Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
One of the following is the chief source of water and soil pollution |
| Answer» Answer :D | |
| 2. |
One of the following is an incorrect statement , point it out, |
|
Answer» Temporary HARDNESS is due to bicarbonates of Ca and MG. |
|
| 3. |
One of the following is false for Hg |
|
Answer» it can EVOLVE hydrogen from `H_(2)S` `Hg+H_(2)Sto"No REACTION"`. |
|
| 4. |
One of the following has O-O Bond |
|
Answer» `H_2S_2O_6` |
|
| 5. |
One of the following is an essential amino acid. |
|
Answer» CYSTEINE |
|
| 6. |
One of the following conversion results in the change of hybridization and gemetry : |
|
Answer» `NH_(3)toNH_(4)^(+)` `sp^(3) " HYBRIDIZATION" sp^(3) " hybridization "` `"Tetrahedral geometryTetrahedral geometry "` `CH_(4)to C_(2)H_(6)` `sp^(3)"hybridized C" sp^(3) "hybridizedC"` `"Tetrahedral geometryTetrahedral geometry "` `H_(2) Oto H_(3)O ` `sp^(3) " hybridization" sp^(3) " hybridization "` `"Tetrahedral geometryTetrahedral geometry "` `BF_(3) to BF_(4)^(-)` `sp^(3) " hybridization" sp^(3) " hybridization "` ` "TRIGONAL plaanar geometryTetrahedral geometry "` |
|
| 7. |
One of the following cannot be prepared by Kolbe's electrolytic process? |
|
Answer» `CH_4` |
|
| 8. |
One of the following amides will not undergo Hofmann bromamide reaction: |
|
Answer» `CH_(3)CONHCH_(3)` |
|
| 9. |
One of the following amide will not undergo Hoffmann bromamide reaction : |
|
Answer» `C_(6)H_(5)CONH_(2)` |
|
| 11. |
One of the essential alpha amino acids is: |
|
Answer» Lysine |
|
| 12. |
One of the distinguishing reactions of aliphatic and aromatic primary amines is |
|
Answer» Carbylamine reaction |
|
| 13. |
One of the constituents of German silver is |
|
Answer» Cu |
|
| 14. |
One of the best photovoltaic material fur the conversion of sunlight into electricity is |
|
Answer» iron |
|
| 15. |
one of the components of stainless steel is : |
|
Answer» Cr |
|
| 16. |
One of the characteristic of transition metals to from the complex ion is |
|
Answer» Having unpaired ELECTRON in d-subshell |
|
| 17. |
One of the approach to the study of real gases involves the analysis of a parameter Z called the compressibility factor Z=(PV_(m))/(RT) where P is pressure, V_(m) is molar volume, T isabsolute temperature and R is the universal gas constant . Such a relation can also be expressed as Z =((V_("m real"))/(V_("m ideal")))(where V_("m ideal ") and V_("m real") are the molar volume for ideal and real gas respectively) . Gas corresponding Z gt 1 have repluive tendenciesamong constituent particles due to their size factor, among constitutent particles . As the pressure is lowered or temperature is increased the value of Z approaches 1. ( reaching the ideal behaviour) For a real gas 'G'Z gt 1 at STP then for 'G' which of the following is true |
