Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
One mole of nitrogen gas at 0.8 atm takes 38 second of diffuse through a pin hole whereas one mole of an unknown compound of Xenon with fluorine at 1.6 atm takes 56.26 second to diffuse through at same hole, then the molecular formula of the compound is (Atomic mass of Xenon : 131.3 u) |
|
Answer» `XeF_2` |
|
| 2. |
One mole of NaCl (s) on melting absorbed 30.5 KJ of heat and its entropy is increased by 28.8JK^(-1). What is the melting point of sodium chloride ? |
| Answer» SOLUTION :T = 1059K | |
| 3. |
One mole of NH_(3) is made to react with one mole of CH_(3) Cl, which of the following is/ are formed |
|
Answer» `CH_(3)NH_(2)` |
|
| 4. |
One mole of NaCl (s) on melting absorbed 30.5 kJ of heat and its entropy is increased by 28.8 JK^(-1). The melting point of NaCl is |
|
Answer» 1059 K Given that : `DeltaH=30.5 kJ mol^(-1)` `DeltaS=28.8 JK^(-1)=28.8xx10^(-3)kJ K^(-1)` By USING `DeltaS=(DeltaH)/(T)=(30.5)/(28.8xx10^(-3))=1059 K`. |
|
| 5. |
One mole of N_(2)O_(4) is heated in a flask with a volume of 10dm^(3). At equilibrium 1.708 mole of NO_(2) and 0.146 mole of N_(2)O_(4) were found at 134^(@)C. The equilibrium constnt will be |
|
Answer» 250 mol `dm^(-3)` `{:("Moles at "eqb^(m),0.146,1.708),(Conc.,,),((mol//dm^(3)),[(0.146)/(10)],[(1.708)/(10)]):}` `thereforeK_(c)=([NO_(2)]^(2))/([N_(2)O_(4)])=([(1.708)/(10)])/([(0.146)/(10)])` `impliesK_(c)=(2.917264)/(100)xx(10)/(0.146)` `IMPLIES K_(c)=(2.917264)/(1.46)=2"mol dm"^(-3)` |
|
| 6. |
One mole of N_2O_4 (g) at 300k is kept in a closed container under one atmospheric pressure. It is heated to 600 k when 20 % by mass of N_2O_4 (g) decomposes to NO_2(g) . The resultant pressure is : |
|
Answer» 1.2 atm |
|
| 7. |
One mole of N_(2)and 3 moles of H_(2)are mixed in a litre flask. If 50% N_(2)is converted into ammonia by the reaction.N_(2)(g)+3H_(2)(g)hArr 2NH_(3)(g)then the total number of moles of gas at equilibrium is : |
|
Answer» `1.5` |
|
| 8. |
One mole of monoatomic gas taken through a cyclic process as shown in figure. Calculate sum_(ABCA) = (dq_(rev))/(T). |
|
Answer» |
|
| 9. |
One mole of monoatomc ideal gas expands adiabatically at initial temp. T against a constant external pressure of 1 atm from one litre to two litre. Find out the final temp. (R = 0.0821 litre. "atm"K^(-1)mol^(-1)): |
|
Answer» T |
|
| 10. |
One mole of methanol when burnt in O_(2), gives out 723 kJ mol^(-1) heat. If one mole of O_(2) is used, what will be the amount of heat evolved |
|
Answer» 723 kJ By burning with `(3)/(2)O_(2)(g)` heat evolved =-723 kJ `therefore` By burning with 1 mole `O_(2)(g)=(-723xx2)/(3)=-482 kJ`. |
|
| 11. |
One mole of methannol when burnt in oxygen gives out 723 kJ mol^(-1) heat. If one mole of oxygen is used, what will be the amount of heat evolved ? |
|
