Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 3. |
P-P linkage is present in |
|
Answer» Pyrophosphoric acid |
|
| 4. |
P overset("Reduction")to Q overset("Dehydration")to R overset("Hydro-chlorination")to S What is P, Q, R, S? |
|
Answer» `P= CH_(3)COOH"" Q= CH_(3)CH_(2)OH` |
|
| 5. |
(P) overset(NaH)rarr (Q) uarr overset(CuSO_(4))rarr (R) darr black precipitate, (P) may be |
|
Answer» `SCl_(2)` |
|
| 6. |
[P] overset(Br_(2))to C_(2)H_(4)Br_(2) underset(NH_(3))overset(NaNH_(2))to [Q] underset(Hg^(2+), Delta)overset(20% H_(2)SO_(4))to [R] overset(Zn-Hg // HCl)to [S] The species P, Q, R and S respectively are |
|
Answer» ethene, ETHYNE, ethanal, ethane P is undergoing `Br_(2)` addition reaction and `Br_(2)` being used in 1 mol. `:.` P is `C_(2)H_(4)` or `CH_(2)=CH_(2)` `underset((P))(H_(2)C=CH_(2))+Br_(2) underset("Addition")overset("Anti")to underset(Br)underset(|)CH_(2)-overset(Br)overset(|)CH_(2) underset(underset("Elimination (twice)")(E_(2)))overset(overset(+)Naoverset(ddot)(barNH_(2)) // NH_(3))to underset((Q))(HC -= CH) underset(underset("HYDRATION reaction")(Hg^(2+), Delta))overset(20% H_(2)SO_(4))to CH_(2)=underset(OH)underset(|)CH overset("Tautomerism")HARR underset((R))(CH_(3)-underset(O)underset(||)C-H) underset(CONC. HCl)overset(Zn-Hg)to underset((S))(CH_(3)CH_(3))` |
|
| 7. |
Poverset(H_(2)//Pd-BaSO_(4))toQunderset((ii)dil.HCl)overset((I Con.NaOH))toR+S R and S form benzyl benzoate when treated with each other .Hence P is |
|
Answer» `C_(6)H_(5)CHO` 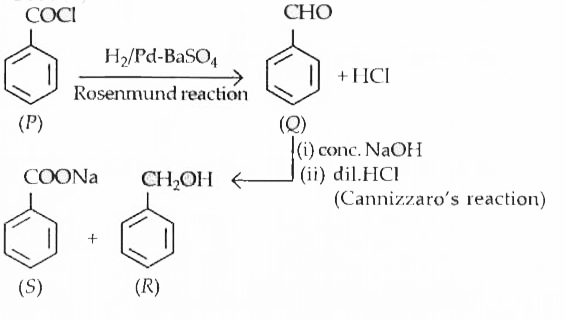 R + S form benzyl BENZOATE on reaction with each other. R + S form benzyl BENZOATE on reaction with each other.
|
|
| 8. |
P underset(2.H_3O^(+))overset(1. CH_3MgBr) to R overset(1. dil. NaOH)underset(2.Delta)to 4-methylpent -3-en-2-one P is |
|
Answer» `Ba(NO_(3))_(2)` |
|
| 9. |
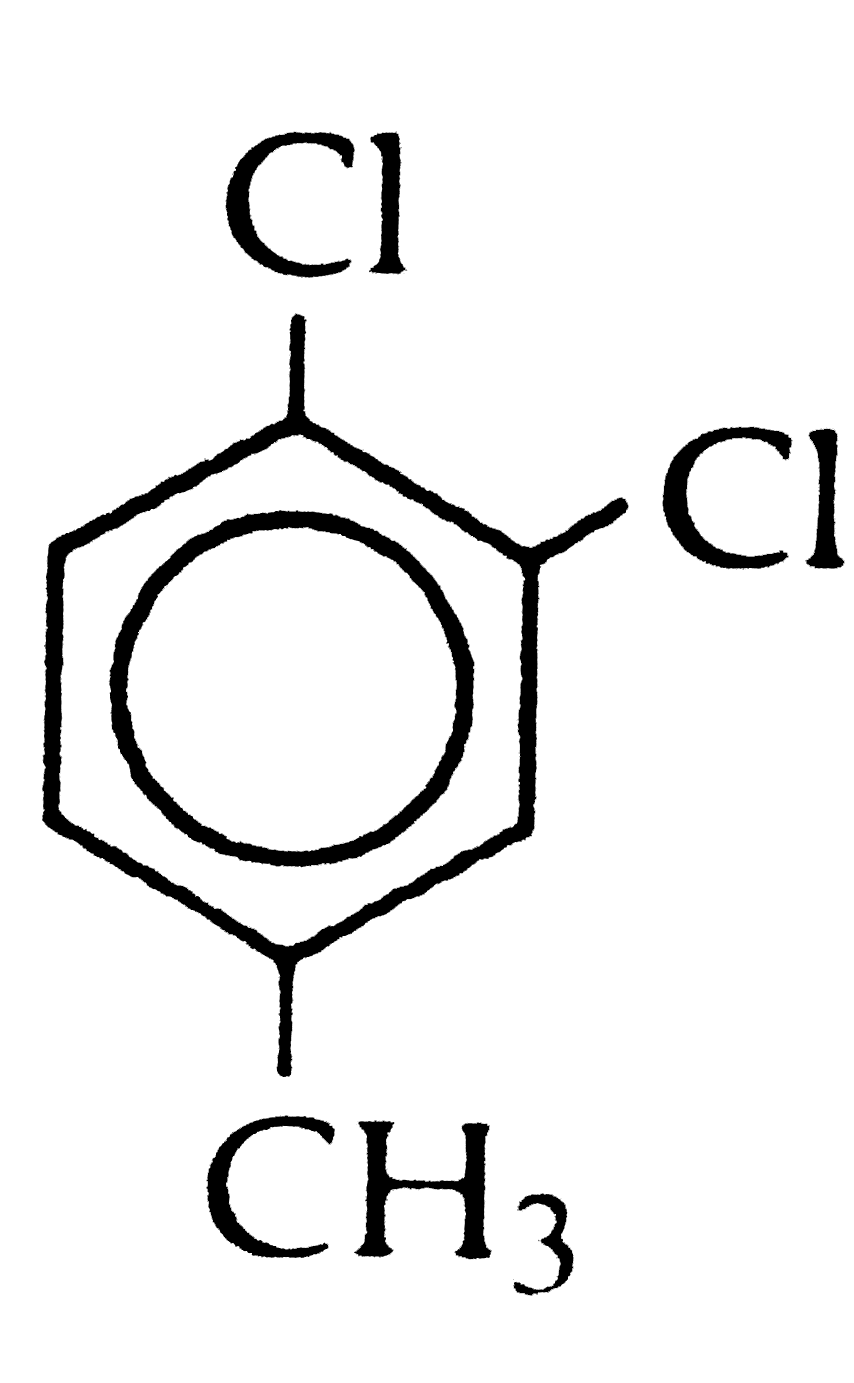
[P] on treatment with H_(2)CrO_(4)in acetone produced [Q]. [Q] compound upon heating give rise to Acetophenone. The compound [P] is: |
|
Answer»
|
|
| 10. |
P-O-P bond is present in |
|
Answer» <P>`H_(4)P_(2)O_(6)` |
|
| 11. |
P-O-P bond is present in : |
|
Answer» METAPHOSPHORIC acid |
