Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Percentage composition in terms of mass of a solution obtaned by mixing 300g 25% solution of NH_(4)NO_(3) with 400g of 40% solution of solute 'X' |
|
Answer» PRECENTAGE by MASS of SOLUTE `33.57%` |
|
| 2. |
Percentage of A_1 in air is about |
|
Answer» 0.01 |
|
| 3. |
Percent purity of iron in cast iron is |
|
Answer» 90 |
|
| 4. |
Per two gram of charcoal, a gas is adsorbed by 0.1g and 0.2g at 10 torr and 80 torr pressure respectively. Calculate the n value in Freundlich adsorption isotherm. |
|
Answer» Solution :`(x)/(m)=K.P^(1//n)(or) (X_(1))/(X_(2))=((P_(1))/(P_(2)))^(1//n)` SUBSTITUTING the values, `(0.1)/(0.2)=((10)/(80))^(1//n) (or)((1)/(2))^(1)=((1)/(2))^(3//n)` Therefore, the VALUE of n is 3. |
|
| 5. |
Per monomeric unit, how much is the increase in mass when cellulose is treated with AcOAc in excess ? |
|
Answer» |
|
| 7. |
Peptization is a process of : |
|
Answer» PRECIPITATING colloidal particles |
|
| 8. |
Peptization is a process |
|
Answer» of PRECIPITATION of colloidal particles |
|
| 9. |
Peptization involves |
|
Answer» Precipitation of colloidal particles |
|
| 10. |
Peptization denotes |
|
Answer» Digestion of FOOD |
|
| 11. |
Peptising agent is added to convert precipitate into colloidal solution. Explain with an example. |
|
Answer» Solution :(i) Ions either positive or negative of peptizing agent (electrolyte) are adsorbed on the particles of precipitate. They repel and hit each other and break the particles of the precipitate into colloidal size. (ii) For EXAMPLE, when we add a small volume of very DILUTE hydrochloric ACID sotion peptisingagent to a fresh precipitate of a silver CHLORIDE, it leads to formation of silver chloride colloidal solution, `UNDERSET("(Precipitate)")(AgCl)overset(HCl)rarrunderset("(Colloid)")(AgCl)` |
|
| 12. |
Peptisation is not used to prepare |
|
Answer» SILVER CHLORIDE sol |
|
| 13. |
Peptisation is a method of preparation of sol. Write a general procedure for peptisation. |
|
Answer» |
|
| 15. |
Peptides are amino acid polymer in which the individual amino acid units are called |
|
Answer» monomer |
|
| 16. |
Peptide linkage is |
|
Answer» `-OVERSET(O)overset(||)(C )-O-` |
|
| 17. |
Peptide bond is a key feature in: |
|
Answer» Polysaccharide |
|
| 18. |
Pepsin enzyme hydrolysis |
|
Answer» Proteinsto AMINO ACIDS |
|
| 19. |
Pepperment can be extracted form plant sources by using solvents like : |
|
Answer» `NH_3` |
|
| 20. |
Pentyl cyanide to 1-aminopentane |
|
Answer» Solution :`CH_(3)-(CH_(2))_(3)-CH_(2)-Cnoverset(H_(2)O)rarr` `CH_(3)-(CH_(2))_(3)-CH_(2)-CONH_(2)overset(Br_(2)//KOH)rarr UNDERSET("(1 - pentanamine)")(CH_(3)-(CH_(2))_(3)-CH_(2)-NH_(2))` |
|
| 21. |
Pentoxides of all the elements of nitrogen family are _____ acidicthan their trioxides. |
|
Answer» Less |
|
| 22. |
Pentavalence in phosphorous is more stable when compared to that of nitrogen even though they belong to same group is due to |