Answer» 723 kJ 1 mol of `O_(2) = (2)/(3)` mol of `CH_(3)OH` :. `(2)/(3)` mol of `CH_(3)OH` on COMBUSTION gives `= (723 xx 2)/(3)` = 482 kJ |
|
| 12. |
One mole of magnesium in the vapour state absorbed 1200 kJ mol^(-1) of energy. If the first and second ionization energies of Mg are 750 and 1450 kJmol^(-1) respectively, the final composition of the mixture is |
|
Answer» `31% Mg^(+)+69% Mg^(2+)` 1 g of Mg (s) absorbs = `(1200)/(24)=50 kJ` `therefore` Energy required to CONVERT Mg (g) to `Mg^(+)`(g) `=0.0417xx750=31.275 kJ`. `therefore`Remaining energy = 50 - 31.275 = 18.725 kJ Number of moles of `Mg^(2+)` formed = `(18.725)/(1450)=0.013` Thus remaining `Mg^(+)` will be = `0.0417 - 0.013 = 0.0287` `therefore % Mg^(+)=(0.0287)/(0.0417)xx100=68.82%` `% Mg^(2+)=100-68.82 = 31.18%`. |
|
| 13. |
Onemole of magnesium nitride on reaction with excess of water gives |
|
Answer» TWO moles of ammonia ` therefore ` 2 mol of `NH_3` |
|
| 14. |
One mole of liquid A (vapour pressure =300 mm Hg ) and 2 moles of a liquid B (Vapour pressure =360 mm Hg) are mixed . The mole fraction of A in the vapour mixture if found to be 5//17 . Which of the following is correct ? |
|
Answer» `(DeltaH)_("mix")gt0` |
|
| 15. |
One moleof ideal gas undergoes following cyclic process (i) Isochoric heating from (P_(1)V_(1)T_(1)) to double temperature.(II)Isobaric expansion from todouble voulme . (III) Linear expansion( on PV curve) to (P_(1),8V_(1)).Isobaric copmression to initial state.Calculate the magnitudeof work done in caloriesif initialtremperature of the gas in is300K?Given : R=2(Cal)/(mol- K) |
|
Answer» Solution :Magniudeof work =`A=P_(1)V_(1)+(1)/(2)xxP_(1)xx 6V_(1)` `=4xx1xx2xx300` `=4xx1xx2xx300` `=2400 CAL` |
|
| 16. |
One mole of ideal monatomic gas is carried through the reversible cyclic process as shown in figure. Calculate the max temperature attained by the gas during the cycle. |
|
Answer» `(25)/(8)((P^(@)V^(@))/(R))` `(y-y_(1)) = (Y_(2)-Y_(1))/(x_(2)-x_(1))(x-x_(1))` `(P-3P^(@))=(P^(@)-3P^(@))/(2V^(@)-V_(@))(V-V^(@)) implies (P-3P^(@)) =-(2P^(@))/(V^(@))(V-V^(@)) ""…..(1)` Replacing P by using `P=(RT)/(V)` in equation (1) `((RT)/(V)-3P^(@))=-(2P^(@))/(V^(@)) (V-V^(@)) implies T=(2P^(@)V(V-V^(@)))/(V^(@)R) + (3P^(@)V)/(R)` For T to be maximum `(dT)/(DV)=0` `(dT)/(dV) =-(2P^(@)(2V-V^(@)))/(V^(@)R)+ (3P^(@))/(R) =0 implies V=(5)/(4)V^(@)` `T_("max")=-(2P^(@)xx(5)/(4)V^(@)((5)/(4)V^(@)-V^(@)))/(V^(@)R)+(3P^(@)(5)/(4)V^(@))/(R)=(25)/(8)((P^(@)V^(@))/(R))` |
|
| 17. |
One mole of idealmonatomicgas in takenin cycliceprocess ABCA as shownin the figure Calculate :(a) Theworkdone by thegas(b) The heatrejectedbythegas in the pathCA (c) The net heatabsorbedbe the bygas in the path BC (d)Maximum temperature obtainedby thegas duringthe cycle. |
|
Answer» |
|
| 18. |
One mole of ideal gas is allowed to expand reversibly and adiabatically from a temperature of 27^(@)C. If the work done by the gas in the process is 3 kJ, the final temperature will be equal to (C_(v)=20J//Kmol) |
