Saved Bookmarks
Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Out of [Sc(H_(2)O)_(6)]^(3+) and [Ti(H_(2)O)_(6)]^(3+)ions whichiscolouredand why? Give reason. |
| Answer» SOLUTION :`[Ti(H_(2)O)_(6)]^(3+)` is coloured while `[Sc(H_(2)O)_(6)]^(3+)` is colourless.This is because `Ti^(3+)`has ONE electron in the d-orbital `( 3d^(1))` which can absorb energy corresponding to yellow wavelength and undergo d-d transition (from `t_(2g)` to `e_(g))`. BU`Sc^(3+)` is `3d^(0)`,i.E., it has no electron in 3d subshell. | |
| 2. |
Out of seven crystal systems how many have body centred unit cell ? |
|
Answer» 4 |
|
| 3. |
Out of potassium nitrate and aluminium nitrate. Which one is required in minimum concentration to coagulate arsenious sulphide Sol. |
| Answer» SOLUTION :ALUMINIUM NITRATE. | |
| 4. |
Out of potassium nitrate and aluminium nitrate which one is required in minimum concentration to coagulate arsenions sulphide sol. |
| Answer» SOLUTION :ALUMINIUM NITRATE. | |
| 5. |
Out of PbS and PbCO_(3) (ores of lead),which one is concentrated by froth floatation process preferably? |
| Answer» SOLUTION :PbS is CONCENTRATED by FROTH floatation PROCESS. | |
| 6. |
OutofPbS andPhCO_3(oresoflead), whichoneisconcentrated byfroth floatation processpreferably ? |
| Answer» SOLUTION :PBS beingasulphideore, ispreferably concentratedbyfrothfloatation PROCESS. | |
| 7. |
Out of physisorption and chemisorption which one has lower enthalpy of adsorption? |
| Answer» SOLUTION :PHYSICAL ADSORPTION. | |
| 8. |
Out of o-nitrophenol annd p-nitrophenol, which is more volatile? Explain. |
| Answer» SOLUTION :o-Nitrophenol exists as discrete molecules due to INTRAMOLECULAR H-bonding (i.e., chelation) and HENCE is more VOLATILE than p-nitrophenol which exists as an associated molecule due to intermolecular H-bonding.For structures of chelated and associated molecules. | |
| 9. |
Out of o - nitrophenol and p - nitrophenol, which is more volatile? Explain. |
| Answer» Solution :o-nitrophenol is more volatile DUE to INTRAMOLECULAR hydrogen bonding. There is INTERMOLECULAR hydrogen bonding in p-nitrophenol which raises its BOILING point and MAKES it less volatile. | |
| 10. |
Out of o-nitrophenol and p-nitrophenol, which is more volatile ? |
| Answer» Solution :Ortho-nitrophenol is more VOLATILE because of intramolecular H-bonding (Chelation) whereas, the intermolecular H-bonding is present in p-nitrophenol. THUS, the p-nitrophenol is associated with the other MOLECULES through H-bonding and so it shows higher BOILING point. | |
| 11. |
Out of o - nitrophenol and o - cresol, which is more acidic? |
| Answer» SOLUTION :o - NITROPHENOL is more ACIDIC because of electron withdrawing nature of nitro group. Methyl group in o - cresol is electron REPELLING and that makes o - cresol less acidic. | |
| 12. |
Out of o-nitrophenol and o-cresol which is more acidic? |
Answer» SOLUTION :Due to -I and -R-effect of the `NO_(2)` GROUP, electron density in the O-H bond decreases while due to +I or hyperconjugation effect, of the `CH_(3)` group, the electron density in the O-H bond increases. 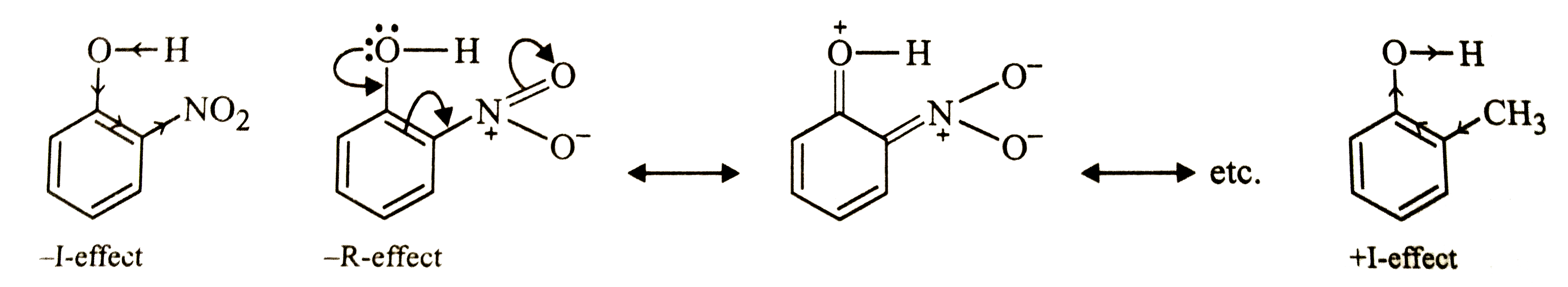 As a RESULT, O-H bond is o-nitrophenol is much weaker than the O-H bond is o-cresol and HENCE o-nitrophenol is much more acidic than o-cresol. |
