Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
One mole of an organic compound requires 0.5 mole of oxygen to produce an acid. The compound may be |
|
Answer» Aldehyde |
|
| 2. |
One mole of an organic compound A with the formula C_(3)H_(8)O reacts completely with two moles of HI ot form X and Y. When Y is boiled with aqueous alkali it forms Z. Z answers the iodoforms test. The compound A is |
|
Answer» Propan-2-ol `CH_(3)-O-C_(2)H_(5)underset("(excess)")overset(HI)to underset((X))(CH_(3)I)+underset((Y))(C_(2)H_(5)I)+H_(2)O` `underset(C_(2)H_(5)Ioverset(aq. KOH)to underset((Z))(C_(2)H_(5)OH)` `C_(2)H_(5)OH` gives IODOFORM test. |
|
| 3. |
One mole of an organic compound is found to require only 0.5 mol of oxygen to produce an acid. Which class of compounds does the starting material belong to ? |
|
Answer» Alcohol |
|
| 4. |
One mole of an organic compound A with the formula C_(3)H_(8)O reacts completely with two moles of HI to from X and Y. When Y is boiled with aqueous alkali it forms Z.Z answers the iodoform test. The compound A is |
|
Answer» A) propan-2-ol `CH_(3)-underset((A))(O)-C_(2)H_(5)underset("(excess)")overset(HI)(to)underset((X))CH_(3)I+C_(2)H_(5)I+H_(2)O` `underset((Y))(C_(2)H_(5)I)overset("aq. KOH")(to)underset((Z))(C_(2)H_(5)OH)` `C_(2)H_(5)OH` gives IODOFORM test . |
|
| 5. |
One mole of an organic compound (A) having M.F. C_(2)H_(6)O reacts with MeMgI to liberate one mole of methane. (A) reacts with CH_(3)COCl to yield a sweet smelling liquid (B). Identify (A) and (B). |
|
Answer» Solution :(i) Since compound (A) with M.F. `C_(2)H_(6)O` reacts with MeMgI to give one mole of `CH_(4)`, the compound (A) must be ETHYL alcohol. `MeMgI-=CH_(3)MgI` `underset("Ethyl alcohol")(CH_(3)CH_(2)OH)+CH_(3)MgI to CH_(3)+Mg(OCH_(2)CH_(3))I` (II) Compound A, i.e., ethyl alcohol reacts with acetyl chloride `(CH_(3)COCl)` to form sweet smelling compound (B). therefore, compound (B) must be ethyl acetate. `underset("Acetyl chloride")(CH_(3)COCl)+underset("Ethyl alcohol")(HOCH_(2)CH_(3)) to underset("Ethyl acetate (B)")(CH_(3)CO-OCH_(2)CH_(3)+HCl)` |
|
| 6. |
One mole of an organic amide (A) upon alkaline hydrolysis gives one mole of NH_3and one mole of monobasic acid of equivalent weight 74. What is the molecular formula of (A)? |
| Answer» SOLUTION :`C_2H_5CONH_2` | |
| 7. |
One mole of an non linear triatomic ideal gas is expanded adiabatically at 300 K from 16 atm to 1 atm. Find the values of DeltaS_("sys"),DeltaS_("surr")&DeltaS_("tota") under the following conditions. (i) Expansion is carried out reversibly. (ii) Expansion is carried out irreversibly (iii) Expansion is free. |
|
