Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Out of the following two coordination entities, which is chiral (optically active) ? (a) cis-[CrCl_(2)(o x)_(2)]^(3-)(b) trans-[CrCl_(2)(o x)_(2)]^(3-) |
Answer» SOLUTION :
|
|
| 2. |
Out of the following which are soluble in-water:(i) AgCN (ii)Ag_2S (iii) AgF (iv)AgNO_3 (v)AgClO_4. |
|
Answer» (i) (ii) (iii) |
|
| 3. |
Out of the following transition elements, the maximum number of oxidation states are shown by |
|
Answer» `SC (Z=21)` |
|
| 4. |
Out of the following three solutions, which has the highest freezing point and why? (i) 0.1 M Urea (ii) 0.1 M Barium phosphate (iii) 0.1 M Sodium sulphate |
|
Answer» |
|
| 5. |
Out of the following three solutions, which has the highest boiling point and why? (i) 1 M Glucose (ii)1 M Potassium chloride (iii) 1 M Aluminium nitrate |
| Answer» SOLUTION :`"1 M AL"(NO_(3))_(3)` | |
| 6. |
Out of the following, the strongest base in aqueous solution is |
|
Answer» METHYLAMINE |
|
| 7. |
Out of the following the one which is not a primary standard is |
| Answer» Answer :B | |
| 8. |
Out of the following the number of metals that can not be obtained by electrolysis of the aqueous solution of their sals are Ag,Mg,Cu,Al,Au,Ca,Na |
|
Answer» |
|
| 9. |
out of the following the correct statement(s) is/are (a) Number of subshells present in M-shell is equal to 3, (b) maximum number of electrons present in l shell is equal to 8, (c) number of electron present in subshell is 3(2l + 1), (d) Cu^+ is paramagnetic |
|
Answer» (a),(B) &(C) |
|
| 10. |
Out of the following the metal which forms polynuclear carbonyl is: |
|
Answer» Na |
|
| 11. |
Out of the followingwhich metal forms polynuclear carbonyl : |
|
Answer» Na |
|
| 12. |
Out of the following, the alkene that exhibits optical isomerism is: |
|
Answer» 2-methyl-2-pentene |
|
| 13. |
Out of the following the best category of proteins is |
|
Answer» polyamides |
|
| 14. |
Out of the following solutions which has the maximum freezing point and why ? (i) 1 M urea (ii) 1 M potassium chloride (iii) 1 M aluminium chloride |
| Answer» SOLUTION :1 M UREA solution. | |
| 15. |
Out of the following the alken that exhibits optical isomerism is: |
|
Answer» 2-Methylpent-2-ene 
|
|
| 16. |
Out of the following the alkene that exhibits optical isomerism is : |
|
Answer» 3-methyl-1-pentene is OPTICALLY active because of chiral C. |
|
| 17. |
Out of the following pairs (of complexes), in which option, first complex is more stable than second complex : |
|
Answer» `[CO(NO_(2))_(6)]^(4-)" and "[Co(NO_(2))_(6)]^(3-)` |
|
| 18. |
Out of the following pairs of electrons, identify the pairs of electrons present in degenerate orbitals. |
|
Answer» (i) `n = 3, I= 2, m_(L) = -2, m_(s)=-1/2`  `3d_(xy))` and `3d_(yz),3d_(x^(2)-y^(2))` represent PAIR of degenerate orbitals. |
|
| 19. |
Out of the following oxides of nitrogen how many of them are neutral oxides? N_(2)O, NO, N_(2)O_(3), NO_(2),N_(2)O_(4), N_(2)O_(5) |
|
Answer» 2 |
|
| 20. |
Out of the following outer electronic configurations of atoms, the highest oxidation state is achieved by which one? |
|
Answer» `(n-1)d^(8)ns^(2)` |
|
| 21. |
Out of the following metals that cannot be obtained by electrolysis of the aqueous solution of their salts is |
|
Answer» Ag |
|
| 22. |
Out of the following identify (i) the *d' block element (ii) the of block element. Ca,Mn,U,Al |
| Answer» SOLUTION :(i) MN (II) U | |
| 23. |
Out of the following, how many oxides are acidic. MnO, Mn_(2)O_(3), MnO_(2), MnO_(3), Mn_(2)O_(7). |
|
Answer» `UNDERSET("netural")OVERSET(+2)(MnO, Mn_(2)O_(3)), underset("Acidic")(MnO_(2)^(+4), MnO_(3)^(+6), Mn_(2)O_(7)^(+7))` |
|
| 25. |
Out of the following gases which will be adsorbed more readily on the surface of charcoal : NH_3 gt CO_2 gt O_2 |
|
Answer» SOLUTION :The order of CRITICAL temperature for the GIVEN GASES is `NH_3 GT CO_2 gt O_2` Hence `NH_3` has strongest intermolecular forces of attraction and hence will be adsorbed more readily. |
|
| 26. |
Out of the following fractions of petroleum, the one having the lowest boiling point is |