|
| 12. |
p-Nitrotoluene on further nitration gives |
|
Answer»
|
|
| 13. |
p-nitrotoluene can be obtained by: |
|
Answer»
|
|
| 14. |
p. Nitrophenol is more acidic than phenol. Explain why? |
Answer» SOLUTION :p-Nitrophenol is more ACIDIC than phenol 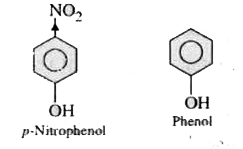 Phenal p-Nitrophenol In case of p-nitrophenol DUE to I EFFECT of -`NO_(2)` group the loss of H-atom as proton from-O-H group is facilitated as compared to phenol. |
|
| 15. |
p-Nitrophenol is having lower pK_(a) value than phenol because |
|
Answer» phenol is more acidic than p-nitro phenol. |
|
| 16. |
p-nitrophenol and o-nitrophenol are separatedby |
|
Answer» Crystallisation |
|
| 17. |
p- Nitrophenol is a stronger acid than phenol because nitro group is : |
|
Answer» ELECTRON DONATING |
|
| 18. |
p-nitrophenol and o-nitrophenol are separated by : |
|
Answer» distillation 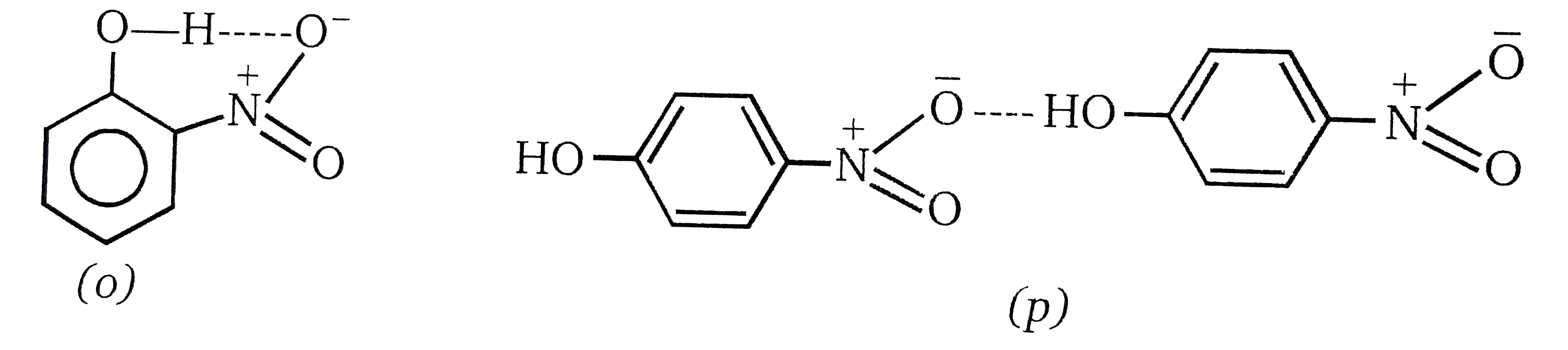 In the o-compound the hydrogen boiling is intramolecular, and so the molecule BEHAVES as a 'single unit'. Hydrogen bonding in the p-compound is intermolecular. Thus, we are DEALING with associated molecules, which behave as a large molecule with high that of the monomer). Since we have assumed that the o-compound has the type of hydrogen bonding SHOWN, this PRESUMABLY is also possible for the p-compound. On the other hand, hydrogen bonding between hydrozyl groups in the p-compound is possible. |
|
| 19. |
p-Nitrobenzoic acid has higher K_(a) value than benzoic acid. Give reasons. |
|
Answer» Solution :Higher the `K_(a)`, stronger is the acid. Thus, p-nitrobenzoic acid is a stronger acid than benzoic acid. This is due to the following two reasons: (i) due to -I and -R-effect of the `-NO_(2)` group, the ELECTRON density in the O-H BOND decreases. as a result, O-H bond becomes weak and hence p-nitrobenzoic acid more easily LOSES a PROTON than benzoic acid 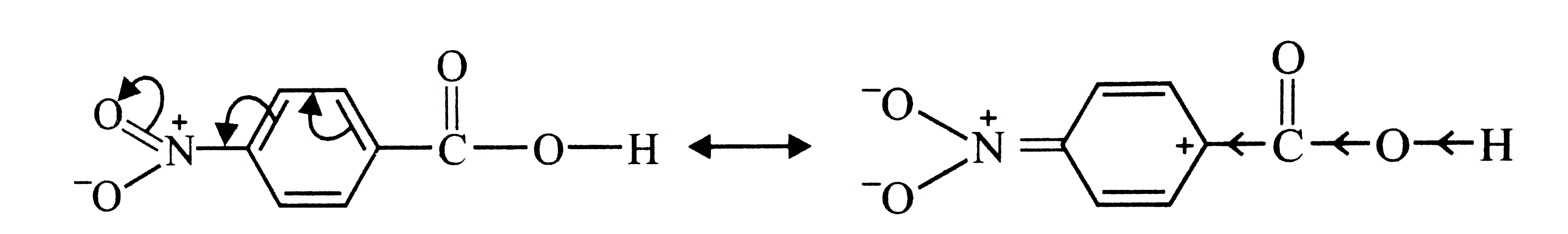 . . (ii) Due to -I and -R-effect of the `NO_(2)` group, dispersal of the -ve charge OCCURS and hence p-nitrobenzoate ion becomes more stable than benzoate ion. 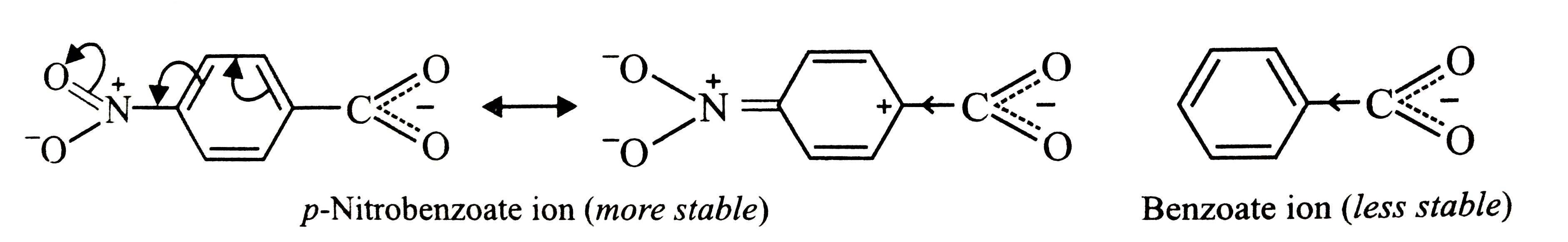 . .
