Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Oxidation states of the metal in the minerals haematite and magnetite, respectively, are |
|
Answer» II, III in HAEMATITE and III in magnetite (ii) Magnetite `(Fe_(3)O_(4))` is an EQUIMOLAR MIXTURE of FeO and `Fe_(2)O_(3)`. Oxidation state of Fe in FeO is II. Oxidation state of Fe in `Fe_(2)O_(3)` is III. |
|
| 2. |
Oxidation states of P in H_(4)P_(2)O_(5), H_(4)P_(2)O_(6) and H_(4)P_(2_)O_(7), respectively are |
|
Answer» <P>`+3,+5` and +4 LET the oxidation state of P in the GIVEN compounds is x. In `H_(4)P_(2)O_(5)` `(+1)xx4+2xx x+(-2)xx5=0` `4+2X - 10 = 0` `2x = 6` `:. x = +3` In `H_(4)P_(2)O_(6)` `(+1) xx4+2xx x + (-2)xx6=0` `4+2x-12=0` `2x=8` `:. x = +4` In `H_(4)P_(2)O_(7)` `(+1)xx4+2xx x + (-2)xx7=0` `4+2x-14=0` `2x=10` `:. x = +5` Thus the oxidation states of `P` in `H_(4)P_(2)O_(5)`. `H_(4)P_(2)O_(6)` and `H_(4)P_(2)O_(7)` are `+3,+4` and `+5` respectively. |
|
| 3. |
Oxidation states of P in H_(4)P_(2)O_(5), H_(4)P_(2)O_(6), H_(4)P_(2)O_(7) are respectively ________ |
|
Answer» `+3,+4,+5` |
|
| 4. |
Oxidation states of carbon atoms in methane and ethane are : |
| Answer» Answer :D | |
| 5. |
Oxidation state/s exhibited by Ce is / are |
|
Answer» `+2` only |
|
| 7. |
Oxidation state of the metal in the minerals haematite and magnetite, respectively are |
|
Answer» II, III in HAEMATITE and III in magnetite |
|
| 8. |
Oxidation state of the metal in the minerals haematite and magnetite respectively are |
|
Answer» II, III in HAEMATITE and III in magnetite |
|
| 10. |
Oxidation state of S in H_(2)SO_(5) and H_(2)S_(2)O_(8) respectively are |
|
Answer» `+6, +6` |
|
| 12. |
Oxidation state of phosphorus is least in |
|
Answer» HYPOPHOSPHORIC ACID |
|
| 13. |
Oxidation state of P in H_(4)P_(2)O_(5), H_(4)P_(2)O_(6), H_(4)P_(2)O_(7) are respectively |
|
Answer» `+3, +5, +4` |
|
| 14. |
Oxidation state of oxygen is + 1 in .......... And +2 in ,....... |
| Answer» SOLUTION :`O_2F_2,OF_2` | |
| 15. |
Oxidation state of oxygen in super oxide is |
|
Answer» O |
|
| 18. |
Oxidation state of oxygen atom in potassium superoxide (KO_2) is |
| Answer» ANSWER :A | |
| 19. |
Oxidation state of nitrogen is incorrectly given for |
|
Answer» `{:(,"COMPOUND","Oxidation State"),((a),Mg_(3)N_(2),""-3):}` |
|
| 20. |
Oxidation state of nitrogen is not correctly given for the compound |
|
Answer» `[CO(NH_3)_5Cl]Cl_2` |
|
| 22. |
Oxidation state of mercury in amalgams is: |
|
Answer» ZERO |
|
| 23. |
Oxidation state of Iron and the charge on the ligand NO in [Fe(H_(2)O)_(5)NO]SO_(4) are …………. . |
|
Answer» `+2` and `0` respectively |
|
| 24. |
Oxidation state of Fe in Fe_(3)O_(4) is |
