Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
p-Amino-N,N-dimethylaniline is added to a strongly acidic solution of X.The resulting solution is treated with few drops of aqueous solution of Y to yield blue colouration due to the formation of methylene blue.Treatment of the aqueous solution of Y with the reagent potassium hexacyanoferrate(II) leads to the formation of an intension blue precipitate.The precipitate dissolves on excess addition of the reagent.Similarly, treatment of the solution of Y with the solution of potassium hexacyanoferrate(III) leads to a brown coloration due to the formation of Z. The compound Z is: |
|
Answer» `Mg_(2)[Fe(CN)_(6)]`  The COMPOUND `X` is `Na_(2)S` and `Y` is `FeCl_(3)` |
|
| 2. |
p-aminobenzoic acid reacts with bromine and H_(2)O to give |
|
Answer»

|
|
| 3. |
p -amino phenol is the product of readucing nitrobezene in……………. |
|
Answer» ACID medium |
|
| 4. |
p-Amino-N,N-dimethylaniline is added to a strongly acidic solution of X.The resulting solution is treated with few drops of aqueous solution of Y to yield blue colouration due to the formation of methylene blue.Treatment of the aqueous solution of Y with the reagent potassium hexacyanoferrate(II) leads to the formation of an intension blue precipitate.The precipitate dissolves on excess addition of the reagent.Similarly, treatment of the solution of Y with the solution of potassium hexacyanoferrate(III) leads to a brown coloration due to the formation of Z. The compound Y is : |