|
|
| 20. |
p-Nitroaniline can be obtained by |
|
Answer»
|
|
| 21. |
p-nitro toluene convert p - nitro benzaldehyde by using |
|
Answer» `KOH + KMnO_(4)` |
|
| 22. |
p-Nitro aniline can be obtained by |
|
Answer»
 Here, `-SO_(3)H` GROUP is knocked out. D choice represnts the protective NITRATION of ANILINE |
|
| 23. |
p-nitro chlorobenzene undergoes nucleophilic substitution faster than chlorobenzene. |
Answer» SOLUTION : p-nitro chlorobenzene undergoes nucleophilic substitution FASTER than chlorobenzene. This is because the anion formed after the ATTACK of nucleophile is stabilised more in case of p-nitro chlorobenzene due to electron withdrawing effect of - `NO_(2)`group. |
|
| 24. |
p - Nitro phenol is more acidic than phenol. Explain. |
Answer» Solution :This because `-NO_(2)` GROUP has `-I` EFFECT.  As a RESULT of it : (a) the LOSS of H atom as proton is facilitated. (b) The phenoxide ion formed after the loss of proton gets stabilised. |
|
| 25. |
p-methoxybenzyl bromide reacts faster than p-nitrobenzyl bromide with ethanol to form an ether product. Explain why? |
| Answer» SOLUTION :It is because of the DIFFERENCE in the STABILITY of carbocations formed in two CASES, | |
| 26. |
