Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
One mole of a non-ideal gas undergoes change of state (2.0 atm, 3.0L, 95(K)rarr(4.0 atm, 5.0 L, 245 K) with a change in internal energy, DeltaU=30.0 L atm. The change in enthalpy (DeltaH) of the process in L atm is |
|
Answer» 40 `=30+4xx5-2xx3=44 L//atm`. |
|
| 2. |
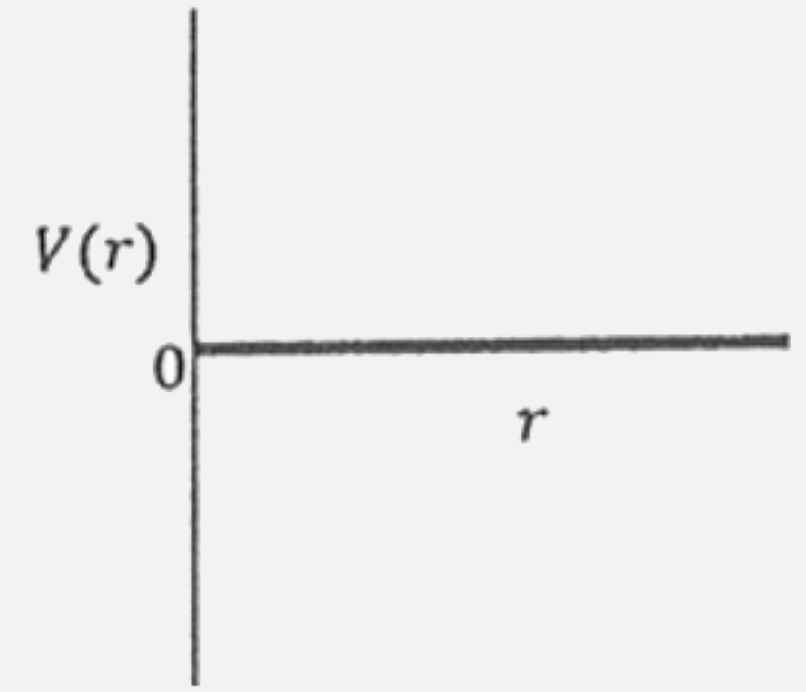
One mole of amonoatomic real gas satisfies the equation p(V-b)=RT where b is a constant. The relationship of interatomic potential V(r) and interatomic distance r for gas is given by |
|
Answer»
|
|
| 3. |
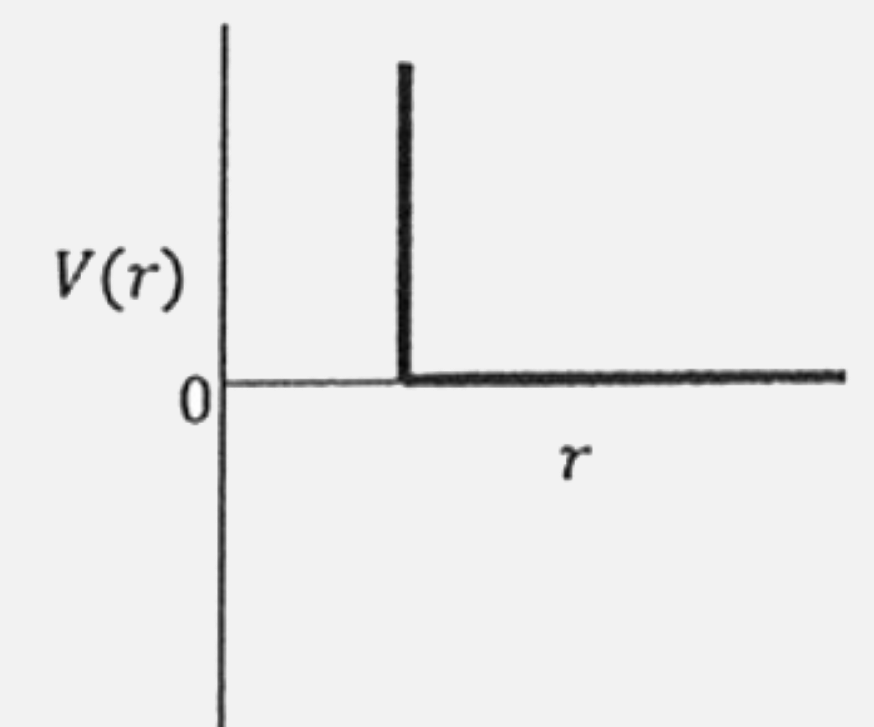
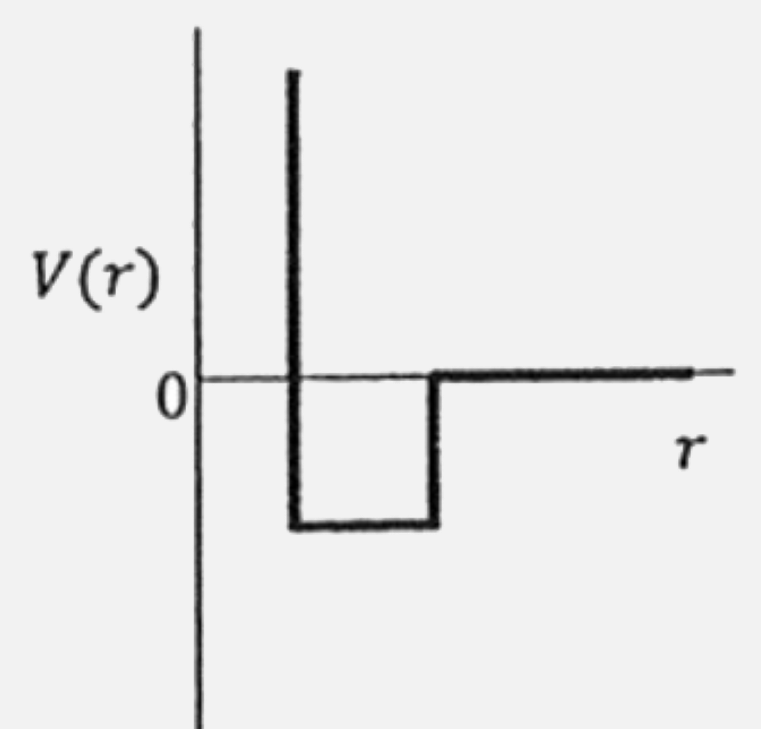
One mole of a monoatomic real gas satisfies the equation p(V -b) = RT where b is a constant. The relationship of interatomic potential V(r ) and interatomic distance r for the gas is given by |
|
Answer»
`rArr PV = PB = RT` `rArr (PV)/(RT) = (Pb)/(RT) +1` `rArr Z = 1+(Pb)/(RT)` Hence `Z gt 1` at all pressure. This means, repulsive tendencies will be dominant when interatomic distance are small. This means, interatomic potential is never negative but BECOMES positive at small interatomic distances. Hence answer is (C ) |
|
| 4. |
One mole of a monoatomic ideal gas is taken through a cycle ABCDA as shown in the P-V diagram. A student wants to determine sign of measurable quantities DeltaU, q, W and DeltaS for reach process separately. If x is the total number of negative values of measurable quantities in overall cycle, then calculate x. |
|
Answer» |
|
| 5. |
One mole ofamonoatomicgas behving ideally is usedas workingsubstancein anengine workingin thecycle as shown in the figure. TheprocessAB, BC, CD and DA are respectively reversible isobaric, adiabatic isochoric and isothermal . The ratioofmaximum tovolume and temperatureduring the cycle is 8sqrt(2) and 4 respectively. Ifthe maximum T is 800 Kand gamma =5//3. Calculate DeltaE (in kJ) forthe process BC.Given R= 8.3 J//K-mol. |
|
Answer» Min. volume at `V_(C)` & `V_(D)` `(V_(A))/(V_(C))= 8sqrt(2)` `V_(A)= 8sqrt(2) V_(C)` nin.Temp .atB max. temp at D or A `(T_(D))/(T_(B))= 4 or(T_(A))/(T_(B))= 4` `therefore T_(B) = 200 K` During isobaric processAb `(V_(A))/(V_(B))= (T_(A))/(T_(B)) rArrV_(B) (T_(B))/(T_(A))= (8sqrt(2))/(4) V_(C) = 2 sqrt(2) V_(C)` During ADIABATIC proessBC `T_(C)= T_(B) ((V_(B))/(V_(C)))^(r-1)= 200 (2sqrt(2))^((5)/(3)-1)=400 K` `= 400 K` `DELTAE= (3)/(2)R[400-200]` `= 2494 .2 J = 2.494 kJ` |