Answer» Solution :For non-linear tri-atomic IDEAL gas `C_(v)=3R,C_(p)=4R` (i) `DeltaS_("sys")=nC_(v)"ln"T_(2)/T_(1)+NR"ln"v_(2)/v_(1)=0` q = 0 `DeltaS_("surr")=-DeltaS_("sys")=0` `DeltaS_("total")=0` (ii) First of all we will have to calculate the temperature of the gas after it has undergoes the said adiabatic reversible expansion we have q = 0 `DeltaU=q+w` `nC_(v)(T_(2)-T_(1))=-P_("ext")(v_(2)-v_(1))` `3R(T_(2)-300)=-1[(RT_(2))/p_(2)-(RT_(1))/2]=-R[T_(2)/1-300/16]` `T_(2)=229.68K` `DeltaS_("sys")=nC_(p)"ln"T_(2)/T_(1)+nR"ln"p_(1)/p_(2)` `=4R"ln"229.68/300+R"ln"16/1=-1.068R+2.77` `R=1.702R` `DeltaS_("surr")=(-q_("irr"))/T=0` `DeltaS_("total")=DeltaS_("sys")=1.702R` (iii) In free adiabatic expansion we have w = 0 `q=0" "DeltaT=0` `DeltaS_("sys")=nR"ln"p_(1)/p_(2)=Rln16=2.77R` `DeltaS_("surr")=(-q_("irr"))/T=0` `DeltaS_("total")=DeltaS_("sys")=2.77R` |
|
| 8. |
One mole of an non linear triatomic ideal gas is compressed adiabatically at 300 K from 1 atom to 16 atm. Calculate Work done under the following conditions. (i) Expansion is carried out reversibly. (ii) Expansion is carried out irreversibly. |
|
Answer» SOLUTION :q = 0 Adiabatic PROCESS (i) `W_("rev")=DeltaU=C_(V)(T_(2)-T_(1))` `P^(1-gamma)T^(gamma)=K` `T_(2)=300(1/16)^((1-4//3)/(4//3))=600K` w = 3R (600 - 300) = 900 R (ii) `W_("irr")=-P_("EXT")(V_(2)-V_(1))=-P_("ext")((nRT_(2))/P_(2)-(nRT_(1))/P_(1))` `-P_("ext")((nRT_(2))/P_(2)-(nRT_(1))/P_(1))=C_(v)(T_(2)-T_(1))` `-16((RT_(2))/16-(RT_(1))/1)=3R(T_(2)-T_(1))` `-(T_(2)-16T_(1))=3R(T_(2)-T_(1))` `-T_(2)+16T_(1)=3T_(2)-3T_(1)` `4T_(2)=19T_(1)` `T_(2)=19/4xx300=1425K` `w=DeltaU=3R(1425-300)=3375R` |
|
| 9. |
One mole of an non linear triatomic ideal gas is expanded adiabatically at 300 K from 16 atm to 1 atm. Calculate Work done under the following conditions. (i) Expansion is carried out reversibly. (ii) Expansion is carried out irreversibly |
|
Answer» Solution :q = 0 `W=DeltaU=C_(v)(T_(2)-T_(1))` `C_(v)` for triatomic NON linear GAS = 3R (i) For rev. process. `P_(1)^(1-GAMMA)T_(1)^(gamma)=P_(2)^(1-gamma)T_(2)^(gamma)""r=4/3` `T_(2)=T_(1)(P_(1)/P_(2))^((1-gamma)/v)=300(16)(1-4/3)/(4//3)=300(2^(4))^(-1//4)=150K` `DeltaU=w-3R(150-300)=-450R` (II) n = 1 `-P_("ext")(V_(2)-V_(1))=C_(V)(T_(2)-T_(1))` `-P_("ext")((RT_(2))/P_(2)-(RT_(1))/P_(1))=3R(T_(2)-T_(1))` `-1((RT_(2))/1-(RT_(1))/16)=3R(T_(2)-T_(1))` `-R/16(16T_(2)-T_(1))=3R(T_(2)-T_(1))` `-16T_(2)+T_(1)=48T_(2)-48T_(1)` `49T_(1)=64T_(2)` `T_(2)=49/64xx300` `T_(2)=229.69` `W_("irr")+C_(v)(T_(2)-T_(1))+3R(229.69-300)=-210.93R` |
|
| 10. |
One moleof an non linear triatomic ideal gasis expanded abiabatically at 300 K form16 atmto1 atmltbr gtFind the value DeltaS_(sys), DeltaS_(surr) & DeltaS_("total")under the following conditions. (i) Expansion is carriedout reversibly (ii) Expansionis carriedout irreversibly (iii) Expansion is free. |
|
Answer» |
|
| 11. |
One mole of annon lineartriatomicideal gas is compressed adiabaticallyat 300 K from 1 atm to 16 atm. Calculate Work doneunder the following conditions.(i) Expansion is carriedout reversibly (ii) Expansion is carried outirreversibly. |
|
Answer» |
|
| 12. |
One moleof an idealmonoatomic gas is put throughtreverslible pathas show in figure.Fill in theblankin thetablegivenbelow: |
|