|
Answer» `MgCl_(2)` 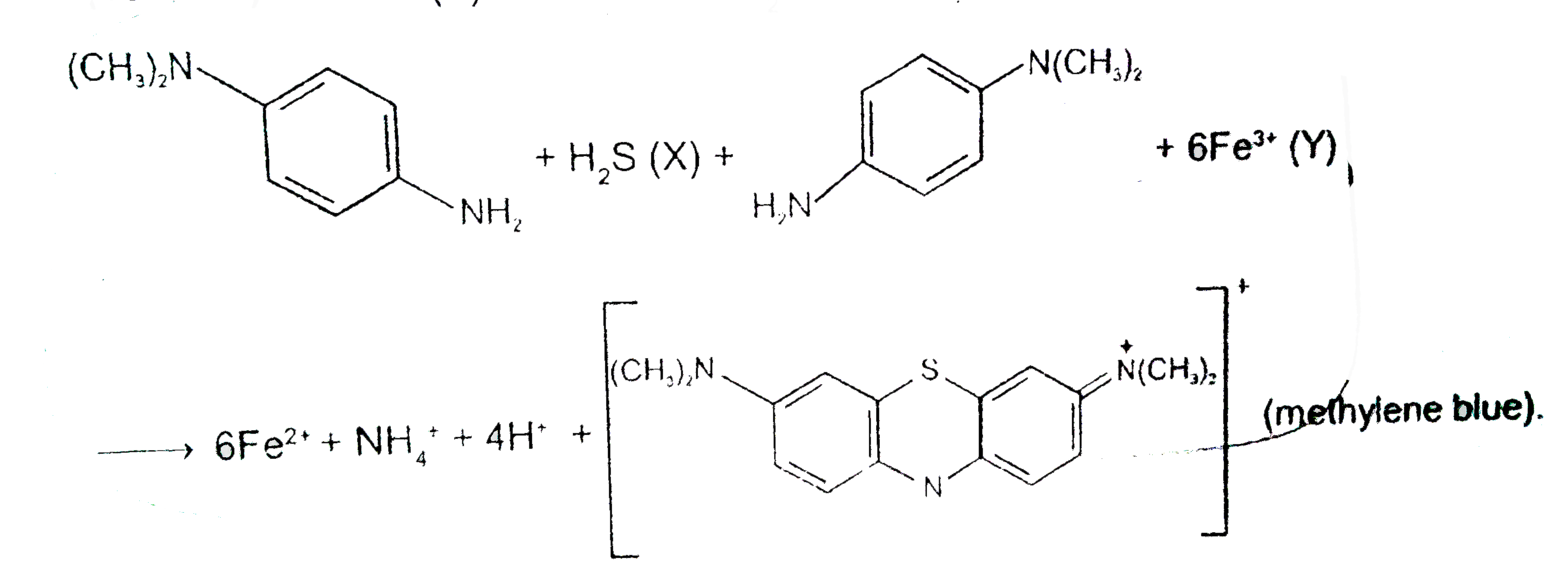 The COMPOUND `X` is `Na_(2)S` and `Y` is `FeCl_(3)` |
|
| 5. |
p-Amino-N,N-dimethylaniline is added to a strongly acidic solution of X.The resulting solution is treated with few drops of aqueous solution of Y to yield blue colouration due to the formation of methylene blue.Treatment of the aqueous solution of Y with the reagent potassium hexacyanoferrate(II) leads to the formation of an intension blue precipitate.The precipitate dissolves on excess addition of the reagent.Similarly, treatment of the solution of Y with the solution of potassium hexacyanoferrate(III) leads to a brown coloration due to the formation of Z. The compound X is: |
|
Answer» `NaNO_(3)` 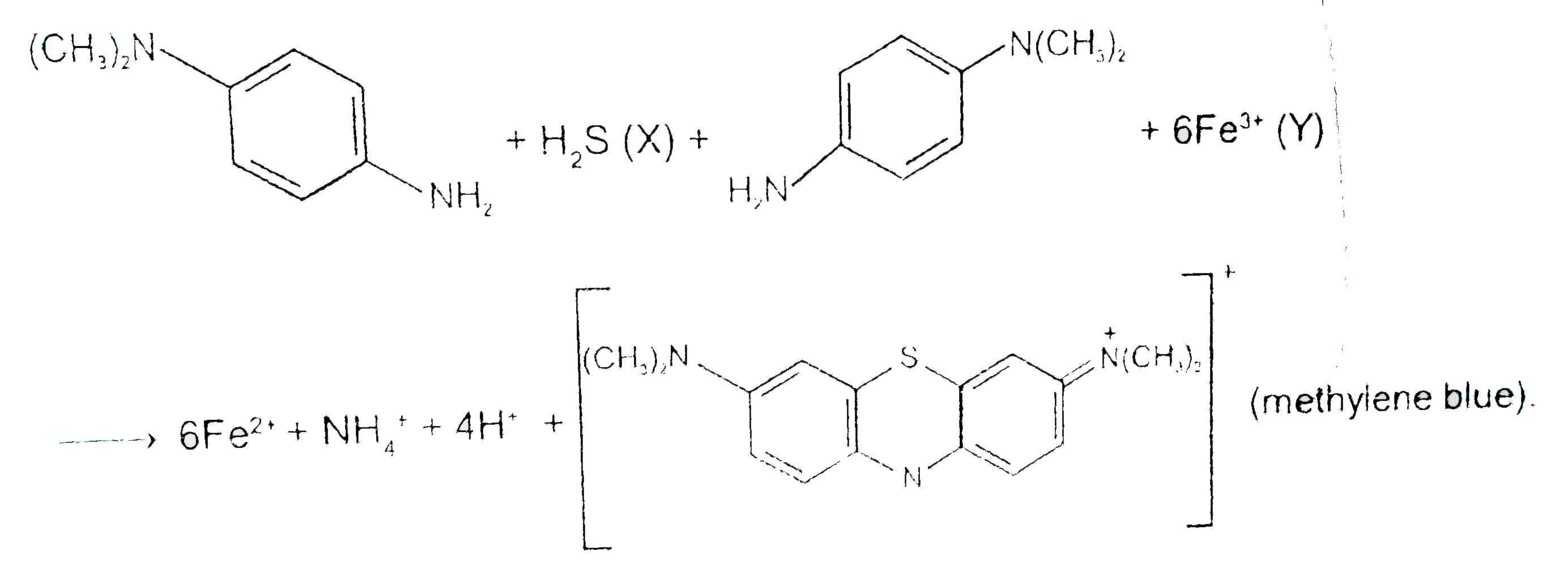 The compound `X` is `Na_(2)S` and `Y` is `FeCl_(3)` |
|
| 6. |
p-Amino-N, N-dimethylaniline is added to a strongly acidic solution of X. The resulting solution is treated with a few drops of aqueous solution of Y to yield blue coloration due to the formation of mothylene blue. Treatment of the aqueous solution of Y with the reagent potassium hexacyanoferrate (II) leads to the formation of an intense blue precipitate. The precipitate dissolves on excess addition of the reagent. Similarly, treatment of the solution of Y with the solution of potassium hexacyanoferrate (III) leads to a brown coloration due to the formation of Z.Q The compound Z is |