|
| 6. |
One mole of a monatomic gas at pressure 2 atm ,279 K taken to final pressure 4 atm by a reversible path described by P//V = constant . Calculate the magnitude of (DeltaE)/(w) for the process . |
|
Answer» |
|
| 7. |
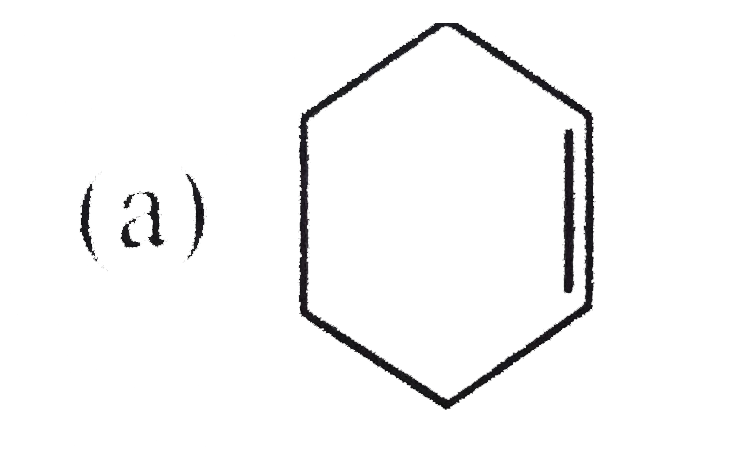
One mole of a hydrocarbon (A) on ozonolysis gives two molesof dicarbonylcompound (A) is : |
|
Answer»
|
|
| 8. |
One mole of a gas occupying 3 dm^3 expands against constant external pressure of 1 atm to a volume of 13 dm^3. The work done is: |
| Answer» Answer :A | |
| 9. |
One mole of a gas is taken from state A to state B as shown in figure. Work done by the gas is alpha xx 10^(beta) J. Find the value of alpha + beta. (Given: T_(1) = 320 K, R = (25)/(3)) |
|
Answer» |
|
| 10. |
One mole of a compound AB reacts with one mole of a compound CD accoeding to the equation AB+CDhArrAD+CB. When equilibrium had been estblished it was found that 3/4 mole each of rectant AB and CD had been converted to AD and CB. There is no change in volume. The equlibrium constant for the reaction is |
|
Answer» `9/16` `{:("MOLE at t=0",1,1,0,0),("Mole at equlibrium",(1-(3)/(4)),(1-(3)/(4)),((3)/(4)),((3)/(4))),(,0.25,0.25,0.75,0.75):}` `K_(C)=(0.75xx0.75)/(0.25xx0.25)=(0.5625)/(0.0625)=9` |
|
| 11. |
One mole of a gas absorbs 200 J of heat at constant volume. Its temperature rises from 298 K to 308 K. The change in internal energy is: |
| Answer» Answer :A | |
| 12. |
One mole o N_(2)O_(4) gasat 300is keip in a closed ctainer at 1 atm. It is heatedto 600 K when 20% by mass of N_(2)O_(4) decomposes to NO_(2(g)). The resultant pressure in the container would be |
|
Answer» `1.2` ATM Total number of moles at equlibrium `=1.2` INITIALLY, `P_(1)V=n_(1)RTor 1xxV=1xxRT_(1)or V=RT_(1)` At equlibrium, `P_(2)V=n_(2)RT_(2)` `or,P_(2)=(1.2)/(V)RT_(2)=(1.2)/(RT_(1))RT_(2)=(1.2xx600)/(300)=2.4atm.` |
|
| 13. |
One mole monoatomic ideal gas was takenthroughprocess ABCD as show in. Calculate(i)W_(AB), W_(BC), W_(CD),W_(DA)(ii)q_(AB), q_(BC), q_(CD),q_(DA) (iii)DeltaH_(AB), DeltaH_(BC), DeltaH_(CD),DeltaH_(DA) [Use :ln(3//2) = 0.40 : In (4//3) = 0. 29] |
|