p-methoxylbenzyl bromide reacts faster than p-nitrobenzyl bromide with ethanol to form an ether product. Explain, why ? |
Answer» Solution : METHOXY GROUP is ELECTRON releasing, therefore, it stabilises carbocation formed by p-methoxybenzyl BROMIDE whereas nitro group being electron withdrawing destabilises carbocation formed by p-nitro BENZYL bromide. That is why p-methoxybenzyl bromide is more reactive than p-nitrobenzyl bromide. |
|
| 27. |
Pi - molecular orbitals can result from the overlapping of: |
|
Answer» p-orbitals |
|
| 28. |
p = K_H. xis the statement of: |
|
Answer» Avogadro's LAW. |
|
| 29. |
P is the probability of finding the 1s electron of hydrogen atom in a spherical shell of infinitesimal thickness, dr at a distanc, r from the nucleus. The volume of this shell is 4pi r^(2) dr. The qualitative sketch of the dependence of P on r is |
|
Answer»
|
|
| 30. |
P is an alcohl which on heating with Al_(2)O_(3) forms an alkene Q.Q on ozonolysis produces R and S. When the mixture of R and S is heated with NaOH, a redox reaction takes place and a mixture of an acid salt and alcohol is formed. The compound (Q) is : |
|
Answer» `CH_(3)-OVERSET(CH_(3))overset(|)(C)=CH-CH_(3)` |
|
| 31. |