|
Answer» `Mg_(2)[Fe(CN)_(6)]` 
|
|
| 7. |
p-Amino-N, N-dimethylaniline is added to a strongly acidic solution of X. The resulting solution is treated with a few drops of aqueous solution of Y to yield blue coloration due to the formation of mothylene blue. Treatment of the aqueous solution of Y with the reagent potassium hexacyanoferrate (II) leads to the formation of an intense blue precipitate. The precipitate dissolves on excess addition of the reagent. Similarly, treatment of the solution of Y with the solution of potassium hexacyanoferrate (III) leads to a brown coloration due to the formation of Z.Q The compound Y is |
|
Answer» `MgCl_(2)` 
|
|
| 8. |
p-Amino-N, N-dimethylaniline is added to a strongly acidic solution of X. The resulting solution is treated with a few drops of aqueous solution of Y to yield blue coloration due to the formation of mothylene blue. Treatment of the aqueous solution of Y with the reagent potassium hexacyanoferrate (II) leads to the formation of an intense blue precipitate. The precipitate dissolves on excess addition of the reagent. Similarly, treatment of the solution of Y with the solution of potassium hexacyanoferrate (III) leads to a brown coloration due to the formation of Z.Q The compound X is |
|
Answer» `NaNO_(3)` 
|
|
| 9. |
p-amino-N, N-dimethyl laniline is added to a strongly acidci solution of x. The resulting solution is treated with a few drops of aqueous solution of Y to yield blue colouration due to the formation of methylene, blue. Treatment of the aqueous solution of Y with the reagent potassium hexaxyanoferrate (II) leads to the formation of an intense blue precipitate. The precipitate of the solution of Y with the solution of potassium hexacyanoferrate (III) leads to a brown colouration due to the formation of Z. |
|
Answer» `Mg_(2)[Fe(CN)_(6)]` |
|
| 10. |
p-amino-N, N-dimethyl laniline is added to a strongly acidci solution of x. The resulting solution is treated with a few drops of aqueous solution of Y to yield blue colouration due to the formation of methylene, blue. Treatment of the aqueous solution of Y with the reagent potassium hexaxyanoferrate (II) leads to the formation of an intense blue precipitate. The precipitate of the solution of Y with the solution of potassium hexacyanoferrate (III) leads to a brown colouration due to the formation of Z. The compound Y, is |
|
Answer» `MgCl` |
|
| 11. |