|
| 13. |
Out of o-nitrophenol and o-cresol which is more acidic ? |
Answer» Solution :`to` The presence of -I or -M GROUP stabilizes the phenoxide ION and thus increases the acidic strength of the phenol whereas the +I or +M group DESTABILIZES the phenoxide ion and as a result the acidic strength decreases. The `-NO_(2)` is strong -M and -I group, whereas the `-CH_(3)` (METHYL group) is +I group, thus, `-NO_(2)` increases the acidic strength of the phenol. So, o-nitrophenol is more acidic. 
|
|
| 14. |
Out of o- and p- dibromobenzenes which one has higher melting point and why? |
| Answer» SOLUTION :p-dibromobenzene has HIGHER melting point than its o isomer. It is DUE to symmetry of p-isomer which fits in CRYSTAL lattice better than the o-isomer. | |
| 15. |
Out of o- and p-dibromobenzene which one has higher melting point and why? |
| Answer» Solution :p-Dibromobenzene being symmetrical fits in the CRYSTAL LATTICE better than the o-isomer and HENCE has higher melting point than the o-isomer. | |
| 16. |
Out of o- and p-dibromobenzene, which one has higher melting point and why? |
Answer» SOLUTION : The structure of p-dibromobenzene is more symmetrical as compared to o-dibromobenzene. As a RESULT, in the crystal LATTICE molecules of p-isomer are more closely packed as compared to the ORTHO isomer. QUITE obviously, the melting point of the p-isomer is higher. |
|
| 17. |
Out of o - and p - dibromobenzene which one has higher melting point and why ? |
|
Answer» <P> SOLUTION :p - DIBROMOBENZENE has higher melting point than its o - isomer. It is due to its SYMMETRICAL STRUCTURE that can be fitted in a crystal structure. |
|
| 18. |
Out of NH_(3) and 'en' which ligand forms a more stable complex with metal and why? |
| Answer» Solution :en' forms a more stable COMPLEX to `NH_(3)` since it is involved in CHELATE FORMATION. | |
| 19. |
Out of Na^(+) andAg^(+) ions, which one is a stronger Lewis acid and why ? |
| Answer» Solution :`Ag^(+)` ion is a stronger Lewis acid than `NA^(+)` ion. Thisis because stronger Lewis acidis ONE which can accepta pair of electrons more easily . `Ag^(+)` ions haspseudo noble GAS configuration ( where outer shell has18 electrons ) wheras `Na^(+)` ion has a noble gas configuartion with 8 electron in the outermost shell. The18 electrons of the outermost shell of `Ag^(+)` ionare DISTRIBUTED as 10 electrons in the inner d-shell and 8 electrons in the outer s and p subshells. The inner d-electrons havepoor screening or shielding effect. As a RESULT, `Ag^(+)` ion hashigher effective nuclearcharge and can take up a pair of electrons more easily. | |
| 20. |
Out of n-type and p-type which is a better conductor of electricity ? |
| Answer» SOLUTION :n-type of SEMICONDUCTOR. | |
| 21. |
Out of molality and molarity which is better for expressing the concerntration of a solution ? |
| Answer» SOLUTION :MOLALITY is BETTER. | |
| 22. |
Out of n-pentane, ethoxyethane and butan-1-ol, which has the highest boiling point ? |
| Answer» SOLUTION :Butan-1-ol | |
| 23. |