P is an alcohl which on heating with Al_(2)O_(3) forms an alkene Q.Q on ozonolysis produces R and S. When the mixture of R and S is heated with NaOH, a redox reaction takes place and a mixture of an acid salt and alcohol is formed. The compounds R and S are : |
|
Answer» `CH_(3)-OVERSET(CH_(3))overset(|)(C)O, CH_(2)=O` |
|
| 32. |
P is an alcohl which on heating with Al_(2)O_(3) forms an alkene Q.Q on ozonolysis produces R and S. When the mixture of R and S is heated with NaOH, a redox reaction takes place and a mixture of an acid salt and alcohol is formed. The alcohol (P) is obtained by : |
|
Answer» `H_(3)C-underset(O)underset(||)(C)-CH_(2)-CH_(3)+CH_(3)MGBR` |
|
| 33. |
p-hydroxy azobenzene is formed by the reaction : |
|
Answer» HOFMANN rearrangement |
|
| 34. |
p-Dichorobenzene has higher melting point and lowerr solubility than those of o- and m-isomers. Discuss. |
| Answer» Solution :The p-isomer being more symmetrical fits closely in the crystal lattice and thus has stornger INTERMOLECULAR forces of attraction than those of o- and m-isomers. Since during melting or DISSOLUTION, the crystal lattice breaks, therfore, a larger amount of ENERGY is needed to melt or dissolve the p-isomer than the corresponding o-and m-isomers. in other words, the melting POINT of the p-isomer is higher and it ssolubility lower than the corresponding o- and -isomers. | |