p-amino-N, N-dimethyl laniline is added to a strongly acidci solution of x. The resulting solution is treated with a few drops of aqueous solution of Y to yield blue colouration due to the formation of methylene, blue. Treatment of the aqueous solution of Y with the reagent potassium hexaxyanoferrate (II) leads to the formation of an intense blue precipitate. The precipitate of the solution of Y with the solution of potassium hexacyanoferrate (III) leads to a brown colouration due to the formation of Z. The compound X, is |
| Answer» Answer :D | |
| 13. |
Ozonolysis of the following gives only acetaldehyde |
|
Answer» 2 - BUTENE |
|
| 14. |
Ozonolysis of terminal alkenes gives ________ as one of the products. |
|
Answer» acetone |
|
| 15. |
Ozonolysis of propene givs |
|
Answer» only formaldehyde. |
|
| 16. |
Ozonolysis of propyne gives: |
|
Answer» `CH_3CHO` |
|
| 17. |
Ozonolysis of C_(7)H_(14) give methanal and 2- methyl pentan -3- one . The C_(7)H_(14) is |
|
Answer» 2- ETHYL 3 - methyl but -1- ene |
|
| 18. |
Ozonolysis of an organic compound gives formaldehyde as one of the products. This confirms the presence of : |
|
Answer» two ethylenic double bonds |
|
| 19. |
Ozonolysis of an organic compound 'A' produces acetone and propionaldehyde in equimolar mixture. Identify 'A' from the following compounds |
|
Answer» 2-Methyl-1-pentene 
|
|
| 20. |
Ozonolysis of an organic compound 'A' produces acetone and propionaldehyde in equimolar mixture. Identify 'A' from the following compounds : |
|