|
Answer» 1 MOLE of the gas occupies 22.4 L at NTP `(PV_(m))/(RT)= (1xx22.4)/(R xxT ) ` At same pressure `=1` atm `(1xxV_(m))/(RT) gt (1 xx 22.4)/(R xx T ) implies V_(m) gt 22.4 L ` at STP for real gas. For `V_(m) = 22.4 L` of real gas , we have to increase the pressure. |
|
| 18. |
One of the acid listed below is formed from P_2O_3. The rest are formed from P_2O_5. The acid formed from hydrolysis is |
|
Answer» `HPO_(3)` |
|
| 19. |
One ,odel is obtained from scu water 11.9 C^(14) atoms disssociate from 1 gm carbon 15.3 C^(14) atoms dissociate from live model then model was how many years old ?t_((1)/(2))=5730 years |
| Answer» SOLUTION :2079 YEARS | |
| 20. |
one of an idealgas is expanded isothermally at 298 K unit its volume is tripled. Findthe values ofDelatS_("gas") and DeltaS_("total") under the followingconditions.(i) Expansion is carried outreversiby .(ii)Expansion is carried out irreversiblywhere836.8J of heatis less absorbed than in (i) (iii) Expansionis free. |
|
Answer» `DeltaS_("TOTAL")= 0` (ii)`DeltaS_(surr)= (-q_(sys))/(T)` `q_(IRR) = q_(rev)- 836.8` `DeltaS_(surr)= -[(q_(rev)-836.8)/(298)]` `DeltaS_("total")= 2.808 J//K` |
|
| 21. |
One molecule of which of the following ligands form only two rings towards a particular central atom? |
|
Answer» en 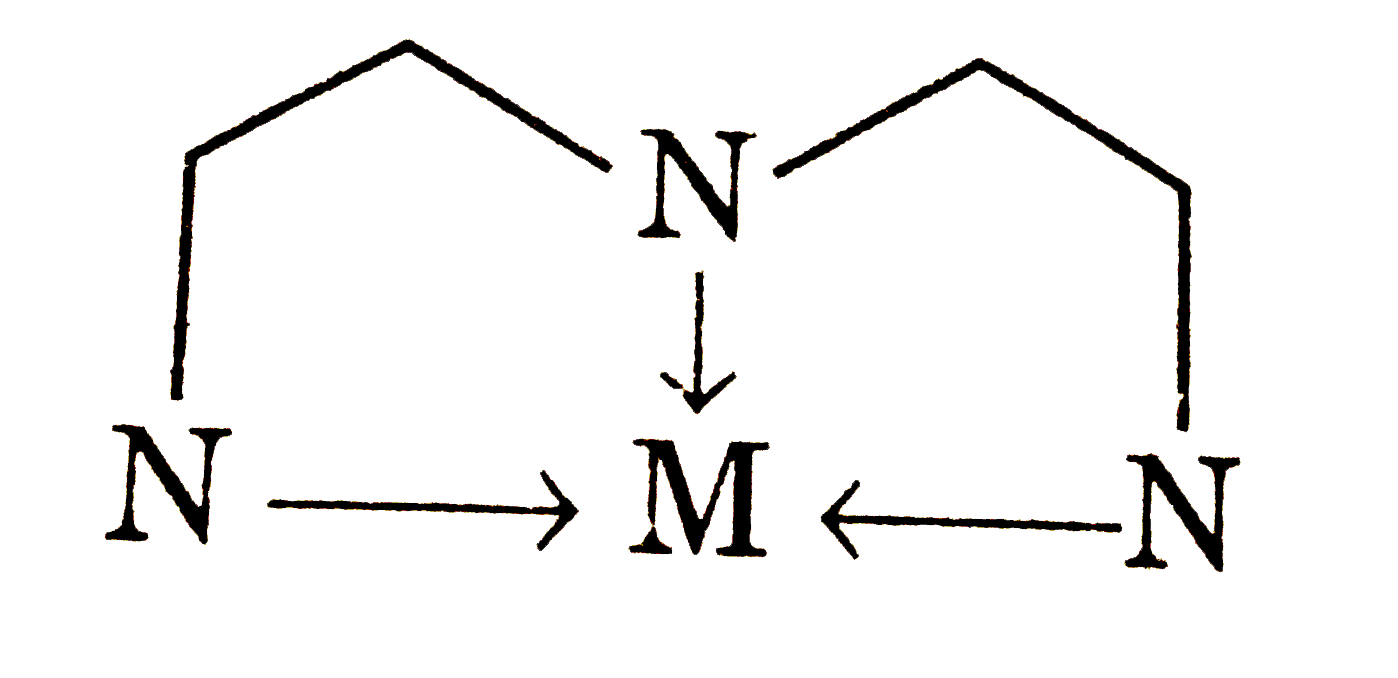
|
|
| 22. |
One moles of an ideal monoatomic gas undergo the state change as shown in following graph (ln 2 = 0.7) Efficiency of the cycle will be : |
| Answer» ANSWER :D | |
| 23. |
One molecule of haemoglobin will combine with four molecules of oxygen. If 1.0 gof haemoglobin combines with 1.53 mL of O, at body temperature (37^@C)and a pressure of 743 torr, what is the molar mass of haemoglobin? |
| Answer» SOLUTION :`6.8 XX 10^4 G "MOL"^(-1)` | |
| 24. |
One molecule of an olefinic compound, on ozonolysis, produces one molecule of acetone,one molecule of glyoxal and one molecule of formadehyde. Identify the compound and write its IUPAC name. |
Answer» Solution :The structure of the olefinic hydrocarbon MAY be obtained by arranging the carbonyl compounds obtained on ozonolysis properly (each of the two oxygen atoms of acetone and formaldehyde is PLACED in front of the 2 oxygen atoms of GLYOXAL) followed by eliminating the four oxygen atoms and joining the carbonyl carbons by double BONDS. Thus, the IUPAC name of the starting olefinic compound is 4-methylpenta -1,3-diene. 