| 35. |
p-dichlorobenzene has highest m.p. than those of ortho and m-isomers? |
| Answer» Solution :p-dichlorobenzene is SYMMETRICAL, FITS into crystal lattice more readily and has HIGHER MELTING POINT | |
| 36. |
p-Dichlorobenzene has higherm.p. and lower solubility than those ofo -and m-isomers. Discuss. |
| Answer» SOLUTION :The p-isomer being more symmetrical fits closely in the CRYSTAL LATTICE. It has stronger intermolecular forces of attraction than those ofo - and m-isomers. During melting or dissolution, the crystal lattice BREAKS, therefore, a LARGER amount of energy is needed to melt or dissolve the p-isomer than the correspondingo - and m-isomers. In other words, the melting point of the p-isomer is higher and its solubility lower than the correspondingo - and m- isomers. | |
| 37. |
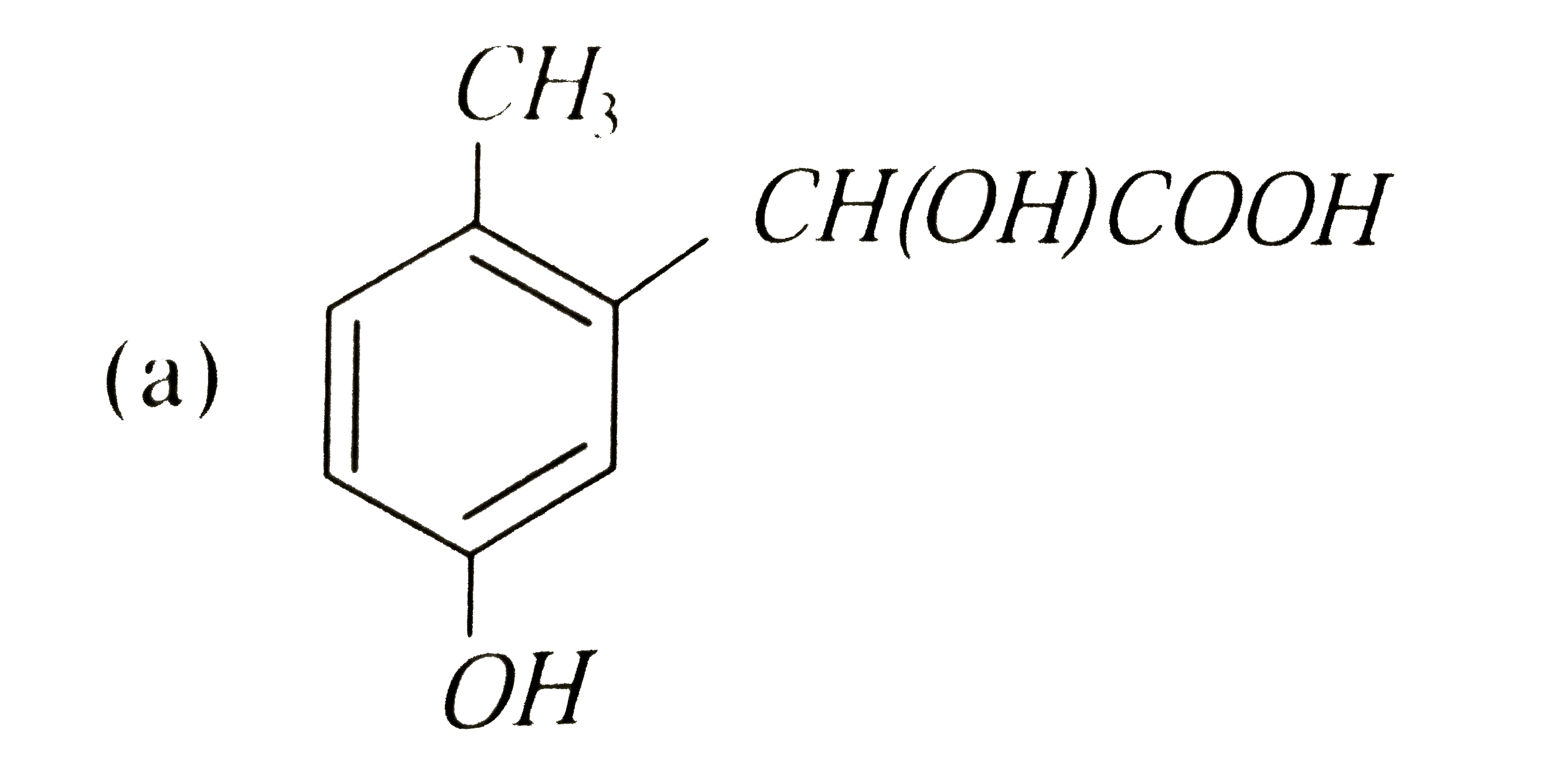
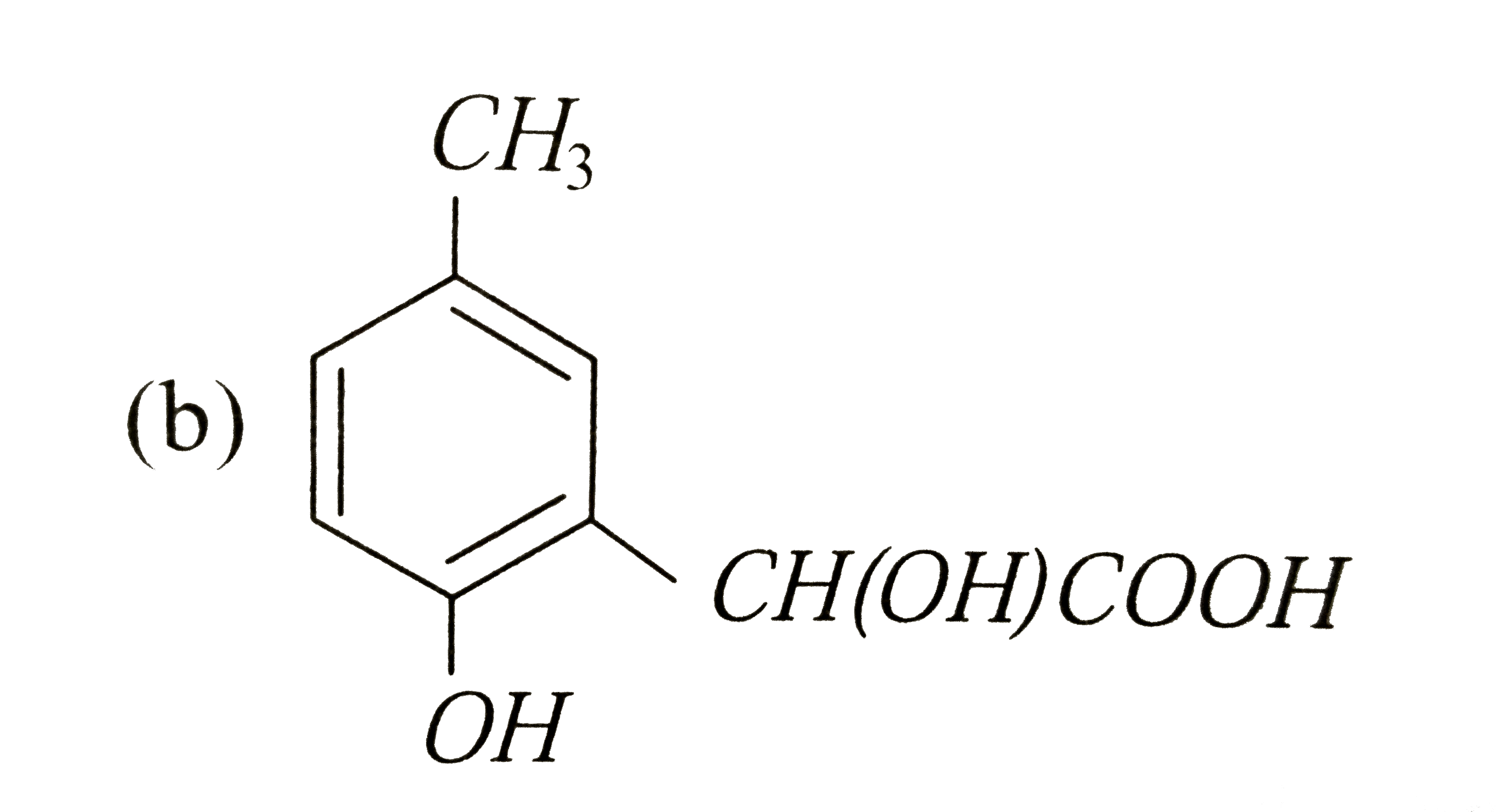
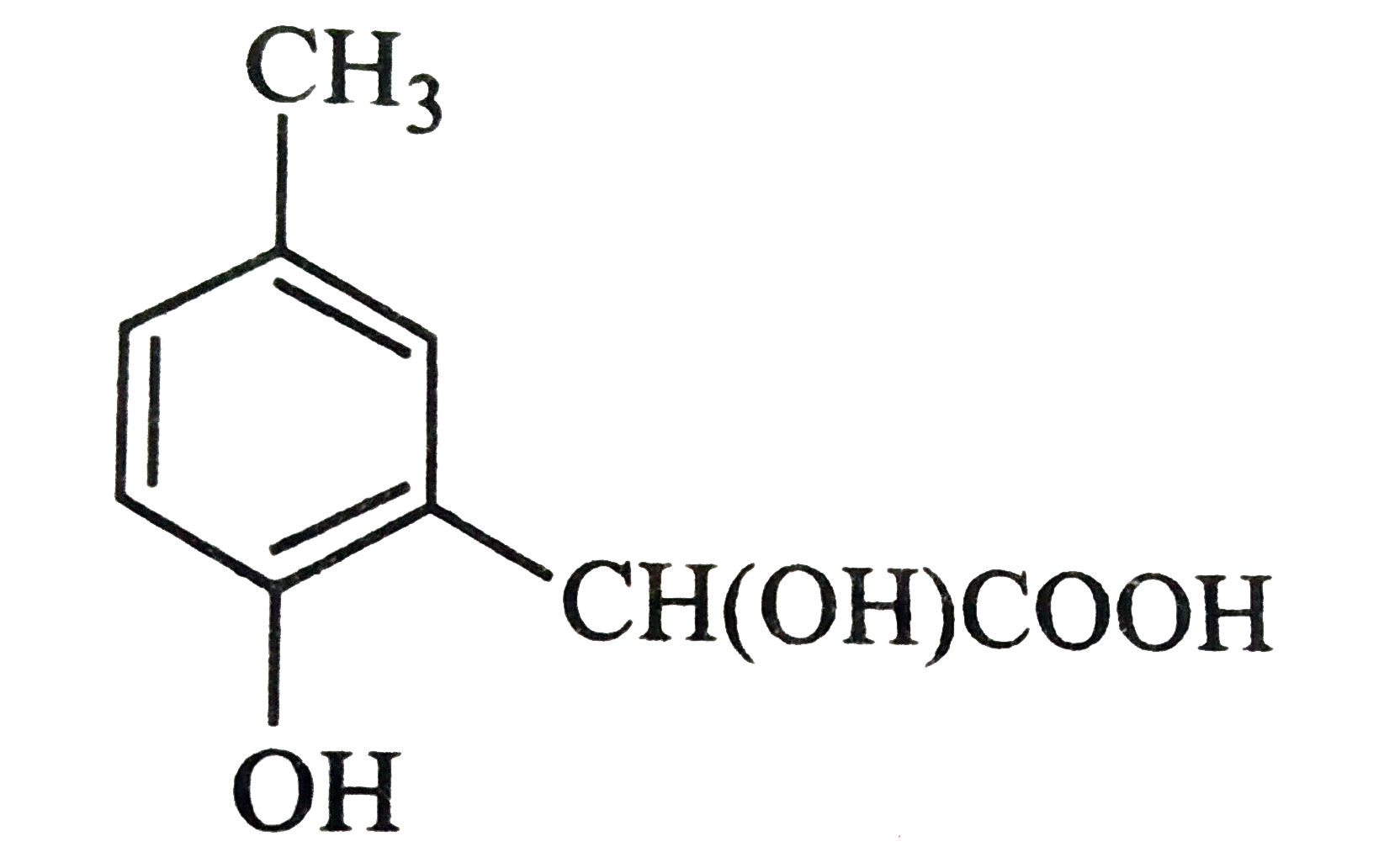
p-cresol reacts with chloroform in alkaline medium to give the compound A which adds hydrogen cyanide to form, the compound B. The latter on acidic hydrolysis gives chiral carboxylic acid. The structure of the carboxylic acid is |