|
Answer» <P>reactivity of phosphorous |
|
| 23. |
Pentose sugar present in RNA is |
|
Answer» `BETA`-D-ribose |
|
| 24. |
Pentan-3-one is not obtained from |
|
Answer» 2,2-dichloro PENTANE `CH_(3)CH_(2)CH_(2)underset(Cl)underset(|)overset(Cl)overset(|)C-CH_(3) overset("Hydrolysis")to CH_(3)CH_(2)CH_(2)underset(OH)underset(|)overset(OH)overset(|)C-CH_(3) underset(-H_(2)O)toCH_(3)CH_(2)CH_(2) underset("pentane-2-one")(underset(O)underset(||)(C)-CH_(3))` |
|
| 25. |
Pentan-3-ol on reaction with aluminium tertiary butoxide in presence of acetone gives |
|
Answer» pentane 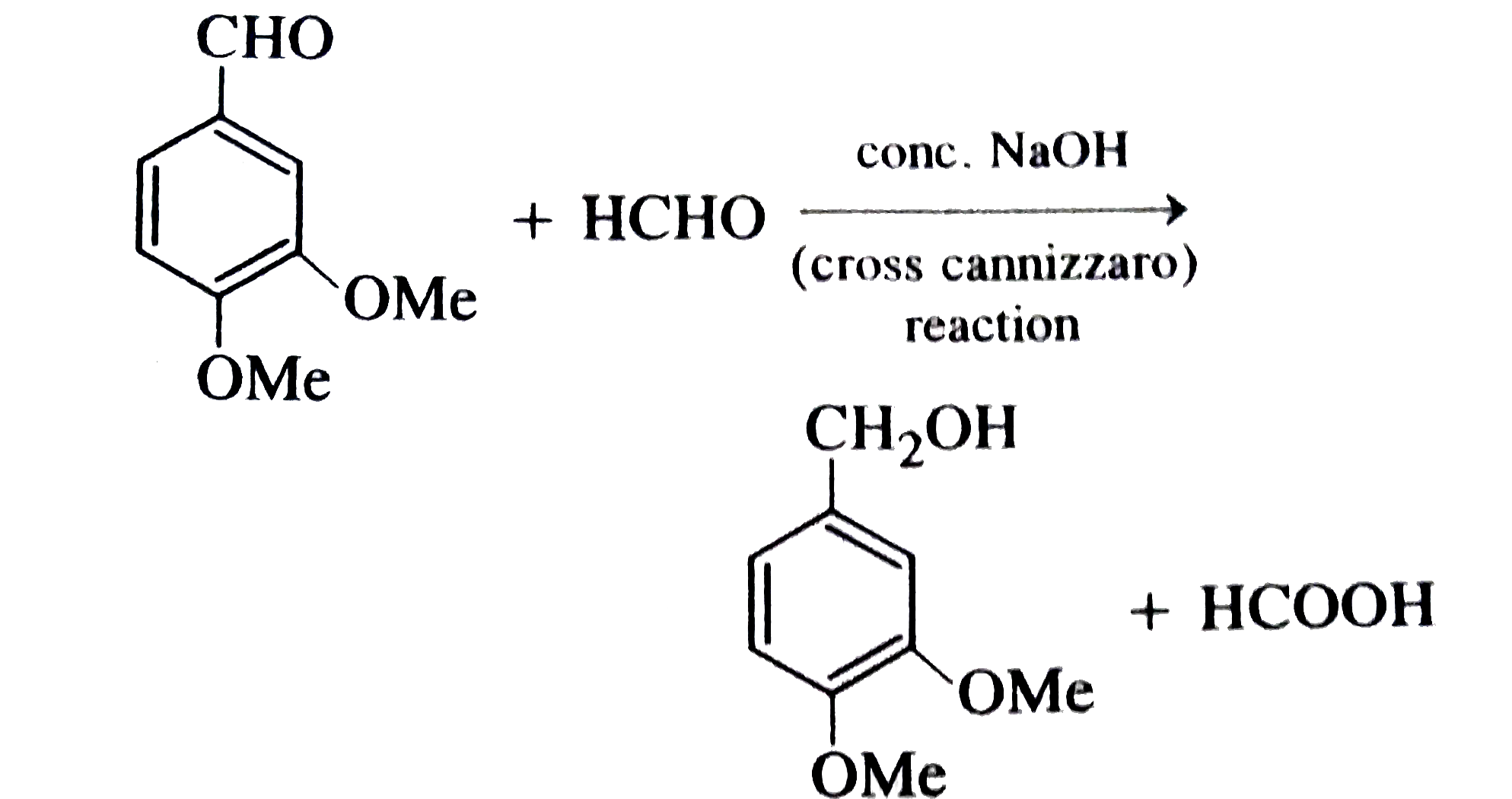
|
|
| 26. |
Pentahalides of phosphorus are known, but not pentahydride. Why ? |
| Answer» Solution :Phophorus exhibits pentavalency in excited state. FLUORINE, chlorine or bromine is more ELECTRONEGATIVE and influences excitation to form `PX_(5)`. However, hydrogen is not that much electronegative to incluence the excitation in phosphorus. Hence `PH_(5)` is not FORMED | |
| 27. |
Pentan -3- en-2-one is reduced by LiAlH_(4) gives |
|
Answer» PENT -4- en-2-ol |
|
| 28. |
Pentan - 2- one and 3- methyl butan - 2- one are |
|
Answer» chain isomers |
|
| 29. |
Pentlandite is an ore of |
|
Answer» Co |
|
| 30. |
Pentalandite is an ore of: |
|
Answer» Fe |
|
| 31. |
Pentahalides of phosphorus are known, but not pentahydride. Why? |
| Answer» SOLUTION :Phosphorous exhibits PENTAVALENCY in excited state. Fluorine, CHLORINE or bromine is more ELECTRONEGATIVE and influences excitation to form `PX_(5)`. However, hydrogen is not that much electronegative to INFLUENCE the excitation in phosphorus. Hence `PH_(5)` is not formed. | |
| 32. |
PentaeryI thrityI monosterate is an example of |
|
Answer» ANIONIC detergent |
|
| 33. |
Pentagonal bipyramidal structure contains bond angles approximately |
|
Answer» 1. `120^(@), 90^(@), 180^(@)` |
|
| 34. |
Pentaammine nitrocobalt(III) cation possesses the property of |
|
Answer» physiorption 
|
|
| 35. |
Penta hydroxy hexanone is ______. |
|
Answer» an ALDO hexose |
|
| 37. |
Penicillin was fist discovered by |
| Answer» Answer :A | |
| 38. |
Penicillin is which type of drug ? |
|
Answer» Antifertility |
|
| 39. |
Penicillin is an example of |
|
Answer» Analgesic |
|
| 40. |
Penicillin was first discovered by |
|
Answer» A. FLEMING |
|
| 41. |
Penicillin is a : |
|
Answer» Hormone |
|
| 42. |
Penicillin and ofloxacin are _______antibiotics |
|
Answer» |
|
| 43. |
Penicillin, ampicillin, cephalosorins, hydrogen peroxide, carbapenems |
| Answer» SOLUTION :HYDROGEN PEROXIDE. It is an ANTISEPTIC where as other are ANTIMICROBIALS. | |
| 44. |
Penicillin has general molecular formula |
|
Answer» `C_(9)H_(11) O_(4)SN_)(2)R` |
|
| 45. |
Penicillin is |
|
Answer» Analgesic |
|
| 48. |
Pegarding the structure of SO_(2) and SeO_(2) which of the following is true- |
|
Answer» The gaseous `SO_(2)` and `SeO_(2)` have same V - shaped molecule both in solid and GAS PHASE |
|