Answer» Solution :(i) `W_(AB)` : Along 'AB' pressure is variable from graph :`(V-10)/(T- 300) = (15- 10)/( 600-300) rArr60 V- 600 = T - 300 rArr 60 V - 300 =T` sub value ofT in ideal gas equation `PV= R (60 V - 300)""rArr""P= 60R-(300R)/(V)` `W_(AB) = - underset(10)overset(15)int PdV = -underset(10)overset(15)int[60R-(300R)/(V)] DV= - 60R(V_(2)- V_(1)) + 300R ln((V_(2))/(V_(1)))` `= - 300R + 300 R(0.4)""("taken ln"(3//2)= 0.10 "data")` `W_(AB) = - 180 R = - 1496.52 J` `W_(BC) = - RT ln ((V_(3))/(V_(2))) = -RT(4//3) "" ("taken ln "(4//3)= 0.29"data")` `= - 8.314 xx 600 xx 0.29 = - 1446.63 "Joule"` `W_(CD)= 0` `W_(DA) = - NRT ln ((V_(1))/(V_(2))) = - 1 xx 8.314 xx 300 ln ((10)/(20)) = + 1728.84"Joule"` (ii) `q_(AB)= DeltaE_(AB)- W =(3)/(2) R xx 300 + 1496.52=450R +180R=630R=5237.82"Joule"` `q_(BC) = - W_(BC) = +1446.63"Joule"` `q_(CD)= DeltaU = nC_(V)DeltaT = 1 xx (3)/(2)R xx(300-600)= -3741.3"Joule"` `q_(DA) = - W_(DA) = 1728.84"Joule"` (iii)`DeltaH_(AB) = C_(P) (T_(2) - T_(1)) =(5)/(3) R xx 300 = 750R = 6235.5"Joule"` `DeltaH_(BC) =0 ` `DeltaH_(CD)= DeltaU_(CD) + nR DeltaT = nC_(p) DeltaT = 1 xx (5)/(2) R xx (-300)= - 6235.5"Joule"` `DeltaH_(DA)= DeltaU_(DA) + nRDelta T = 0"Joule"` |
|
| 14. |
One mole each of urea, glucose and sodium chloride were dissolved in one litre of water Equal osmotic pressure will be produced by solutions of |
|
Answer» Glucose and sodium CHLORIDE |
|
| 15. |
One mole each of urea, glucose and sodium chloride were dissolved in one litre of water . Equal osmotic pressure will be produced by solutions of : |
|
Answer» UREA and glucose |
|
| 16. |
One mole each of urea, glucose and sodium chloride were dissolved in one litre of water. Equal osmotic pressure will be produced by solutions of |
|
Answer» UREA and glucose |
|
| 17. |
One mole Benzene in presence of light on reaction with 'x' moles of Cl_2 forms B.H.C. what is 'x' ? |
Answer» 
|
|
| 18. |
One mole each of a monoatomic, diatomic and triatomic gases are mixed, C_p/C_v for the mixture is : |
|
Answer» 1.4 |
|
| 19. |
One mole benzene diazonium chloride is treated with HBr(excess) in the presence of CuBr. Now, volume of N_(2) liberated at STP weighs |
|
Answer» 11.2 lit. |
|
| 20. |
One molar solution of sodium chloride will have the relative lowering of vapour pressure closest to: |
|
Answer» `5.8%` (wt//vol.) UREA SOLUTION |
|
| 21. |
One molar solution of sodium chloride will have the relative lowering of vapour pressure closest to : |
|
Answer» <P>5.8% (wt./vol) urea SOLUTION (M.M.=60) mole FRACTION of NACI and urea should be same. |