|
Answer»
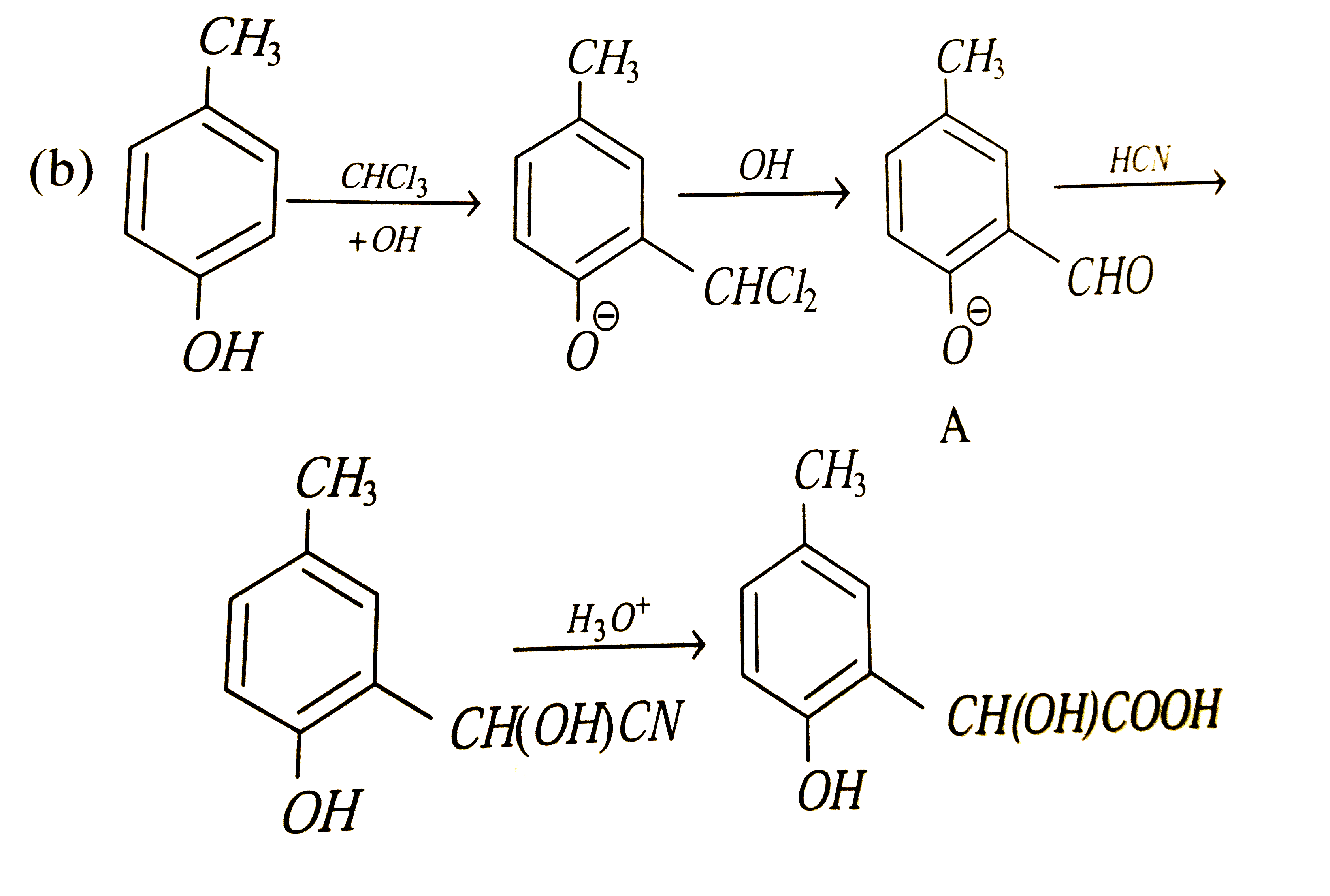
|
|
| 38. |
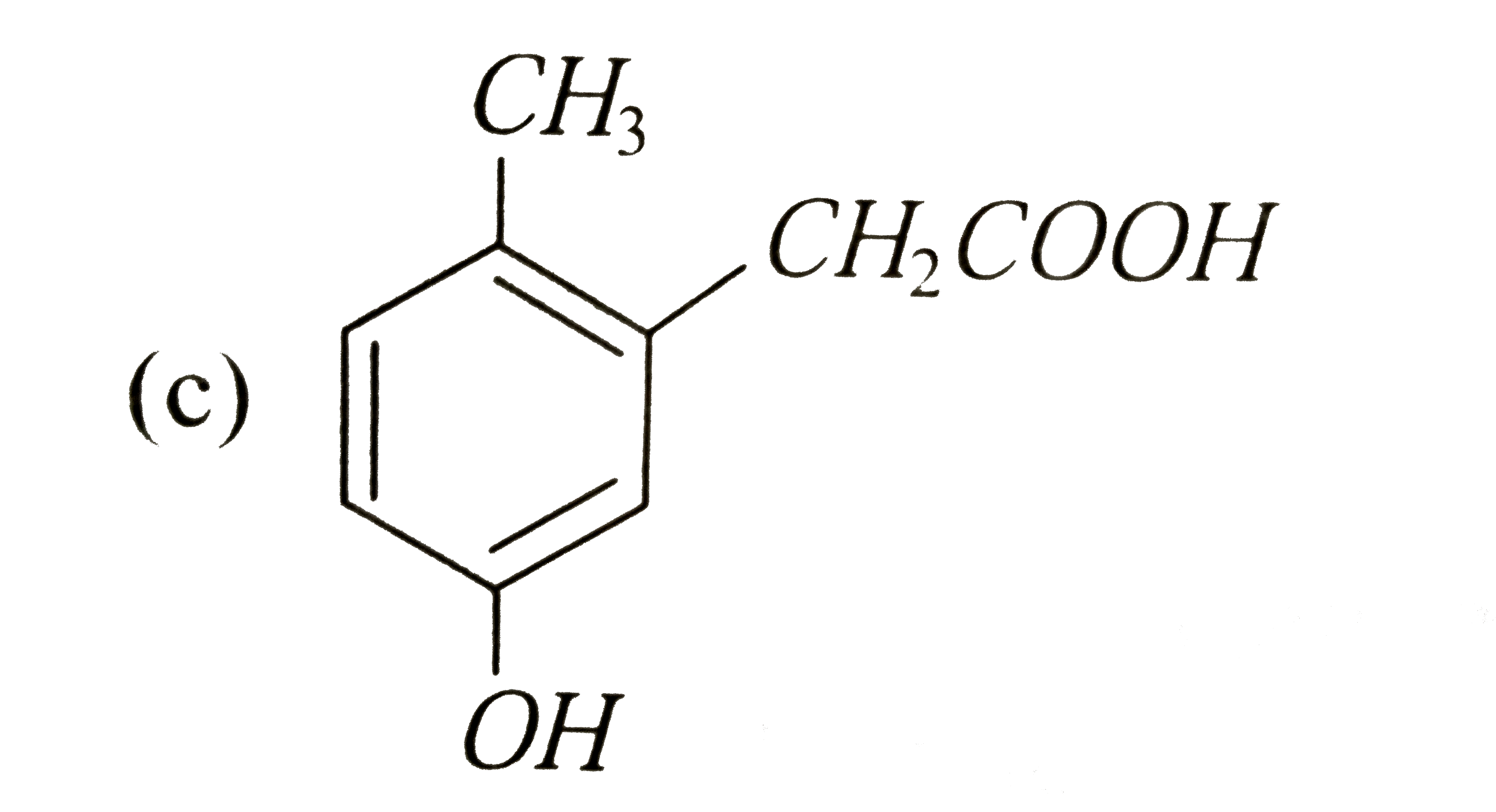
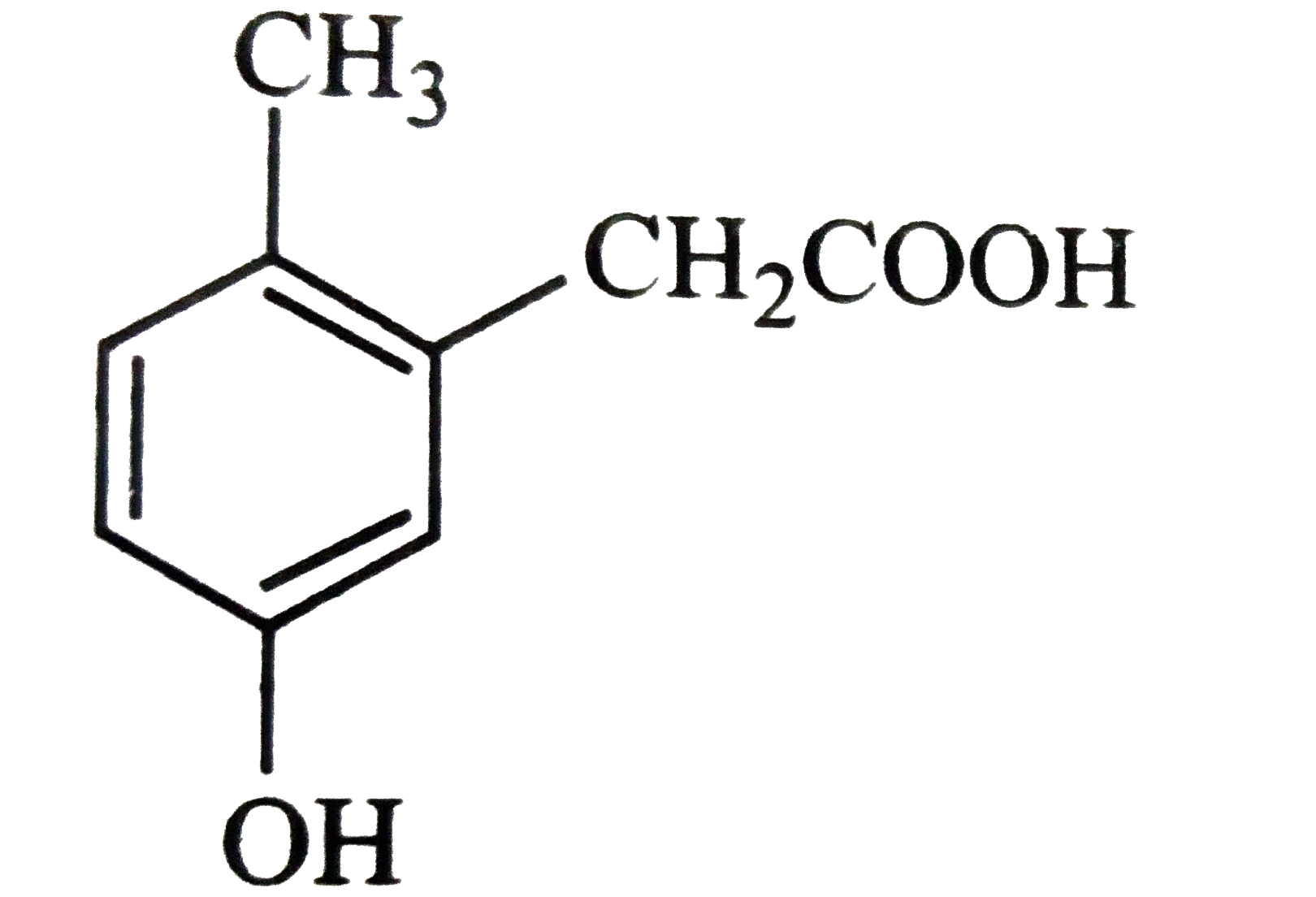
p-Cresol reacts with chloroform in alkaline medium to give the compound A which adds hydrogen cyanide to form the compound B. the latter on acidic hydrolysis gives chiral carboxylic acid. The structure of the carboxylic acids is |
|
Answer»
|
|
| 39. |
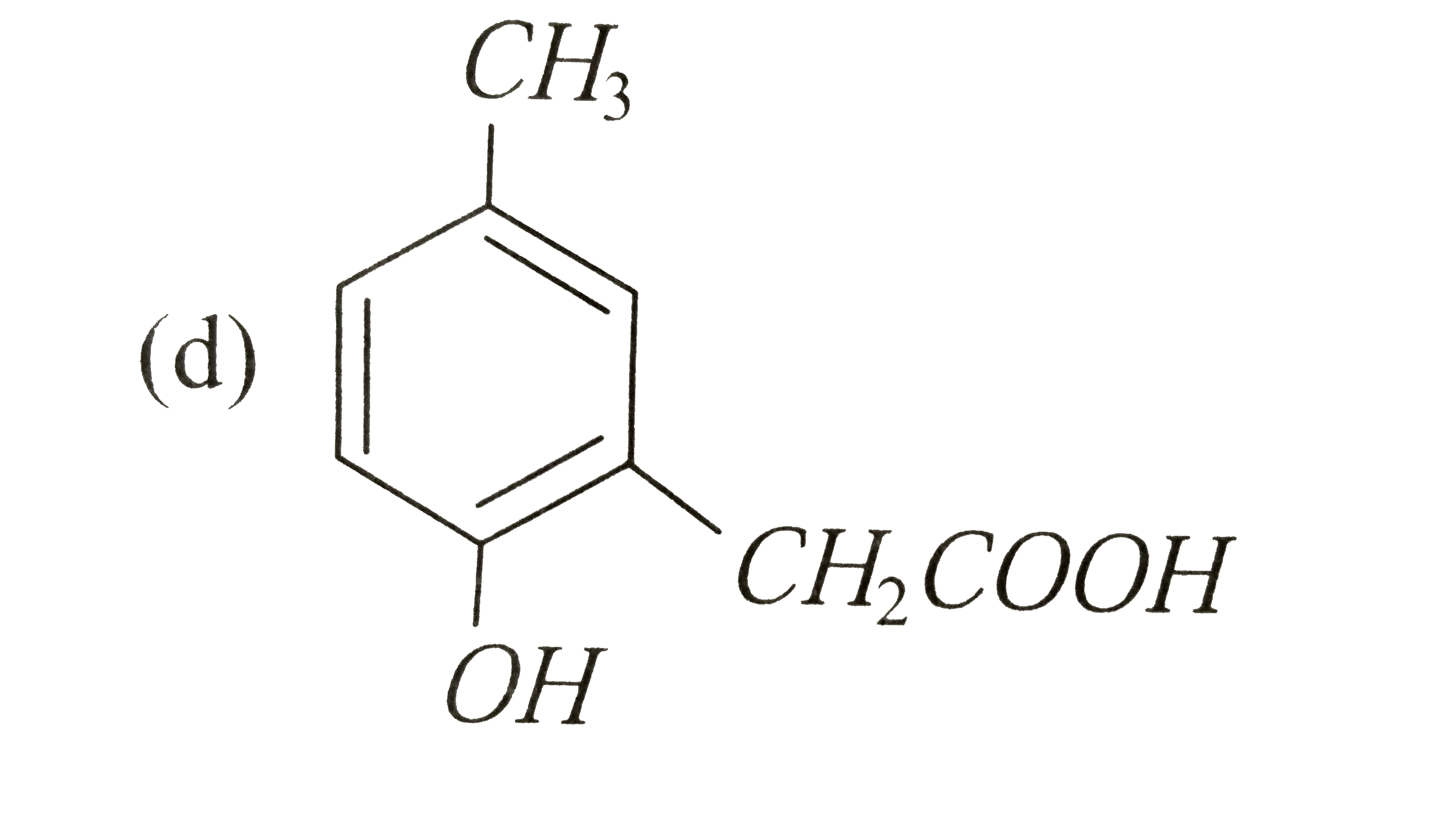
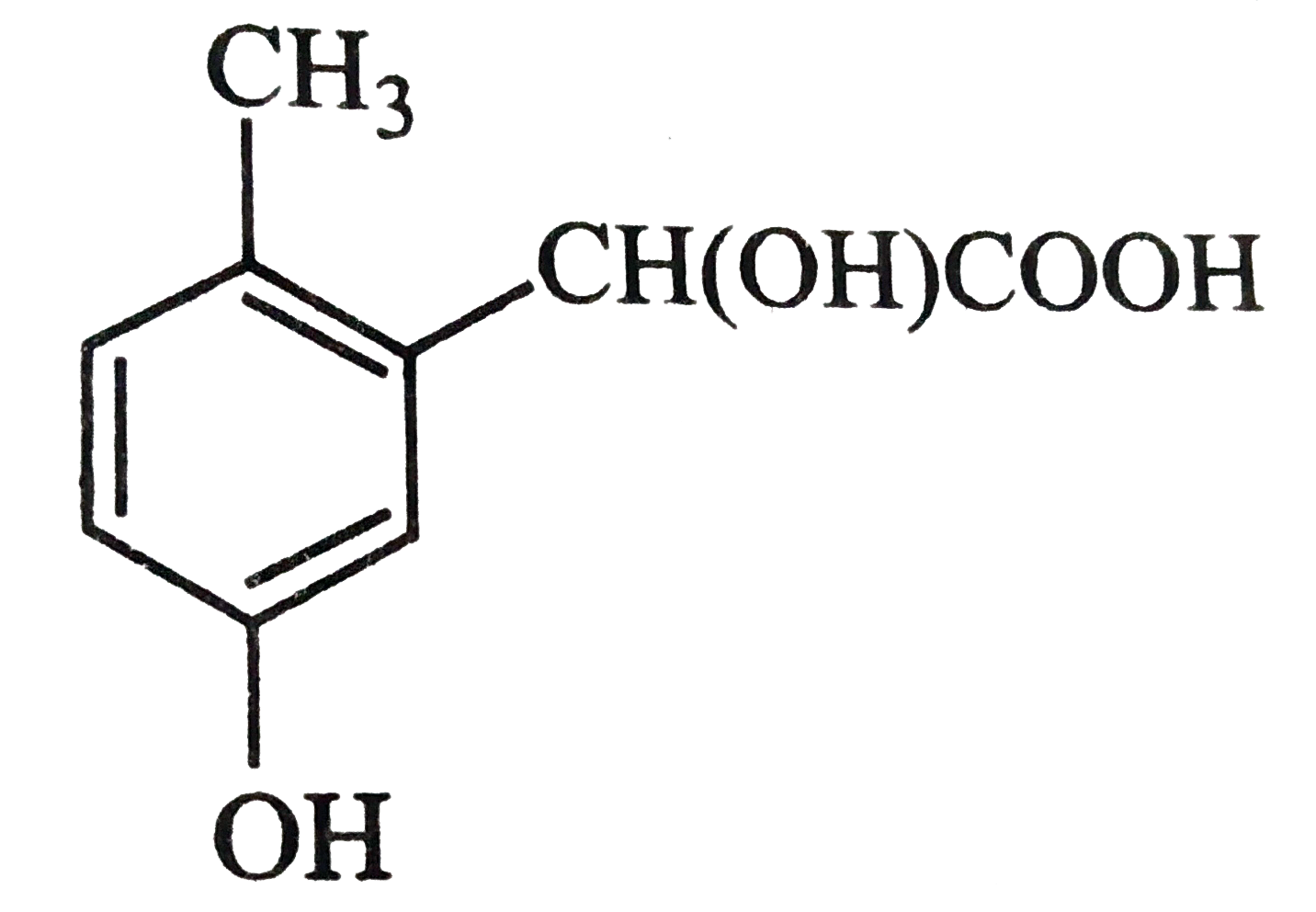
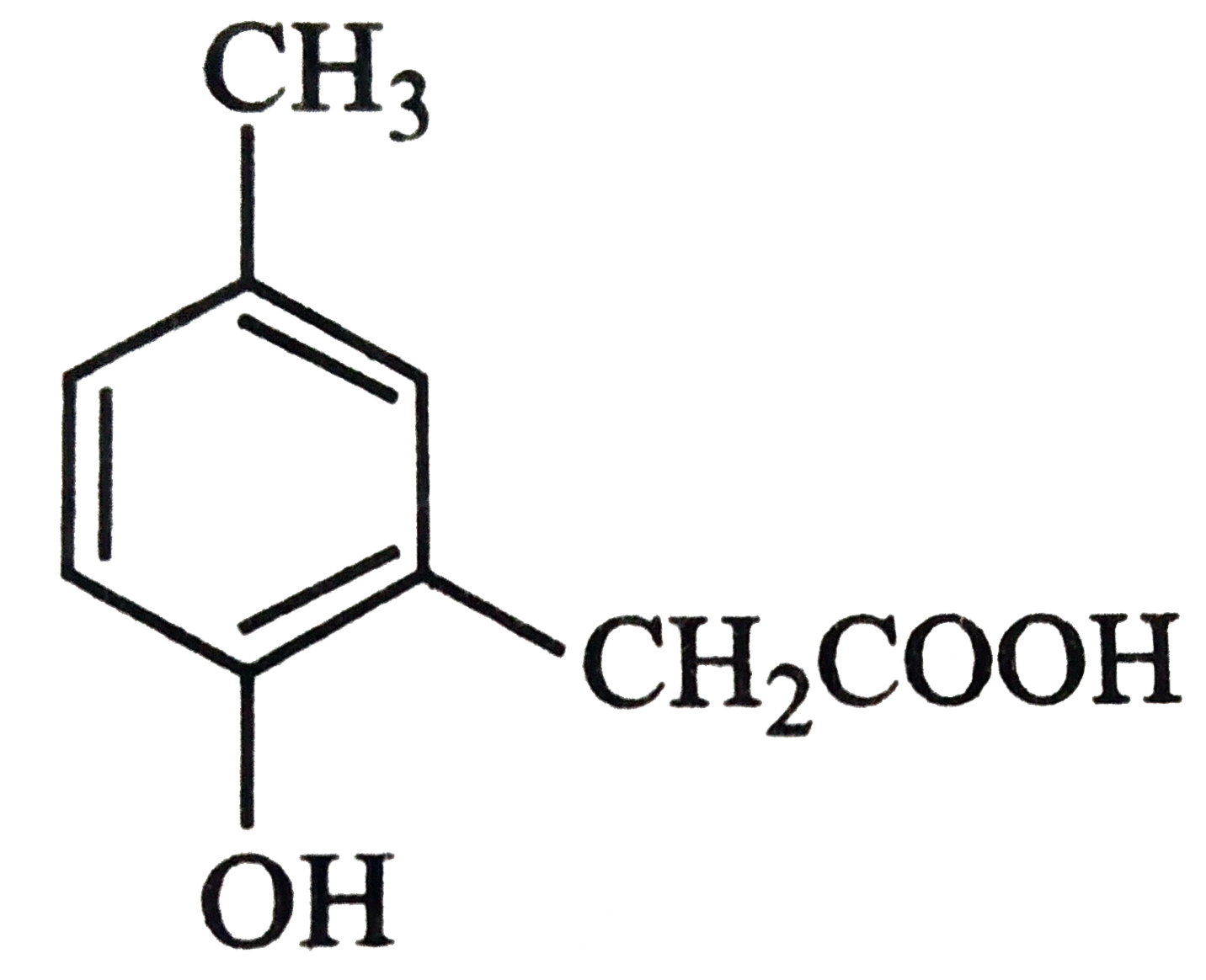
p-cresol reacts with chloroform in alkaline medium to give the compound A which adds hydrogen cyanide to form, the 29. compound B. The latter on acidic hydrolysis gives chiral carboxylic acid. The structure of the carboxylic acid is |
|
Answer»
 ` (##NEET_CHE_DPP_C25_E01_022_S02.png" width="80%"> |
|
| 40. |
p-chlorotoluene on chlorination in the presence fo FeCl_(3) gives |
|
Answer»
|
|
| 41. |
p-Cl-C_(6)H_(4)NH_(2)" and "PhNH_(3)""^(+)Cl^(-) can be distinguished by |
|
Answer» `NAOH` |
|
| 42. |
p-Chloroaniline and anilinium hydrochloride can be distinguished by |
|
Answer» SANDMEYER REACTION |
|
| 43. |