|
Answer» 100 K |
|
| 19. |
One mole of ice is coverted into water at 273 K. The entropies of H_(2)O(s) and H_(2)O(l) are 38.20 and 60.01 J mol^(-1) K^(-1) respectively. The enthaply change for the conversion is |
|
Answer» `59.54 J mol^(-1)` `DELTA S = S[H_(2)O(l)] - [H_(2)O(s)]` `60.01 - 38.20` `= 21.81 J mol^(-1)` `Delta H = T Delta S = 21.81 xx 273` `= 5954 J mol^(-1)` |
|
| 20. |
One mole of ice is converted into water at 273 K. The entropies of H_2O(s) and H_2O(l) are 38.20 and 60.01 J mol^-1 K^-1 respectively. The enthalpy change for the conversion is: |
|
Answer» `59.54 J mol^-1` |
|
| 21. |
One mole of hydrocarbon (A) reacts with one mole of bromine giving a dibromocompound C_(5)H_(10)Br_(2). Substance (A) on treatment with cold, dilute alkaline KMnO_(4) solution forms a compound C_(5)H_(12)O_(2). On ozonolysis, (A) gives equimolar quantities of propanone and ethanal. Deduce the structural formula of (A) |
Answer» SOLUTION :
|
|
| 22. |
One mole of hydrogen peroxide (H_2O_2)has a mass same as that of |
|
Answer» 0.1 mol of sucrose ` (C_12H_22O_11)` 2 mol of `NH_3 = 2 xx 17= 34 g ` |
|
| 23. |
one mole of hydrogen iodide is heated in a closed container of 2 litre. At equilibrium half mole of hydrogen iodide has dissociated. What is the value of the equilibrium constant ? |
|
Answer» 1 |
|
| 24. |
One mole of Helium gas undergoes a reversible cyclic process ABCDA as shown in the figure.Assuming gas to be ideal, answer the following questions : Polytropic process for ideal gas is given as PV^(th)=constant. For polytropic process for an ideal gas.the expression for work obtained is : w=(P_1V_1[(V_2/V_1)^x-1])/((y)) Report your answer as (x+y). |
|
Answer» 0 `=-INT K/V^n dV=-K/(1-n)[V^(-n+1)]_(V_1)^(V_2)=K/(n-1)[V_2^(1-n)-V_1^(1-n)]` `=(P_1V_1^n)/(n-1)[V_2^(1-n)-V_1^(1-n)]=(P_1V_1^(n+1-n))/(n-1)[(V2/V_1)^(1-n)-1]` |
|
| 25. |
One mole of Helium gas undergoes a reversible cyclic process ABCDA as shown in the figure.Assuming gas to be ideal, answer the following questions : What is the value of 'q' for the overall cyclic process: |
|
Answer» `-100 Rln2` `=-1Rxx30ln2+1xx(5R)/2xx(400-300)+1Rxx400ln2+1xx(5R)/2xx(300-400)` (`because q_(AB)=-W_(AB)=-1Rxx300ln2` since process is reversible isothermal for which `DeltaU=0`) (`because q_(BC)=DeltaH_(BC)=1xx(5R)/2xx(400-300)` since process is reversible ISOBARIC ) (`because q_(CD)=-W_(CD)=1Rxx300ln2` since process is reversible isothermal for which `DeltaU=0` ) (`because q_(DA)=DeltaH_(AB)=1xx(5R)/2xx(300-400)` since process is reversible isobaric ) So, q=100 Rln2 |
|
| 26. |
One mole of Helium gas undergoes a reversible cyclic process ABCDA as shown in the figure.Assuming gas to be ideal, answer the following questions : What is the net work involved in the process A to C : |
|
Answer» `-100 R(1-ln8)` |
|
| 27. |