|
| 22. |
One molal solution of a complex of cobalt chloride with NH_(3) in water showed an elevation in boiling point equal to 2.08^(@). Assuming that the complex is completely ionized in the solution, the complex is (K_(b) for water = "0.52 K kg mol"^(-1)) |
|
Answer» `[CO(NH_(3))_(6)]Cl_(3)` `2.08xxixx0.52xx1 or i=4` This means that the complex produces 4 ions in the solution. Hence, the complex is `[Co(NH_(3))_(6)]Cl_(3)`. |
|
| 23. |
One molal solution of a complex of cobalt chloeide with NH_(3) in aqueous s0olution shows boiling point elevaton of 2.08^(@). Assuming that the complex is compleltely ionised in solution, the xomplex is (K_(b)"for water"=0.52 K kg mol^(-1)). |
|
Answer» `[Co(NH_(3))_(6)]CI_(3)` This shows that the complex produces FOUR ions in aqueous solution. Correct option is (a). `[CO(NH_(3))_(6)]CI_(3)overset(("AQ"))HARR[CO(NH_(3))_(6)]^(+)+3CI^(-)` |
|
| 24. |
One moel of an organic compound (A) with the formula C_3H_8O reacts completel with two moles of HI form X and Y. When Yis boiled with aqueous alkali it forms Z.Z answers the indoform test. Thecompound (A) is …………………. . |
|
Answer» PROPAN - 2 - ol 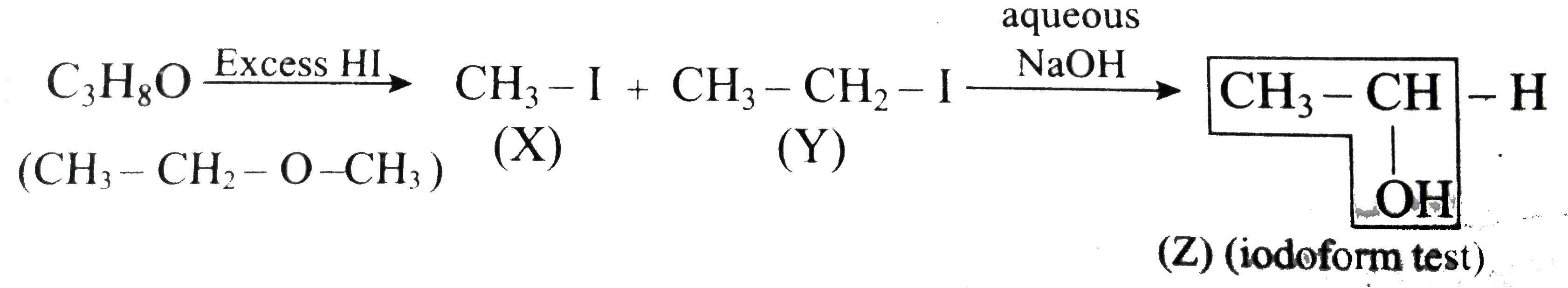
|
|
| 25. |
One mL of an organic compound was dissolved in ethanol and a very small drop of dilute alkali was added to it and them a drop of phenolphalein was added. The red colour of the indicator was seen . The mixture. was then heated . The colour disappeared in a few minutes . The organic compound is most likely to be |
|
Answer» an ALDEHYDE |
|
| 26. |
One microgram of radioactive sodium, ""_(11)^(24)Na with half life of 15 hour was injected into a living system. How long will it take for the radioactivity to fall to 25% of the initial value ? |
|
Answer» 22.5 hr |
|
| 27. |
One method to produce hydrogen on an industrial on an industrial scale is the reaction of methane with overheated water vapour at 1100 K to form hydrogen and carbon monoxide . The reaction is known as steam reforming The K_(p)of the reforming reaction at 1100K is 28.6 When 1 mol of methane and 1 Kmol fo water arereacted at 1100 K , calculate the percentage conversion of methane at equilibrium at a total pressure of 1.6 bar. In another experiment 1.0 Kmol of CH_(4) and 1.0 mol of H_(2)O are taken in a sealed vessel at 400 K and 1.6 bar. The temperature is raised to 1100K. |