|
Answer» KEROSENE |
|
| 27. |
Out of the following elements,which one is the most reactive chemically? |
|
Answer» Ba |
|
| 28. |
Out of the following compounds, which will be dehydrated most readily? |
Answer» Solution :The dehydration can take place in the acidic medium in the presence of conc. `H_(2)SO_(4)` . A carbocation us EXPECTED to be formed in each case as the intermediate in the rate dertermining step. 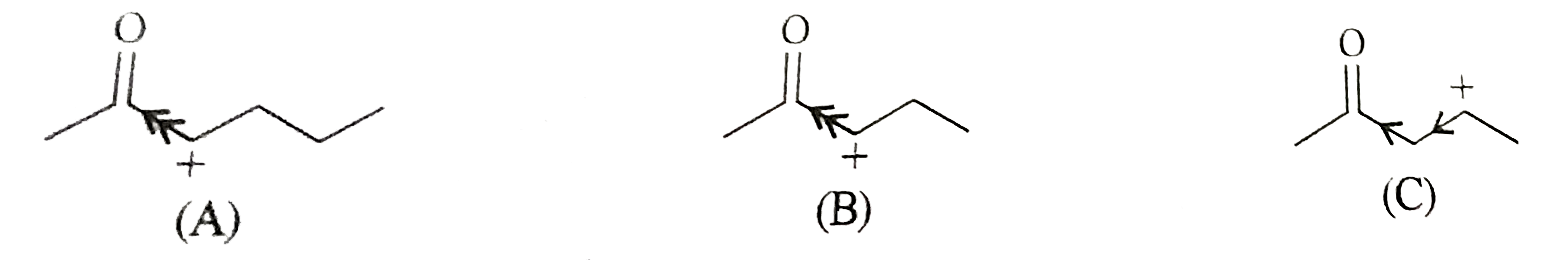 In the structures (A) and (B) the positively charged carbon atom is adjacent to the electron withrawing carbonyl group which tends to destabilise the carboncations by incresing the magnitude of the positive charge on these. HOWEVER, in carbocation (C), the carbonyl group is not directly ATTACHED to the positively charged carbon atom. THEREFORE, its positive charge weakning effect is comparactively is more stable. This mean that the compound (C) will be dehydrated most easily. |
|
| 29. |
Out of the following four compounds, which is expected to show optical activity. {:(""R''),("|"),(R-overset(..)(N)-HR-overset(..)(N)-R'", "R-overset(..)(N)-R' R-N-R),("||||"),(""H""H""R''""R''),("(I)(II)(III)(IV)"):} |
|
Answer» Only III |
|
| 30. |
Out of the following elements which one do you expect to be most reactive chemically |
|
Answer» Mg |
|
| 31. |
Out of the following compounds the most acidic is |
|
Answer» `As_(2)O_(3)` |
|
| 32. |
Out of the following, choose the one which is not a part of internal energy? |
|
Answer» KINETIC energy |
|
| 33. |
Out of the following compounds, which is the most basic ? |
|
Answer» `CH_(3)NH_(2)` |
|
| 34. |
Out of the following, choose a correct expression for finding out the cystal field stabilization energy in tetrahedral coordination entities. |
|
Answer» (## AAK_MCP_27_NEET_CHE_E27_030_A001 .PNG" WIDTH="30%"> |
|
| 35. |
Out of the following, choose a correct statement. |
|
Answer» [Cu(NH_3)_6]^(2+)` is a colourless ION |
|
| 36. |
Out of the following bond order of oxygen species identify the correct answer |
|
Answer» `O_2^(-)gtO_2gtO_2^+` |
|
| 37. |
Out of the following 1.0 m aqueous solutions, which one will exhibit the largest point depression ? |
|
Answer» `C_(6)H_(12)O_(6)` |
|
| 38. |
Out of the folloowing, the alkene that exhibits optical isomerism is |
|
Answer» 3-methyl-1-pentene It has a chiral carbon and hence, it is optically active. All other ALKENES do not have a chiral carbon and hence are optically INACTIVE. |
|
| 39. |
Out of the first 80 elements,the number of elements having 3d orbitals are: |
|
Answer» 10 |
|
| 40. |
Out of the compounds K_(2)SO_(4),MgCl_(2),FeSO_(4),NiCl_(2) and ZNO which of the following pair will show paramagnetism- |
|
Answer» `K_(2)SO_(4),MgCl_(2)`  
|
|
| 41. |
Out of the elements with atomic numbers given below which one would be a halogen: |
|
Answer» 25 |
|
| 42. |
Out of the atomicnumbers given below suggest which one of the following will be a d-group element and name the element. |
|
Answer» 12 |
|
| 43. |
Out of tetracarbonyl nickel (0) and tetraamminecopper (II) sulphate, which is magnetic and why ? |
| Answer» SOLUTION :Tetraamminecopper (II) SULPHATE is paramagnetic whereas tetracarbonyl NICKEL (0) is diamagnetic. | |
| 44. |
Out of temperature and pressure, which does not affect the solubility of a solid in the solvent ? |
| Answer» SOLUTION :PRESSURE | |
| 46. |
Out of sulphur and proteins, which one froms multimolecular colloids ? |
| Answer» SOLUTION :SULPHUR | |
| 47. |
Out of sulphur sol and proteins, which one forms multimolecular colloids ? |
| Answer» SOLUTION :SULPHUR FORMS MULTIMOLECULAR COLLOIDS. | |
| 48. |
Out of sulphur sol and proteins , which one forms macromolecular colloids? |
| Answer» SOLUTION : PROTEINS FORM macromoleucular COLLOIDS . | |
| 49. |
Out of specific, equivalent and molar conductivities, the quantity which decreases with dilution is____. |
| Answer» SOLUTION :SPECIFIC CONDUCTIVITY. | |
| 50. |
Out of sodium dichromate and potassium dichromate, which is more soluble ? |
| Answer» SOLUTION :SODIUM DICHROMATE | |