|
|
| 25. |
One moleculeof H_(2)SO_(4) forms how many no of series of salt's |
| Answer» SOLUTION :There are (1) -ous seriesof SALTS , (2) -ic SERIES of salt. | |
| 26. |
One mole of CO_2 contains |
|
Answer» `6.02 xx 10^23` ATOMS of C |
|
| 27. |
One mole of which of the following has the highest entropy |
|
Answer» Mercury |
|
| 28. |
One mole on non-volatile solute is dissolved in two mole of water. The vapour pressure of the solution relative to that of water is: |
|
Answer» 2/3 |
|
| 29. |
One mole of which o the following has the highest entropy? |
|
Answer» Liquid nitrogen |
|
| 30. |
One mole of which of the following has the highest entropy ? |
|
Answer» mercury |
|
| 31. |
One mole of water at 100^@C is converted into steam at 100^@C at a constant pressure of 1 atm. The change in entropy is ____________. (heat of vaporization of water at 100^@C=540cal/g) |
|
Answer» 8.74 Here, heat of vaporisation = 540 cal/gm `=540xx18 cal mol^(-1)` Temperature of WATER = 100 + 273 = 373 K `THEREFORE` entropy change = `(540xx18)/(373) = 26.06 cal mol^(-1) K^(-1)`. |
|
| 32. |
One mole of unsaturated hydrocarbon on ozonolysis gives one mole each of CH_3. CHO, HCHO and OHC. CHO. The hydrocarbon is |
|
Answer» `CH_3.CH_2C-=C.CH_3` |
|
| 33. |
One mole of the complex compound Co(NH_(3))_(5)Cl_(3), gives 3 moles of ions on dissolution in water. One mole of the same complex reacts with two moles of AgNO_(3) solution to yield two moles of AgCl (s). The structure of the complex is |
|
Answer» `[CO(NH_(3))_(3)Cl_(3)]*2NH_(3)` |
|
| 34. |
One mole of the complex compound Co(NH_3)_5Cl_3 , gives 3 moles of ions on dissolution in water. One mole of the same complex reacts with two moles of AgNO_3 solution to yield two moles of AgCl_((s)) . The structure of the complex is _________. |
|
Answer» `[CO(NH_3)_3Cl_3]2NH_3` |
|
| 35. |
One mole of the complex compound Co(NH_(3))_(5)Cl_(3)gives 3 moles of ion on dissolution in water. One mole of the same complex reacts with two moles ofAgNO_(3) solution to yield two moles ofAgCl(s). The structure of the complex is– |
|
Answer» `[CO(NH_(3))_(4)CL]Cl_(2).NH_(3)` |
|
| 36. |
One mole of the complex compound CoCl_3 xNH_3 gives 3 moles of ions on dissolution in water. One mole of the same complex reacts with two moles of AgNO_3 solution to yield two moles of AgCl. The value of x is |
|
Answer» 5 |
|
| 37. |
One mole of stachyose on hydrolysis yields |
|
Answer» 1 MOLE of glucose + 1 mole of fructose + 2 MOLES of galactose |
|
| 38. |
One mole of solid Zn is placed in excess of dilute H_(2)SO_(4)" at "27^(@)C in a cylinder fitted with a piston. Find the value of DeltaE, q and w for the process if the area of piston is 500cm^(2) and it moves out by 50 cm against a pressure of 1 atm during the reaction. The heat given to surrounding is 36.5 KJ. Zn(s)+2H^(-)(aq)hArrZn^(2+)(aq)+H_(2)(g) |
| Answer» SOLUTION :`DeltaE=-39.03KJ//"MOLE",q=-36.5kJ:w=-2.53kJ` | |
| 39. |
One mole of SO_(3) was placed in a litre reacton vesel at a certain temperaturee. The following equilibrium was established 2SO_(3)hArrSO_(2)+O_(2) At equlibrium 0.6 moles of SO_(2) were formed. The equlibrium constant for the reaction will be |
|