Out of N, O,Pand S, the highest ionisation enthalpy is of: |
|
Answer» N |
|
| 24. |
Out of MgCl_(2)and AlCl_(3) which one is more effective in causing coagulation of negatively charged sol and why ? |
| Answer» SOLUTION :`AlCl_(3)` due to GREATER POSITIVE charge on `AL^(3+)` than `Mg^(2+)` | |
| 25. |
Out of MgCl_2 and AlCl_3, which one is more effective in causing coagulation of negatively charged sol and why? |
| Answer» Solution :`AICI_3` is more effective in causing coagulation of negatively charged sol. This is because `Al^(3+)` ion carries greater POSITIVE charge than `MG^(2+)` ion. According to Hardy-Schulze RULE, greater the charge on an ion, greater is the capability to coagulate oppositely charged sol. | |
| 26. |
Out of M H_(2)SO_(4) and 1H J_(2)SO_(4) which is mor econcentreated and why ? |
| Answer» SOLUTION :`1M H_(2)SO_(4)` is more concerntrated. | |
| 27. |
Out of Lu(OH)_(3) and La(OH)_(3) which is more basic and why? |
|
Answer» SOLUTION :(i) As we move from `Ce^(3+)` to `Lu^(3+)`, the BASIC character of `Lu^(3+)` IONS decreases. (ii) Due to the DECREASE in the SIZE of `Lu^(3+)` ions, the ionic character of Lu-OH bond decreases, covalent character increases which results in the decrease in the basicity, (iii) Hence, `La(OH)_(3)` is more basic than `Lu(OH)_(3)` |
|
| 28. |
Out of K,Na, Be and Kr, the highest ionisation enthalpy is of: |
|
Answer» Kr |
|
| 29. |
Out of HCl and NaCl, which do you expect will have greater value for wedge_(m)^(@) and why? |
| Answer» Solution :HCL will have gerater VALUE for `wedge_(m)^(@)` because `H^(+)` ions are smaller than `Na^(+)` ions. Consequently, `H^(+)` ions will have GREATER ionic mobility and hence ionic CONDUCTANCE than `Na^(+)` ions. | |
| 30. |
Out of H_(2)Oand H_(2)S, which one has higher bond angle and why ? |
| Answer» Solution :Oxygen is more electronegative than sulphuur, therefore, bond PAIR electrons of the O-H bond will be closer to oxygen than the bond pair electrons of S-H bond which will be LITTLE away from the SULPHUR atom. As a result, bond pair-bond pair REPULSIONS would be stronger in O-H bonds than in the S-H bonds. Consequently, bond angle in `H_(2)O (104.5^(@))` is HIGHER than in `H_(2)S(92.1^(@))` | |
| 31. |
Out of H_2O and H_2S, which one has higher bond angle and why ? |
| Answer» SOLUTION :MOVING down the group the atomic size increases and electronegativity of central atom DECREASES. As a result, the ELECTRON density on a central atom decreases. Also, the `sp_3` CHARACTER of a bond becomes less distinct. Thus `H_2O` has a greater bond angle than `H_2S`. | |
| 32. |
Out of H_2O and H_2S which has higher bond angle and why? |
| Answer» Solution :Bond angle in `H_2O` is GREATER because OXYGEN is more electronegative than SULPHUR. Therefore bond pair electrons of O - H bond will be closer to oxygen. There will be greater bond-pair bond-pair repulsion between the two O-H BONDS. | |
| 33. |
Out of following which compound is used for preservation of wood |
|
Answer» `NACL` |
|
| 34. |
Out of following number of laboratory test which will be positive for ? (a) Haloform test (b) 2,4-DNP test (c ) Cerric ammonium nitrate test (d) Esterification (e ) Tollen's reagent test (f) Fehling test |