One mole of Helium gas undergoes a reversible cyclic process ABCDA as shown in the figure.Assuming gas to be ideal, answer the following questions : What is the value of DeltaH for the overall cyclic process: |
|
Answer» `-100 Rln2` |
|
| 28. |
One mole of fluorine is reacted with two moles of hot and connectrated KOH . The products formed are KF, H_(2)O and O_(2). The molar ratio of KF, H_(2)O and O_(2) respectively is |
|
Answer» `1 : 1 : 2` `{:(F_(2),KOH,KF,O_(2),H_(2)O),(1,2,2,(1)/(2),1):}` |
|
| 29. |
One mole of glucose on respiration produces : |
|
Answer» 36 MOLE of ATP |
|
| 30. |
One mole of ethyl alcohol was treated with one mole of acetic acid at 25^(@)C. Two-third of the alcohol change into easter at equlibrium. The equlibrium constant for the reaction will be |
|
Answer» 1 `{:("Initial CONC.","1 mol","1 mol","0 MOLE","0 mole"),("At equlibrium", 1-2/3=1/3 "mol",1/3"mol",2/3"mol",2/3"mol"):}` `K_(C)=((2)/(3)xx(2)/(3))/(1/2xx1/3)=4` |
|
| 31. |
One mole of ethyl alcohol was treated with one mole of acetic acid at 25^@C. 2/3of the acid changes into ester at equilibrium Calculate the equilibrium constant for the reaction : |
|
Answer» 1 |
|
| 32. |
One mole of ethyl acetate on treatment with an excess of LiAIH_4 in dry ether and subsequent acidification produces |
|
Answer» 1 mol ACETIC acid + 1 mol ETHYL alcohol |
|
| 33. |
One mole of each PdCl_(2).4NH_(3), PtCl_(2).2NH_(3),PtCl_(4).2HCl and CoCl_(3).4NH_(3) gave 2, 0, 0 and 1 mole of AgCl, respectively when excess of AgNO_(3) solution is added. Assign the secondary valences to the metals in these complexes. |
| Answer» SOLUTION :4, 4, 6 and 6 | |
| 34. |
One mole of each of the following alkenes is catalytically hydrogenated. The quantity of heat evolved will be the lowest in the case of |
|
Answer» 1-butene |
|
| 35. |
One mole of CrCl_(3).6H_(2)O compound reacts with excess AgNO_(3) solution to yield two moles of AgCl (s). The structural formula of the compound is |
|
Answer» `[Cr(H_(2)O)_(5)Cl]Cl_(2).H_(2)O` |
|
| 36. |
One mole of Co(NH_(3))_(5)Cl_(3) gives 3 moles of ions on dissolution in water. One mole of this reacts with two moles of AgNO_(3) to give two moles of AgCl. The complex is : |
|
Answer» `[Co(NH_(3))_(4)Cl_(2)]Cl.NH_(3)` |
|
| 37. |
One mole of complex compound Co(NH_(3))_(5)Cl_(3) gives 3 moles of ions on dissolution in water. One mole of the same complex reacts with two moles ofAgNO_(3) solutionto yield two moles of AgCl(s). The structure of the complex is : |
|
Answer» `[Co(NH_(3))_(3)Cl_(3)].2NH_(3)` |
|
| 38. |
One mole of CO_(2) gas at 27^(@) C is compressed suddenly so that its pressure becomes 8/27 of its original pressure. Final temperature of gas would be |
|
Answer» `300 XX (2/3)^(0.33)` |
|
| 39. |
One mole of CO_2corresponds to : |
|
Answer» 22.4 L at 1 ATM and `25^@C` |
|
| 40. |