Answer» `0.36` `K_(C)=([SO_(2)]^(2)[O_(2)])/([SO_(3)])=(0.6xx0.6xx0.3)/(0.4xx0.4)=0.675.` |
|
| 40. |
One mole of SO_(3) was placed in a vessel of 1 litre capacity at a certain temperature when the following equilibrium was established. 2SO_(3)iff2SO_(2)+O_(2) At equilibrium, 0.6 moles of SO_(2) were formed. The equilibrium constant of the reaction will be |
|
Answer» `0.36` `K=((0.6)^(2)(0.3))/((0.4)^(2))=0.675` |
|
| 41. |
One mole of radioactive nuclide , X with half life of ten days decays as ""_(Z)^(A) X to ""_(Z- 4) ^(A - 8) Y + 2 ""_(2) ^(4) He . If the no. of moles of Helium formed in 20 days is 0.25 x then x = ….. |
|
Answer» t = 20 days , `n_x` consumed = `(1 - (1)/(4)) = (3)/(4)` mole of X, 1 mole of .X. from `to` 2 mole of He GAS `(3)/(4)` mole of .X. from `to 2 xx (3)/(4)` mole of He gas , `V_(He) = 2 xx (3)/(4) xx 22.4 = 33.6I`, `n_(He "gas") to 2 xx (3)/(4) = (3)/(2)` MOLES , `0.25 xx = (3)/(2) , x = (3)/(2 xx 0.25)= (3)/(0.50) = 6` |
|
| 42. |
One mole of propanone and one mole of formaldehyde are the products of ozonolysis of one mole of an alkene. The alkene may be |
|
Answer» 2-methylpropene |
|
| 43. |
One mole of Phenols is warmed with sodium metal. If we assume 100% yield, volume of H gas liberated at S.T.P is |
|
Answer» 11.2L |
|
| 44. |
One mole of potassium dichromate completely oxidises the following number of moles of ferrous sulphate in acidic medium |
|
Answer» 1 eq. of `K_(2)Cr_(2)O_(7)=` eq. of `FeSO_(4)` `1xx6=x xx1` |
|
| 45. |
One mole of potassium chlorate is thermally decomposed and excess of aluminium is burnt in the gaseous product. How many moles or aluminium oxide are formed? |
|
Answer» `1` `4Al+3O_(2)to2Al_(2)O_(3)` 2 moles of `KClO_(3)` gives 2 moles of `Al_(2)O_(3)` `:.` 1 MOL of `KClO_(3)` gives 1 mol of `Al_(2)O_(3)` |
|
| 46. |
One mole of phenol is warmed with sodium metal . If we assume 100% yield , volume of H_2 gas liberated at S.T.P is |
|
Answer» `11.2L ` |
|
| 47. |
One mole of oxygen gas at STP is equal to ……….. |
|
Answer» `6.022xx10^(23)` molecules of oxygen |
|
| 48. |
One mole of nitrogen gas at 0.8 atm takes 38 seconds to diffuse through a pinhole, whereas one mole of an unknown compound of xenon with fluorine at 1.6 atm takes 57 seconds to diffuse through the same hole. Calculate molecular formula of the compound. |
|
Answer» <P> Solution :`(r_(1))/(r_(2))=sqrt((M_(2))/(M_(1)))xx(P_(1))/(P_(2))`or `(n_(1))/(t_(1))xx(t_(2))/(n_(2))=sqrt((M_(2))/(M_(t)))xx(P_(1))/(P_(2))` or `(1)/(38)xx(57)/(1) =sqrt((M)/(28))xx(0.8)/(1.6)` `therefore M=252` |
|
| 49. |
One mole of nitrogen is mixed with 3 mole of hydrogen in a closed 3 litre vessel 20 % of nitrogen is converted into NH_3 .Then what is the K_C for (1)/2(N_2) + (3)/2(H_2) ⇌ NH_3 |
|
Answer» 0.36 LITRE `MOL^(-1)` |
|
| 50. |
One mole of oxygen at 273 K and one mole of sulphur dioxide at 546 K are taken in two separate containers, then |
|
Answer» kinetic energy of both are equal Molecular mass of `O_(2) = 2 XX 16 = 32` For SULPHUR DIOXIDE, n = 1 mole. T = 546 K = 2(273)K Molecular mass of `SO_(2) = 32 + 2(16) = 64` Kinetic energy = (3/2)RT `(K.E_(I))/(K.E_(II)) = ((3)/(2) xx Rxx T)/((3)/(2) xx R xx 2T) = 1/2` `therefore` Kinetic energy of `SO_(2) gt` Kinetic energy of `O_(2)`. |
|