Answer» `1`-PENTENE 
|
|
| 21. |
Ozonolysis of an alkene produces only one dicarbonylcompound . The structure of the alkene is |
|
Answer» `CH_3 - CH = CH - CH_3` 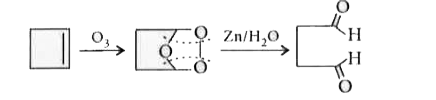
|
|
| 22. |
Ozonolysis of an alkene [A] followed by decomposition with water and a reducing agent gave a mixture of two isomers of the formula C_(3)H_(6)O. The structure of alkene is : |
|
Answer» `CH_(2) = CH CH_(2)CH (CH_(3))_(2)` |
|
| 23. |
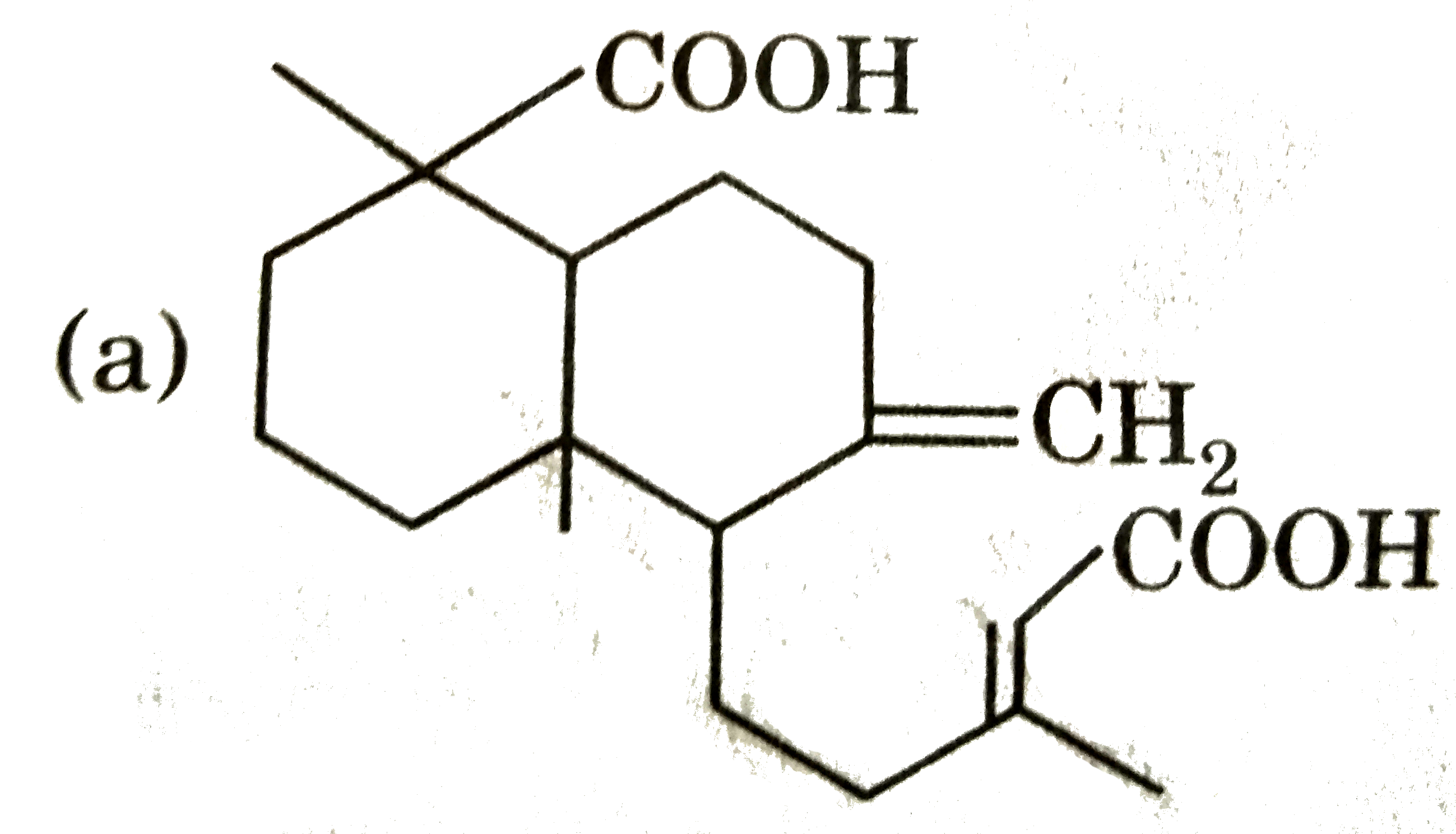
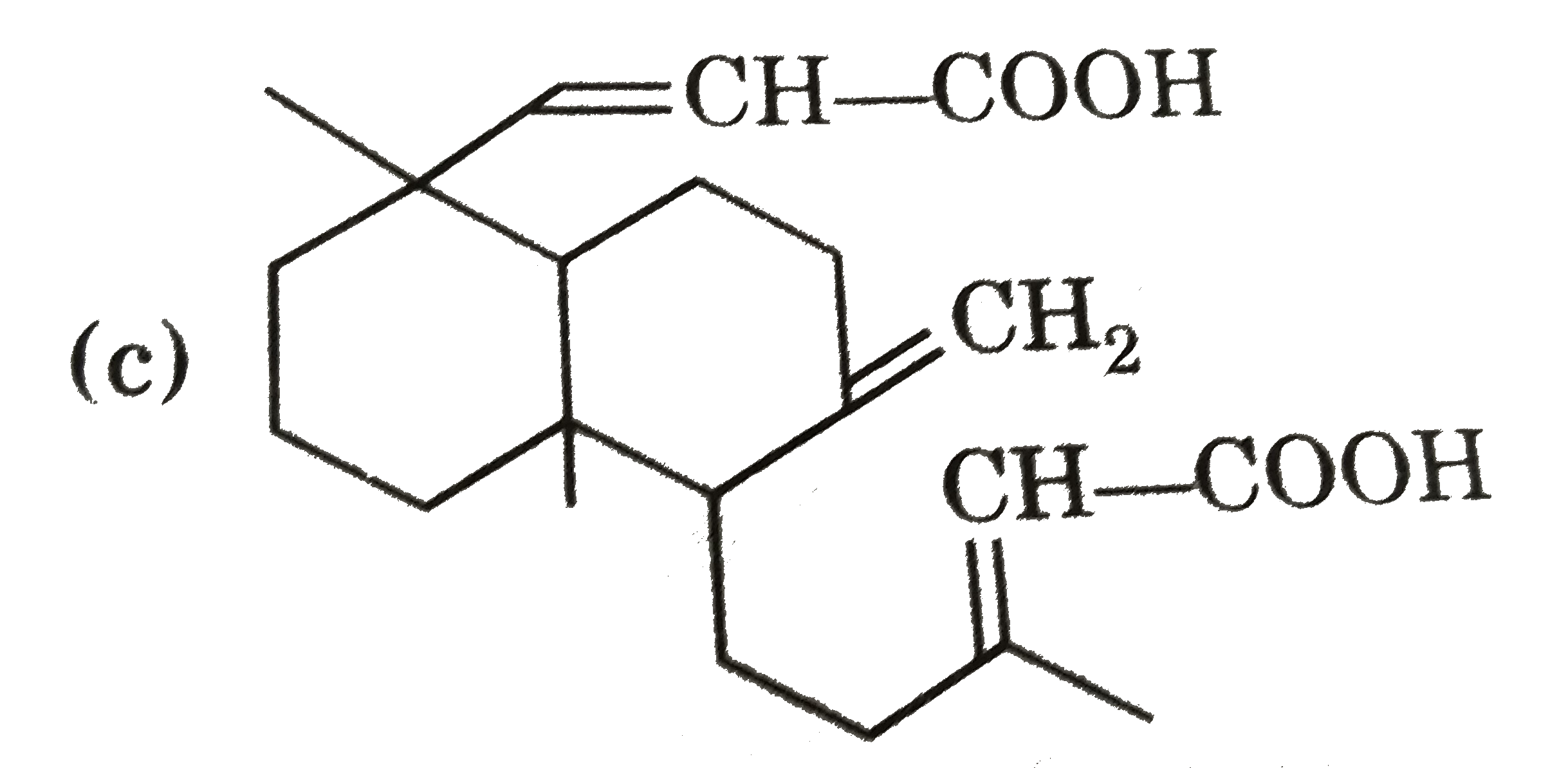
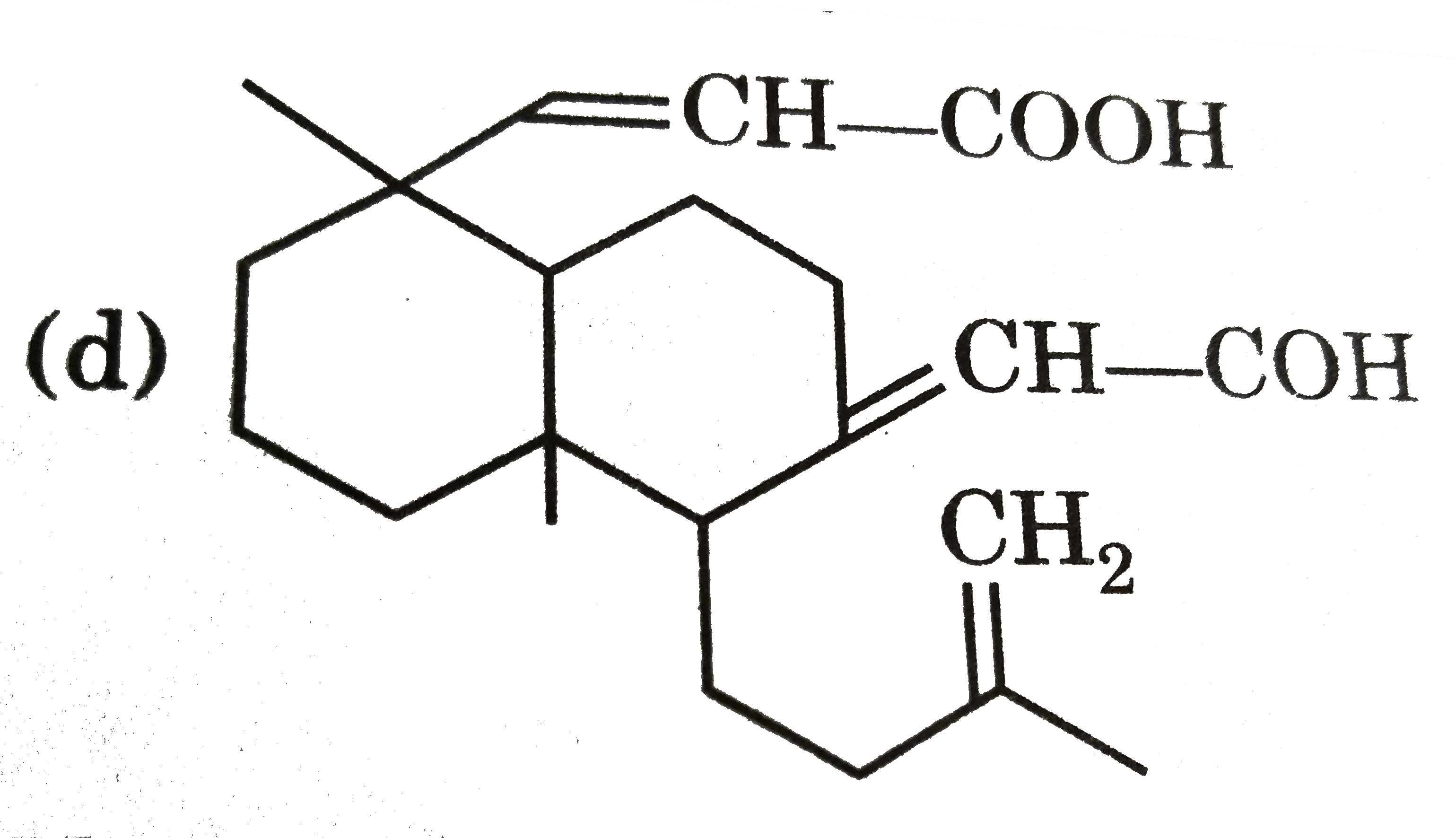
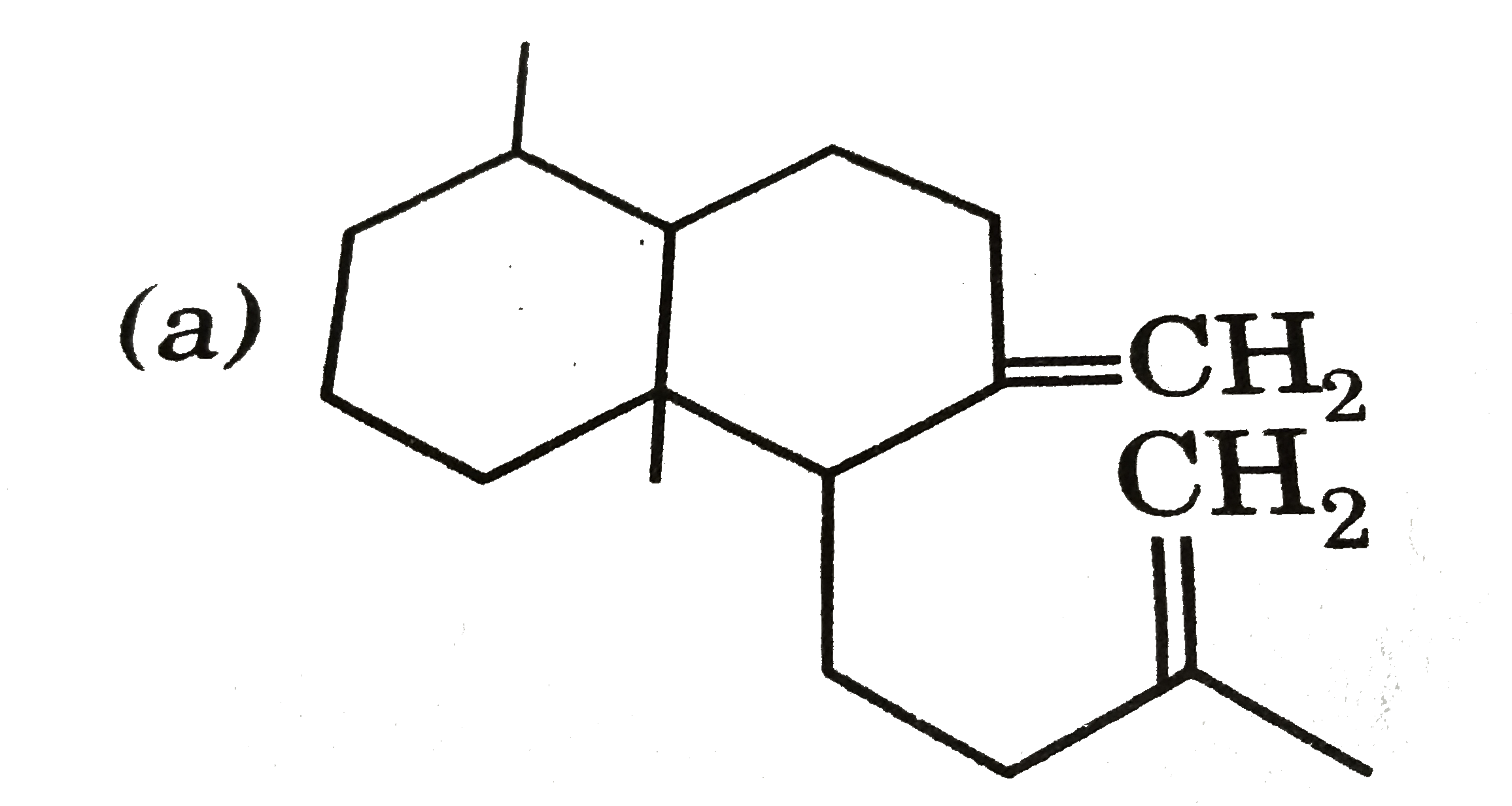
Ozonolysis of a compound Agathene dicarboxylic acid gives following compounds : on complete reduction by Na-EtOH Agathene dicarboxylic acid give hydrocarbon C_(20)H_(38) which have 5 chiral carbon in True statement about Agathene dicarboxylic acid is : |
|
Answer» It GIVES PPT. with Tollen's reagent. |
|
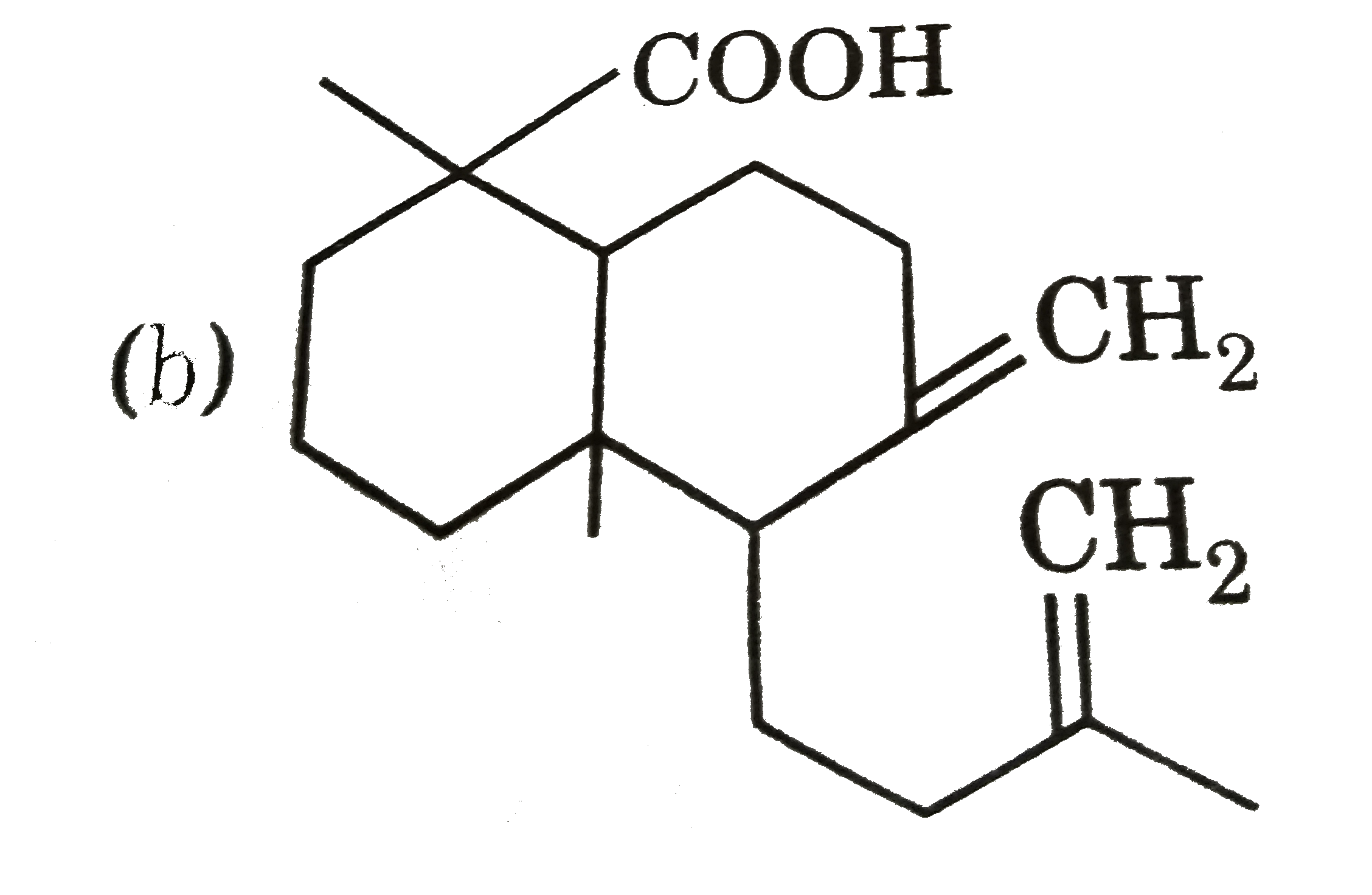
| 25. |
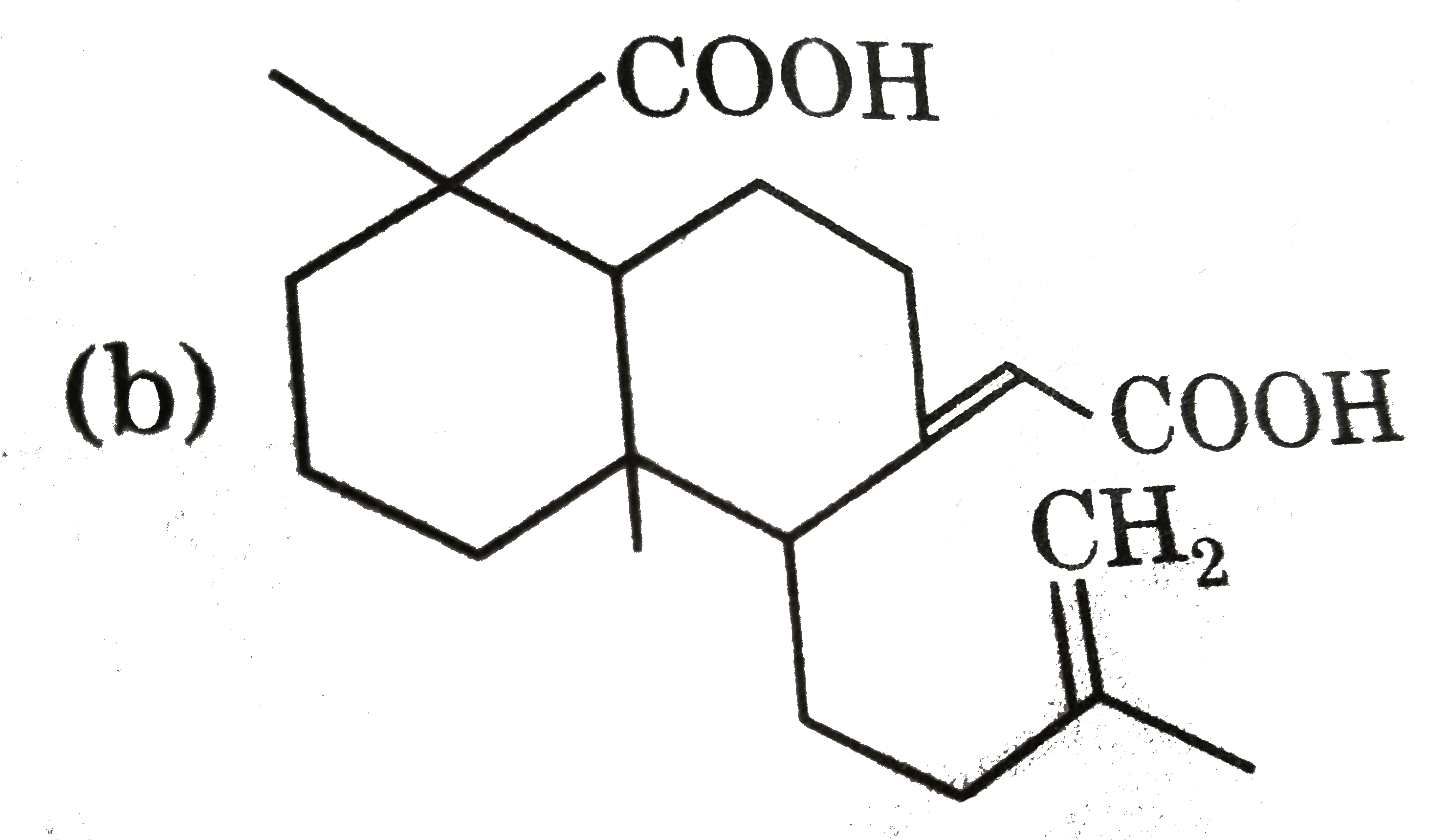
Ozonolysis of a compound Agathene dicarboxylic acid gives following compounds : on complete reduction by Na-EtOH Agathene dicarboxylic acid give hydrocarbon C_(20)H_(38) which have 5 chiral carbon in The structure of Agathene dicarboxylic acid is : |
|
Answer»
|
|
| 26. |