|
Answer» |
|
| 28. |
One method to produce hydrogen on an industrial on an industrial scale is the reaction of methane with overheated water vapour at 1100 K to form hydrogen and carbon monoxide . The reaction is known as steam reforming Calcuate the pressure in the vessel at 1100K and the % conversion of methane. |
|
Answer» |
|
| 29. |
One metal possess bcc arrangement. Its unit cell has edge length 3.04 Å, then the volume of unit cell = ....... "cm"^3. |
|
Answer» `2.81 xx 10^(-23)` |
|
| 30. |
One litre solution X has x g of urea dissolved in it. Another one litre solution Y has xg of KCl dissolved in it. Find the ratio of lowering of vapour pressures of the solutions X and Y. |
|
Answer» Solution :The ratio of concentration = The ratio of number of MOLES The MOLE ratio =`x/60:x/74.6=7.46 :6` Urea is unionised even in dilute solutions and hence, van.t HOFF factor, i = 1. KCL is almost ionised in dilute solutions and hence, van.t Hoff factor, i 2. Hence the ratio of particles in two solutions 7.46 : 6 x 2 =7.46 : 12 =1 : 16 Since lowering of vapour pressure is a colligative PROPERTY, the ratio of lowering = 1: 1.6 |
|
| 31. |
One litre solution of MgCl_(2) is electrolyzed completely by passing a current of 1A for 16 min 5 sec.The ogiginal concentration of MgCl_(2) solution was(Atomic mass of Mg=24) |
|
Answer» `5 xx 10^(-3)` M = 1 `xx` 965 = 965 C w = ZIt(where w= amount of substance deposited) `w = (12)/(96500) xx 965 ( :' Z = (E)/(96500)) = (12)/(100)` Conc. of `MgCl_(2) = ("Weight")/("Mol.mass" xx V(L)) = (12)/(100xx24xx1)` `= 1/(200) = 5 xx 10^(-3)` M |
|
| 32. |
One litre solution of alkali ( NaOH ) is prepared by dissolving impure solid of alkali which contian 5% Na_(2) CO_(3) and 8% CaCO_(3) and 10%NaCl. A 10ml portion of this solution required 9.8 ml of a 0.5 M H_(2)SO_(4) solution for neutralization. Calculate weight of alkali dissolved initially gm eq. of Na_(2) CO_(3) + gm eq. of CaCO_(3) + gm eq. of NaOH = gm of H_(2) SO_(4) |
|
Answer» SOLUTION :`(5x)/( 100) XX ( 1)/( 53) + ( 8 x)/( 100) xx ( 1)/( 50) + (77x)/( 100) xx( 1)/( 40) = 0.98` `x = 45 g ` |
|
| 33. |
One litre of water was added to 500mL of 32% HNO_(3) of density 1.20g//mL . What is the percent concentration of HNO_(3) in the solution obtained ? |
|
Answer» |
|
| 34. |
One litre of water contains 10^-7 mol hydrogen ions. The degree of ionisation in water will be |