One mole of Cl_(2(g)) which may be assumed to obey the ideal gas law, initially at 300 K and 1.01325 xx 10^(7) Pa, is expanded against a constant exteranl pressure of 1.01325 xx 10^(5) Pa to a final pressure of 1.01325 xx 10^(6) Pa . As a result of the expansion , the gas cooled to a temperature of 239 K (which is the normal boiling point of Cl_(2)) and 0.100 mol of Cl_(2) condensed. The enthalpy of vaporization of Cl_(2(l)) is 20.42 KJ "mol"^(-1) at the normal volume is C_(v)=28.66 JK^(-1) "mol"^(-1) and the density of Cl_(2(l)) is 1.56 g cm^(-1)(at 239 K) . Assume that the molar heat capacity at constant pressure for Cl_(2(g)) is C_(p)=C_(v)+R. (1 atm = 101325 xx 10^(5) Pa , R=8.314510 Jk^(-1) "mol"^(-1) = 0.0820584 L atm K^(-1)"mol"^(-1)) (i) Either draw a complete molecular orbital energy diagram or write the complete electronic configuration of Cl_(2) Predict the bond order of Cl_(2) and thus whether this molecules will be diamagnetic , ferromagnetic ,or paramagnetic. (ii) For the changes decribed above, calculate the change in the internal energy (DeltaE) and the change in the entropy (DeltaS_("sys")) of the system. |
|
Answer» |
|
| 41. |
One mole of CO_2contains |
|
Answer» ` 6.02 XX 10^23`atoms of C ` = 6.02 xx 10^23` atoms of C |
|
| 42. |
One mole of calcium phosphide on reaction with excess water gives: |
|
Answer» ONE mole of PHOSPHINE |
|
| 43. |
One mole of calcium phosphide on reactionwith excess of water gives |
|
Answer» ONE mole of phosphine |
|
| 44. |
Onemole of calcium phosphideon reaction with excess of water gives |
|
Answer» ONE mole ofphosphine |
|
| 45. |
One mole of As_(2)O_(5) on treatment with furning hydrochloric acid gives how much mole of Cl_(2)? |
|
Answer» |
|
| 46. |
One mole of any substance contains 6.022xx10^(23) atoms/molecules. Number of molecules of H_(2)SO_(4) present in 100 mL of 0.02 M H_(2)SO_(4) solutions is …………….. . |
|
Answer» `12.044xx10^(20)" molecules"` `"100 mL of 0.02 M "H_(2)SO_(4)" contain "H_(2)SO_(4)=(1)/(1000)xx100xx0.02" mole"=0.002" mole"=2xx10^(-3)" mole"` `=2xx10^(-3)xx(6.022xx10^(23))" molecules"=12.044xx10^(20)" molecules."` |
|
| 47. |
One mole of anhydrous salt AB dissolves in water and liberates 15 J mol^(-1) of heat .The value of Delta H_(hydration)^° of AB is -20.05 J mol^(-1) .Hence the enthalpy of dissolution of hydrated salt AB.3H_2O(s) is |
|
Answer» `-5.5J` `MOL^(-1)` |
|
| 48. |
One mole of aniline warmed with the mixture of NaNO_(2)+HCl. If we assume 100% yield, volume of N_(2) gas liberated at S.T.P is |
|
Answer» `11.2 L` |
|
| 49. |
One mole of an unsaturated hydrocarbon on reductive hydrolysis gives one mole each of formaldehyde, acetaldehyde and methyl glyoxal. The hydrocarbon is |
|
Answer» `CH_(2)=CH-CH=CH-CH_(3)` 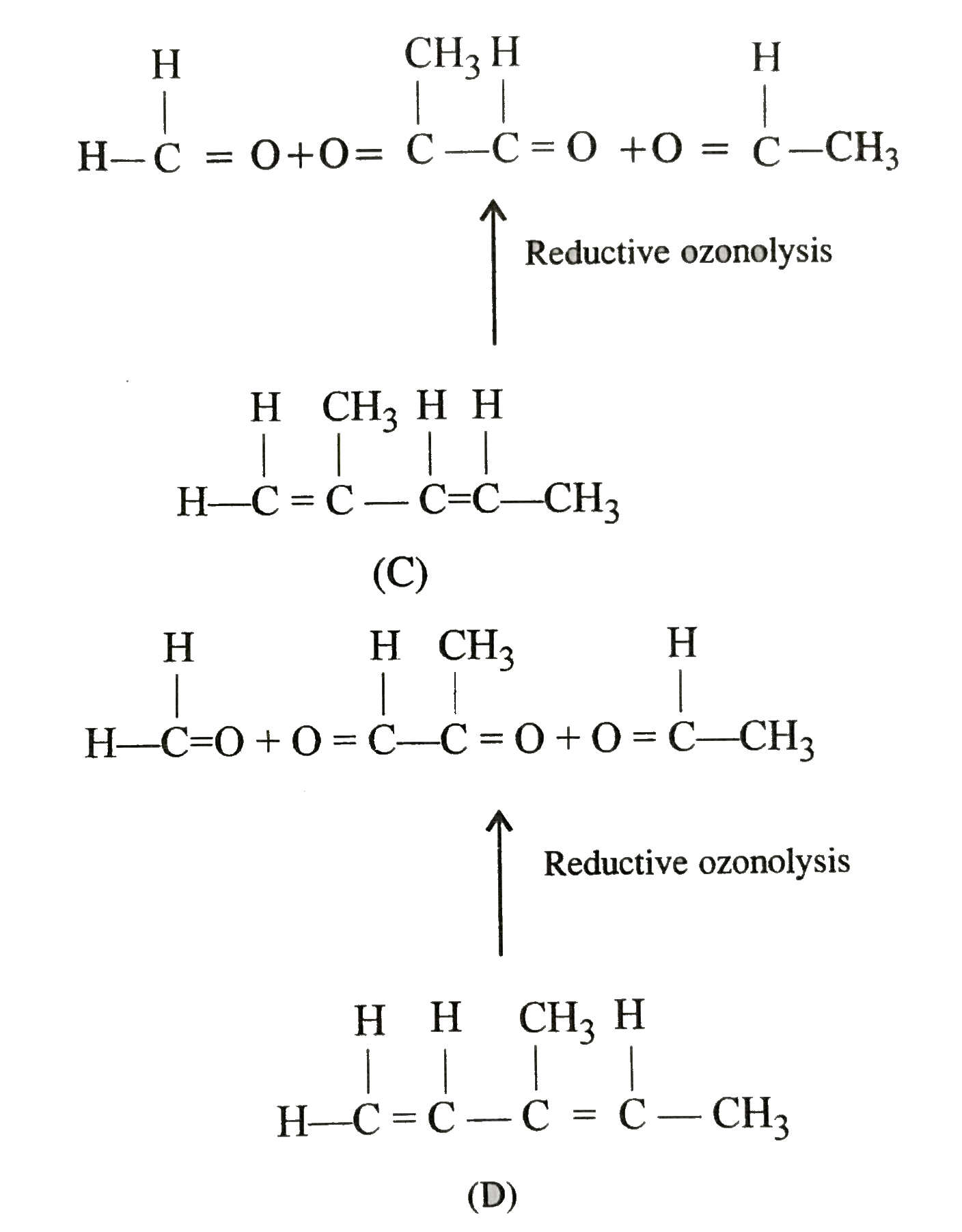
|
|
| 50. |
One mole of an organic compound requires 0.5 mole of oxygen to produce an acid.The compound may be: |
|
Answer» Alcohol |
|