Ozonolysis of a compound Agathene dicarboxylic acid gives following compounds : on complete reduction by Na-EtOH Agathene dicarboxylic acid give hydrocarbon C_(20)H_(38) which have 5 chiral carbon in How many chiral carbon are present in agathene diocarboxylic acid? |
|
Answer» 2 |
|
| 27. |
Ozonolysis of a compound Agathene dicarboxylic acid gives following compounds : on complete reduction by Na-EtOH Agathene dicarboxylic acid give hydrocarbon C_(20)H_(38) which have 5 chiral carbon in Total stereoisomers therotically possible for Agathene dicarboxylic acid are: |
|
Answer» 16 |
|
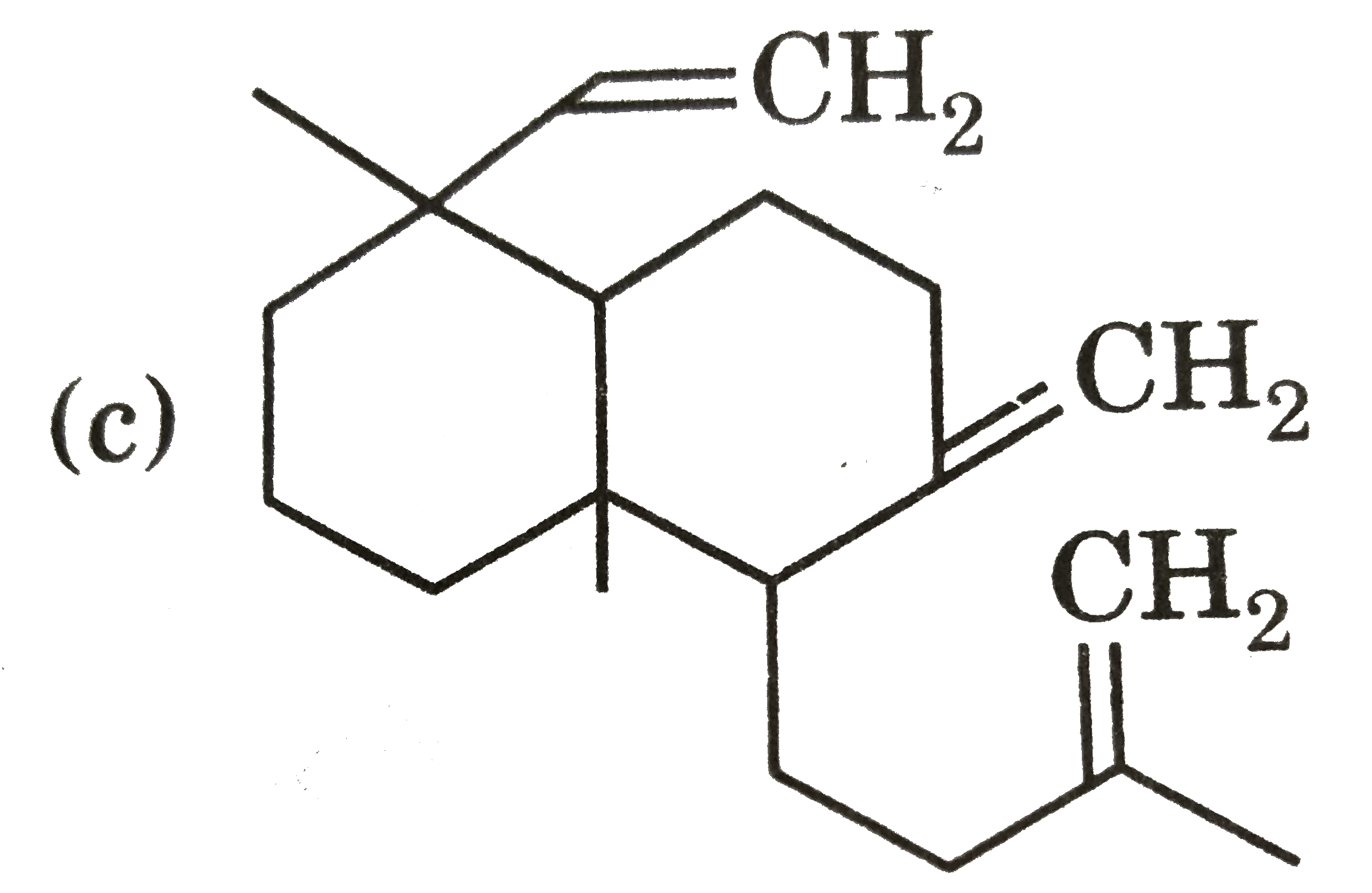
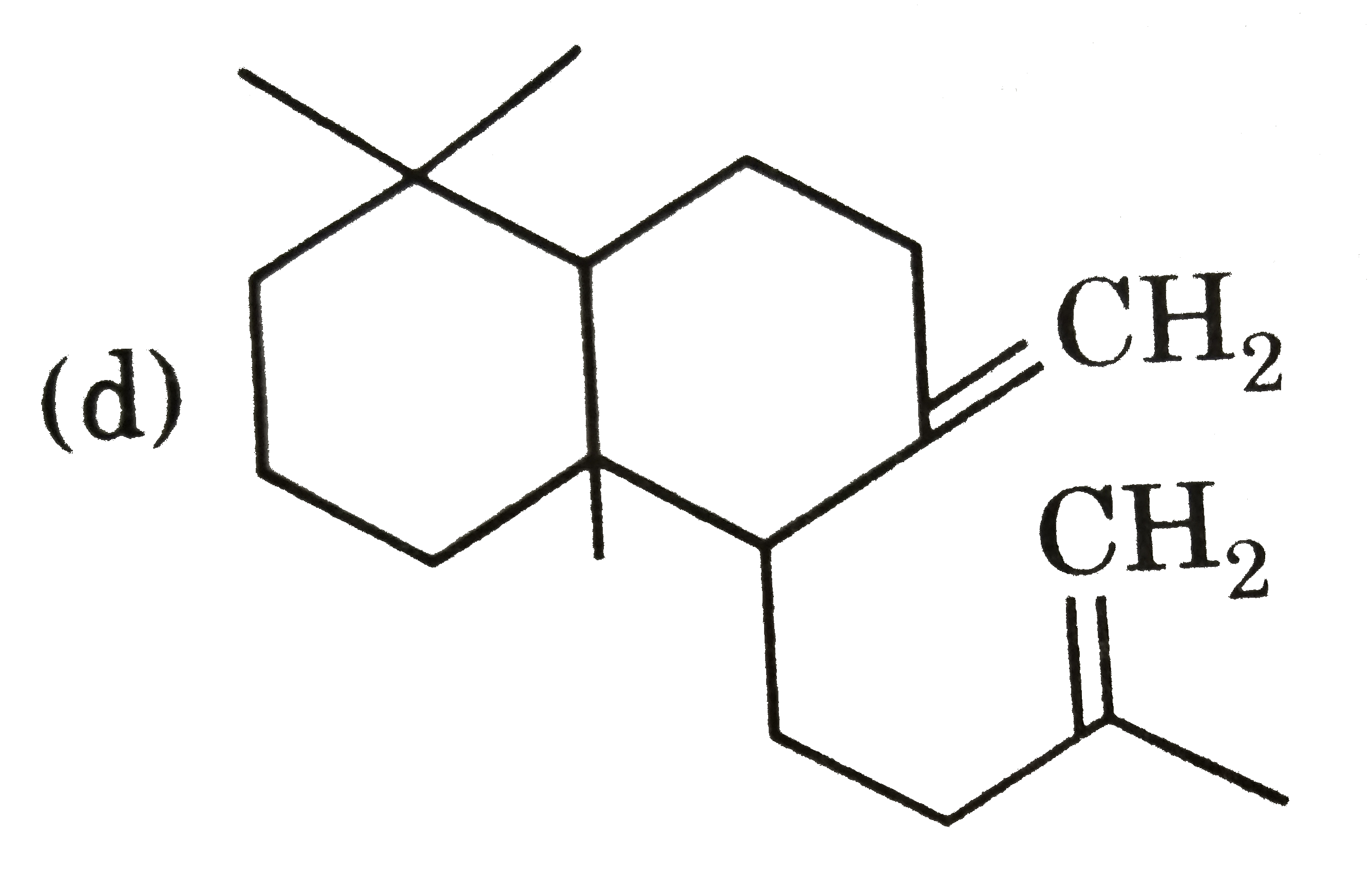
| 28. |
Ozonolysis of a compound Agathene dicarboxylic acid gives following compounds : on complete reduction by Na-EtOH Agathene dicarboxylic acid give hydrocarbon C_(20)H_(38) which have 5 chiral carbon in Structure of product formed when Agathene dicarboxylic acid is heated with soda lime is : |
|
Answer»
|
|
| 29. |
Ozonolysis of 2,3-Dimethyl-1-butene followed by reduction with zinc and water gives? |
|
Answer» Methanoic ACID and 3-methyl-2-butanonoe |
|
| 30. |
Ozonized oxygen can be prepared by the reaction of H_(2)O with |
|
Answer» `CONC. H_(2)SO_(4)` |
|
| 31. |
During ozonolysis of 1mole of benzene, number of moles of ozone consumed is |
Answer» 
|
|
| 32. |
Ozonization of water is carried out to remove |
|
Answer» BACTERIAL impurities |
|
| 33. |