|
Answer» `1.8 TIMES 10^-7` So, degree of IONISATION `=(10^-7 times 18)/1000=1.8 times 10^-7` |
|
| 35. |
One litre of water contains 10^(-7) mole hydrogen ions. The degree of ionization in water will be |
|
Answer» `1.8 xx 10^(-7)%` So degree of ionization `= (10^(-7) xx 18)/(1000) = 1.8 xx 10^(-7)%` |
|
| 36. |
One litre of oxygen at STP is made to react with three litres of carbon monoxide at STP. Calculate the mass of each substance found after the reaction. Which one is the limiting reactant? |
|
Answer» Solution :The chemical equation representing the reaction is : `UNDERSET("2 vol.")(2CO)+underset("1 vol.")(O_(2))RARR underset("2 vol.")(2CO_(2))` Step 1. To calculate the VOLUME and mass of CO (at STP) left unused after the reaction. Applying Gay Lussac's Law of Gaseous Volumes, 1 Vol. of `O_(2)` reacts with CO = 2 Vol. `therefore"1 litre of "O_(2)" will react with CO = 2 litres at STP"` `"But the volume of CO taken = 3 litre at STP"` `therefore"Volume of CO (at STP) left unused "=3-2-1" litre."` Now, by mole concept, mass of 22.4 litres of CO at STP = Molecular mass is grams `=12+16=28g` `therefore"Mass of 1 litre of CO at STP"=(28)/(22.4)xx1=1.25g` Thus, the mass of CO left unused = 1.25g Step 2. To calculate the volume and mass of `CO_(2)` formed from 1 litre of `O_(2)` at sTP. Applying Gay Lussac's Law of Gaseous Volumes, 1 Vol. of `O_(2)` produces `CO_(2)=" 2 Vol."` `therefore"1 litre of "O_(2)" will produce CO"_(2)=" 2 litres at sTP"` By mole concept, Mass of 22.4 litres of `CO_(2)` at STP = Molecular mass in grams `=12+2xx16=44g` `therefore"Mass of 2 litres of "CO_(2)" at STP"=(44)/(22.4)xx2=3.928g` Thus, the mass of `CO_(2)` produced = 3.928g. Step 3. As oxygen has been COMPLETELY used up, hence oxygen is the limiting REACTANT. |
|
| 37. |
one litre of water contains 10^(-7) mole H^+ ions what is degree of ionisation of water ? |
|
Answer» `1.8xx10^(-7)%` |
|
| 38. |
One litre of N/2 HCl solution is heated in a beaker. It was observed that when the volume of the solution was reduced to 600 ml, 3.25 g of HCl is lost. Calculate the normality of the new solution. |
|
Answer» SOLUTION :`"1 L of "(N)/(2)" HCl contain HCl "=(1)/(2)xx"36.5 g = 18.25 g , HCl now present "=18.25-3.25g=15g` `"NEW volume = 600 ML = 0.600 L,Normality "=(15)/(36.5)" mol"xx(1)/(0.600L)=0.685N.` |