|
Answer» |
|
| 35. |
Out of following number of aplymers which areformed by condensation polymerization (x) : Teflon, Bakelite, Nylon-6, Poylstyrene, PVC |
|
Answer» |
|
| 36. |
Out of following forms of sulphur S_(8),S_(6) and S_(2) which one is paramagnetic in nature and why ? |
|
Answer» |
|
| 37. |
Out of Fe,Cu,Sn and Hg, the most reactive metal is ____and least reactive metal is_____. |
| Answer» SOLUTION :`FE,HG(E_(Fe//Fe^(2+))^(@)=+0.44V,E_(Sn//Sn^(2+))^(@)=+0.14V,E_(Cu//Cu^(2+))^(@)=-0.34V,E_(Hg//Hg^(2+))^(@)=-0.86)` | |
| 38. |
Out of ethyl bromide and ethyl chloride which has higher boiling point and why? |
| Answer» Solution :Due to BIGGER size of Br over Cl, the van der Waals forces of attraction are stronger in ethyl BROMIDE than in ethyl chloride. Therefore, ethyl bromide has higher BOILING point than ethyl chloride. | |
| 39. |
Out of ethyl amine and ethyl alcohol which has higher boiling point and why ? |
| Answer» Solution :Because there is stronger intermolecular H-bonding in ethanol than ETHYL AMINE due to more electronegativity of OXYGEN than nitrogen. | |
| 40. |
Out of Cu_(2)S, HgS, Ag_(2)S and ZnS roasting will convert the minerals into metal in case of |
| Answer» Answer :D | |
| 41. |
Out of Cu,Ag,Fe and Zn the metal which can displace all other from their saltsolutions is : |
|
Answer» Ag |
|
| 42. |
Out of Cu_(2)Cl_(2) and CuCl_(2) which is more stable and why? |
| Answer» SOLUTION :`CuCl_(2)` is more STABLE because of higher stability of `Cu^(2+)` ION due to high negative value of HYDRATION enthalpy of `Cu^(2+)` than `Cu^(+)` | |
| 43. |
Out of Cu_(2)Cl_(2) and CuCl_(2) which is more stable and why ? |
| Answer» SOLUTION :`CuCl_(2)` is more STABLE than `Cu_(2)Cl_(2)` . This is because `Cu^(2+) (aq)` has greater negative enthalpy of hydration than `Cu^(+) (aq)`. | |
| 44. |
Out of Cu_(2)Cl_(2) and CuCl_(2), which is more stable and why ? |
| Answer» Solution :`CuCl_(2)` is more stable than `Cu_(2)Cl_(2)`. GREATER stability of `CU^(2+)` (AQ) is due to more NEGATIVE `Delta_("hyd")H^(@)` of `Cu^(2+)` (aq) than that of `Cu^(+)`(ag). | |
| 45. |
Out of Cu^(2+),Ni^(2+),Co^(2+) and Mn^(2+) of those thatdissolve in dil HCI only one give precipitate when H_(2)S is passed. Identifythe corresponding order which do notgive precipitation : |
|
Answer» `Ni^(2+)` |
|
| 46. |
Out of Cr^(2+),Ni^(2+),Co^(2+) and Mn^(2+) those dissolve in dil. HCl, only one gives a precipitate when H_(2)S is passed. Identify the corresponding one |
|
Answer» `Ni^(2+)` |
|
| 47. |
Out of Cs^(+),Li^(+),Mg^(2+) and Na^(+),the largest size of : |
| Answer» Answer :A | |
| 48. |
Out of Cr^(2+) and Cr^(3+), which one is stable in aqueous solution? |
| Answer» Solution :`Cr^(3+)` is more stable than `Cr^(2+)` in aqueous solutions. It is because of GREATER hydration ENTHALPY of `Cr^(3+)` ions due to its SMALL size and higher charge. | |
| 49. |
Out of Cr^(2+) and Cr^(3+), which one is stable in aqueous solution ? |
| Answer» Solution :`CR^(3+)` is more STABLE in AQUEOUS solution. This is due to greater hydration ENTHALPY due to smaller SIZE and higher charge. | |