Ozonised oxygen can be obtained from H_2O the action of |
|
Answer» CONC. `H_2SO_4` |
|
| 34. |
Ozonised oxygen can be obtained from H_(2)O by the action of |
|
Answer» CONC. `H_(2)SO_(4)` |
|
| 35. |
Ozonisation of water is carried to remove |
|
Answer» BACTERIAL IMPURITIES |
|
| 36. |
Ozonisation of water is carried out to remove |
|
Answer» BACTERIAL impurities |
|
| 37. |
Ozone with KI solution produces |
|
Answer» `Cl_(2)` |
|
| 38. |
Ozone when reacts with potassium iodide solution liberates certain product, which turns starch paper blue. The liberated substance is. . . . |
|
Answer» Oxygen |
|
| 39. |
Ozone with dry iodine give |
|
Answer» `I_(4)O_(4)` |
|
| 41. |
Ozone uses all oxygen atoms in the oxidation reaction with |
|
Answer» `SO_(2)` only |
|
| 42. |
Ozone turns tetramethyl base paper: |
|
Answer» Green |
|
| 43. |
Ozone turns benzidine paper: |
|
Answer» Violet |
|
| 44. |
Ozone turns benzidine paper : |
| Answer» Solution :`O_(3)` turns Benzidine paper to brown. | |
| 45. |
Ozone tarnishes silver metal. How many number of moles of O_(2) are evolved? |
|
Answer» |
|
| 46. |
Ozone readily dissolves in : |
|
Answer» `H_(2)O` |
|
| 47. |
Ozone tails mercury. The reaction is ……..of Hg: |
|
Answer» Reduction |
|
| 50. |
Ozone reacts with K_4[Fe(CN)_6] to form .......... |
| Answer» Solution :`6K_(4)[Fe(CN)_(6)] + O_(3) + 3H_(2)O to 6K_(3)[Fe(CN)_(6)] + 6KOH` | |