p-chloroaniline and anilinium hydrochloride can be distinguished by : |
|
Answer» SANDMEYER reaction |
|
| 44. |
p- chloroaniline and aniline hydrochloride can be distinguished by using |
|
Answer» CARBYLAMINE reaction 
|
|
| 45. |
p-chloroamine and anilinium hydrochloride can be distinguished by |
|
Answer» SANDMEYER REACTION |
|
| 46. |
p block elements of 6^(th) period are represented as :- |
|
Answer» `[XE] 4F^(14) 5d^(10)6p^(1-6)` |
|
| 47. |
P-benzoquinone is obtained from phenol by |
|
Answer» reduction |
|
| 48. |
p- anisidine can be represented by the forumula |
|
Answer»
|
|
| 49. |
P and Q are isomers of dicarboxyhc acid C_(4)H4O_(4) .Both decolourize Br_(2)//H_(2)O On heating, P forms the cyclic anhydride. Upon treatment with dilute alkaline KMnO_(4), P ass well as Q could produce one or more than one from S. T and U. Compounds formed form P and Q are, respectively. |
|
Answer» OPTICALLY ACTIVE s and optically active pair (T,U) |
|
| 50. |
P and Q are ideal gases which do not react with each other. The mass of one mole of P is four times that of Q. At STP, which of the following are true? |
|
Answer» <P> The. average K.E of MOLECULES of P is EQUAL to that of Q |
|























 WIDTH="30%">
WIDTH="30%">