|
Answer» Solution :`overset(**)(F)e_(3)O_(4)` `3X+(-8)=0, 3x-8=0` `3x=8 implies x=8/3`. |
|
| 26. |
Oxidation state of copper is +1 in |
|
Answer» MALACHITE |
|
| 30. |
Oxidation state of chlorine is highest the compound : |
|
Answer» `CI_2` |
|
| 32. |
Oxidation state of carbone in its hydrides………….. |
|
Answer» `+4` |
|
| 34. |
Oxidation state of +1 for phosphorus is found in: |
|
Answer» `H_3PO_3` |
|
| 35. |
Oxidation state of +1 for phophorous is found in |
|
Answer» <P>`H_(3)PO_(3)` |
|
| 36. |
Oxidation state, covalency of sulphur and total number of lone pairs of electrons in S_(8) molecule are respectively |
|
Answer» `+6,6,16` |
|
| 37. |
Oxidation state if iron in ferric is |
| Answer» SOLUTION :It is the correct answer. | |
| 38. |
Oxidation state exhibited by group 13 elements is ___________ |
|
Answer» `+1,+2` and `+3` |
|
| 39. |
Oxidation products of glucose are |
|
Answer» Sucrose |
|
| 40. |
Oxidation product of .X. (molecular formula C_3H_6O is .Y. (molecular formula C_3H_6O_2). The compound .Y. is: |
|
Answer» ACETIC ACID |
|
| 41. |
Oxidation product 1,2-cyclopentanediol with HIO_(4) gives |
|
Answer» `Hunderset(O)UNDERSET(||)C-CH_(2)-CH_(2)-CH_(2)-underset(O)underset(||)C-H` 
|
|
| 42. |
Oxidation potential of given hydrogen half cell at 25^(@)C is 0.118V then what is the pH of H^(+) ion solutioin ? P|underset((1" bar"))(H_(2(g)))|H_((xM))^(+) |
|
Answer» 1 |
|
| 43. |
Oxidation potential of unimolar calomel electrode is |
|
Answer» `0.242 V` |
|
| 44. |
Oxidation potential of unimoles of calomel is |
|
Answer» `+ 0.25` V |
|
| 45. |
Oxidation of which of the following by air in the presence of vanadium pentoxidegivesphenol ? |
|
Answer» TOLUENE |
|
| 46. |
Oxidation oftoluenewith CrO_3 to benzaldehydeis carriedout in thepressureto aceticanhydride |
Answer» SOLUTION :DURINGTHE oxidationoftoluenewith ` CrO_3`as soonasbenzaldehyde is FORMED, itreactswithaceticanhydride to frombenzylinediacetatethischecks thefurtheroxidationof benzaldehydre to BENZOICACID 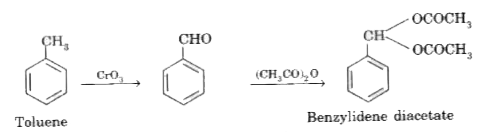
|
|
| 47. |
Oxidation of toluene with CrO_(3) in the presence of (CH_(3)CO)_(2)O gives a product 'A' which on treatment with aqueous NaOH produces |
|
Answer» `C_(6)H_(5)CHO` 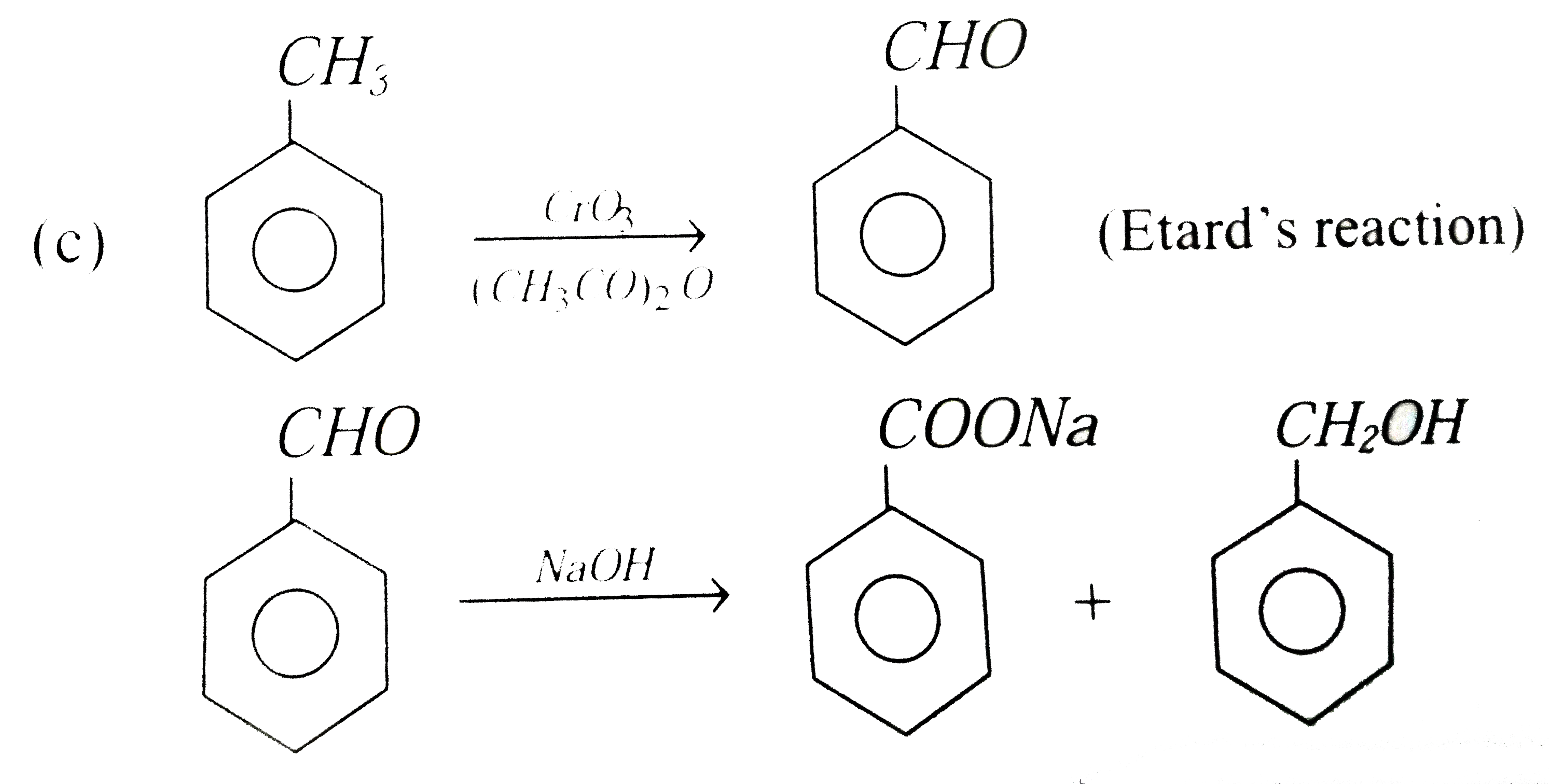
|
|
| 48. |
Oxidation of toluene with CrO_(3) in the presence of (CH_(3)CO)_(2)O gives a product 'A' which on treatment with aq. NaOH gives : |
|
Answer» 2, 4- Diacetyl toluene |
|
| 49. |
Oxidation of toluene to benzaldehyde by chromyl chloride is called |
|
Answer» Rosenmund reaction |
|
| 50. |
Oxidation of toluene by chromium oxide in acetic anhydride gives |
|
Answer» BENZALDEHYDE |
|