|
| 39. |
One litre of milk weight 1.035 kg. The butter fat is 4% (v/v) of milk and has density of 875 kg//m^(3). If the density of fat free skimed milk is 'x' kg//m^(3), the value of (4.8 x) is ? |
|
Answer» |
|
| 40. |
One litre of milk weighs 1.035kg. The butter fat it contains to the extent of 4% by volume has a density of 875kg//m^(3). Find the density of the fat-free skimmed milk . (1m^(3)=10^(3) litres) |
|
Answer» SOLUTION :Weight of `1m^(3)` milk `=1.035xx10^(3)=1035kg` Weight of the FAT in `1m^(3)` of milk `=0.04xx875kg` (`1m^(3)` milk CONTAINS `0.04m^(3)` of fat ) `=35kg` `:.` density of skimmed milk `=("wt.of skimmed milk")/("vol.of skimmed milk")` `=((1035-35)kg)/((1-0.04)m^(3))=1042kg//m^(3)` |
|
| 41. |
One litre of gas A at 2 atm pressure at 27^(@)C and two litres of gas B at 3 atm pressue at 127^(@)C are mixed in a 4 litre vessel. The temperature of the mixture is maintained at 327^(@)C. What is the total pressure of the gaseous mixture? |
|
Answer» 3.93 atm `(2XX1)/300+(3xx2)/400=(P_("mix")xx4)/600implies(8+18)/1200=(4P_("mix"))/600` `P_("mix")=(26xx600)/(4xx1200)=3.25` atm |
|
| 42. |
One litre of an aqueoous solution contains 15 g of the solute of molar mass 150 g mol^(-1). If the density of solution is 1.005 g/mL, calculate the mole fraction of the solute. |
|
Answer» Mass of the solute in the solution = 15 g Mass of the solvent (water) in the solution=(1005-15)=990 g `"No. of moles of the solute "(n_(B))=((15G))/((150"g MOL"^(-1)))=0.1mol` `"No. of moles of the solvent"(eta_(A))=((990g))/((18"g mol"^(-1)))=55mol` `"Mole fraction of the solute"(x_(B))=n_(B)/(n_(B)+n_(A))=((0.1 mol))/((55.1 mol))=0.00181.` |
|
| 43. |
One litre of an unknown gas weighs 1.25 g at NTP. The possible formula of the gas is : |
|
Answer» `N_2` 22.4 L of gas st N.T.P. WEIGHT ` = 1.25 xx 22.4 = 28` ` therefore ` MOLECULES mass= 28 molecular FORMULA = CO |
|
| 44. |
Onelitre of a solution contains 18.9 g of HNO_(3) and one litre of another solution contains 3.2 g of NaOH. In what volume ratio must these solution be mixed to obtain a neutral solution ? |
|
Answer» `3:8` |
|
| 45. |
One litre of a mixture of O_(2) and O_(3) at NTP was allowed to react with an excess of acidified solution of KI. The iodine liberated required 40 mL of M/10 sodium thiosulphate solution for titration. The weight percent of ozone in the mixture is |
