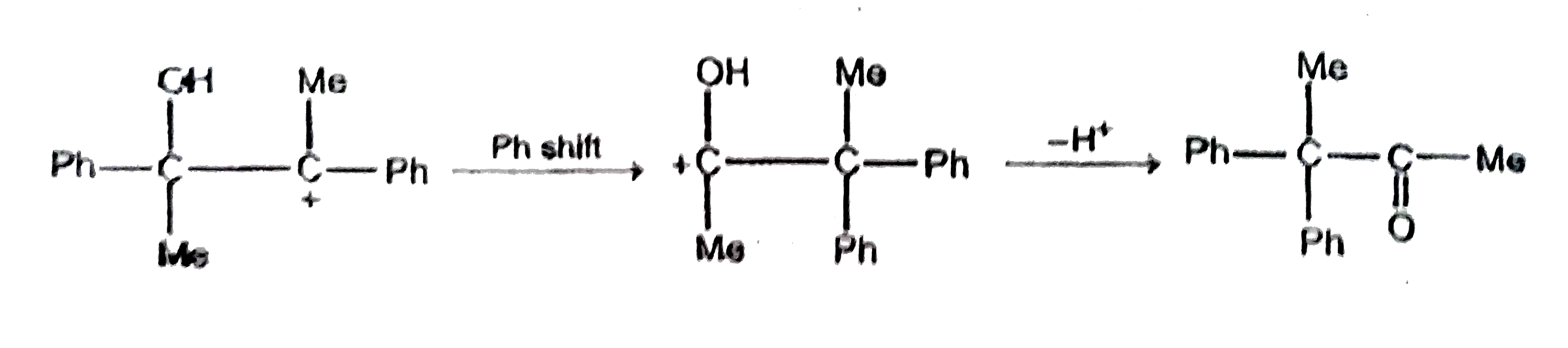
|
|
| 49. |
Ph-underset(O)underset(||)(C)-CH_(3)overset(NaOCl)(to)A+ "salt" A+CH_(3)-underset(O)underset(||)(C)-CH_(3)toB 'B' is |
|
Answer» `CH_(3)-underset(O)underset(||)(C)-CH=underset(CH_(3))underset(|)(C)-PH` |
|