|
Answer» 0.0557 `{:(2 KI+H_(2)SO_(4)+(O)rarr K_(2)SO_(4)+H_(2)O+I_(2)),(2 Na_(2)SO_(3)+I_(2) rarr Na_(2)S_(4)O_(6)+2" NAI"),(bar(2 Na_(2)S_(2)O_(3)+2 KI+O_(3)+H_(2)SO_(4) rarr O_(2)+K_(2)SO_(4)+H_(2)O+Na_(2)S_(4)O_(6)+2" NaI")):}` 2 moles of `Na_(2)S_(2)O_(3)` react with one MOLE of `O_(3)` No. of moles `Na_(2)S_(2)O_(3)` in 40 ml of `M/10 Na_(2)S_(2)O_(3)` `=1/10xx1/1000xx40=4xx10^(-3)` mole `:. O_(3)` reacted (present) `=2xx10^(-3)` mole VOLUME of `O_(3)=2xx10^(-3)xx22.4 L=0.0448 L` `:.` Volume of `O_(2)=1-0.0448=0.9552 L` Weight of `O_(3)=2xx10^(-3)xx48=0.096 g` Weight of `O_(2) =(0.9552)/22.4xx32=1.3646 g` `:.` Weight of mixture `=0.096+1.3646` `=1.4606 g` `:.` % of `O_(3)` by weight `=0.096/1.4606xx100=6.573%`. |
|
| 46. |
One litre of a sample of hard water contains 4.44 mg CaCl_2 and 1.9 mg of MgCl_2.What is the total hardness in terms of ppm of CaCO_3 ? |
|
Answer» 1.9 mg `MgCl_2` is equivalent to 2 mg `CaCO_3` TOTAL wt. of `CaCO_3=6` mg HARDNESS in PPM =`(6xx10^(-3))/10^3 xx10^6=6` ppm |
|
| 47. |
One litre of a mixture of O_(2) and O_(3) at NTP was allowed to react with an excess of acidified solution of KI. The iodine liberated required 40 mL of M/10 sodium thiosulphate solution for titration. Knowing that ultraviolet radiation of wavelength 300 nm can decompose ozone and assuming that onw photon can decompose one molecule of ozone, the number of photons required for the complete decomposition of ozone in the original mixture will be |
|
Answer» `1.20xx10^(21)` `=2xx10^(-3)xx6.022xx10^(23)=1.2044x10^(21)` `:.` No. of photons REQUIRED `=1.2044xx10^(21)`. |
|
| 48. |
One litre of a mixture of O_(2) and O_(3) at NTP was allowed to react with an excess of acidified solution of KI. The iodine liberated required 40 mL of M/10 sodium thiosulphate solution for titration. The volume of oxygen left unreacted in the mixture is |
|
Answer» 0.655 L |
|
| 49. |
One litre of a mixture of carbon mono oxide and carbon dioxide is passed through a tube containing red hot charcoal. The volume becomes 1.6 litres. Find the compositionof the gaseous mixture. |
|
Answer» |
|
| 50. |
One litre of a gas is at a pressure of 10^(-6) mm of Hg at 25^(@)C. How many molecules are present in the vessel? |
|
Answer» `3.2xx10^(6)` or `V_(2)=1.2xx10^(-6)"cc (at STP)"` No. of MOLECULES `=(6.02xx10^(23))/(22400)xx1.2xx10^(-6)` `=3.2xx10^(13)` |
|