Answer» `"State-2": """P= 1 atm""""V= 44.8L""""T= 546K"` `"State-3": """P= 0.5 atm""""V= 44.8L""""T= 273K"` Step -A : Isobaricprocess `W= - PDeltaV = - nRDeltaT=- R(273)` `DeltaH=nC_(p,m)DELTAT = (5)/(2)Rxx 273` `DeltaE=nCv_(m)DeltaT = (3)/(2)Rxx 273` `q_(p) = DeltaH =(5)/(2)R xx 273` Step -B : ISOCHORIC process `W=0` `DeltaH=nC_(p,m)DeltaT = -(5)/(2)Rxx 273` `DeltaE=nC_(v,m)DeltaT = -(3)/(2)Rxx 273` `q_(p) = DeltaE =-(3)/(2)R xx 273` Step-C : Isothermal process `W= - nRT ln.((V_(2))/(V_(1)))= 273ln2` `DeltaH=0` `DeltaE = 0` `q= - W "" =- 273 R ln 2` |
|
| 13. |
One mole of an ideal monoatomic gas mixed with 1 mole of an ideal diatomic gas. The molar specific heat of thr mixture at constant vlume is : |
|
Answer» 3 cal |
|
| 14. |
One mole of an ideal monoatomic gas is caused to go through the cycle shown in figure.Then the change in the internal energy of gas from a tob and b to c is respectively : |
|
Answer» `(9P_@V_@)/2,6 RT_@` |
|
| 15. |
One mole of an ideal monoatomic gas is heated according to path AB and AC. If temperature of state B and C are equal then calculate (q_(AC))/(q_(AB)) xx 10. |
|
Answer» |
|
| 16. |
Onemoleof an ideal monoatomic gas iscarried through the reversilycycle of thegiven figure consisiting of stepA,and C andinvolvingstate1,2 and 3. Fill in the blankspacein thethe tablegivenbelow assumingreversible steps. |
|
Answer» `2 to 3 rArr "" " Isothermala preocess"` `3 to 1 rArr "" " Isobaric preocess"` `"State - 1:"""P=(NRT)/(V)= 1 atm""V= 22.4 L ""T= 273K` `"State - 2:"""T= 546K""V= 22.4 L ""P= 2atm` `"State - 3:"""T= 546K""V= 44.8 L ""P= 1atm` Step - A:Isochoric process `""W = 0` `""DeltaE= n C_(v.m)DELTAT= (3)/(2) R xx 273` `""DeltaH= n C_(p.m)DeltaT= (5)/(2) R xx 273` `""q_(v)= DeltaE= (5)/(2) R xx 273` Step- B : Isothermal processltb`""DeltaE=0` `""DeltaH=0` `""W= - nRT ln .((V_(1))/(V_(2))) = - 1 xx R xx546 ln 2` `""= - 546R ln 2` `""q = - W =546R ln 2` Step-C : Isobaricprocess `""DeltaE = nC_(v.m)DeltaT= -(3)/(2) R xx 273` `""DeltaH = nC_(p.m)DeltaT= -(5)/(2) R xx 273` `""W =- PDeltaV= - nRT = R xx 273` `""q_(p) = DeltaH = -(5)/(2) R xx 273` |
|
| 17. |
One mole ofan ideal monoatomic gasexpanded irreversibly in two stage expansion . "State"-1""("8.0 bar, 4.0 liters, 300K") "State"-2""("2.0 bar, 16 liters, 300K") "State"-3""("1.0 bar, 32 liters, 300K") Total heatabsored by thegas in theprocess is : |
|
Answer» <P>116 J `W_(1)= - 2 [16-4] xx 100 ""= - 2400J` `W_(2) = - [32 -16] = - 16 xx 100= - 1600 J` ""`-4000` `q = - W = 4000 J` |
|
| 18. |
Onemole ofan idealmonoatomic gas (C_(V.M)= 1.5 R)is subjected to the followingsequence of steps : (a)The gasis heated reversiblyat constantpressure of 1 atm from 298 K to 373 K. (b) Next, thegas isheatedreversiblyand isothermallyto doubleits volume. (c)Finally , thegasis cooled reversiblyandadiabatically to 308 K . Calcuated q, w, DeltaH for the overall porcess. |
|
Answer» `= - nRDeltaT` `=- 1xx 8.14 XX (373 - 298)` ` = -623.55 J//mol` `DeltaH = q = nC_(P)DeltaT` `= - 2.5 xx 8.314 xx 75"" (C_(P)=2.5 R)` `= 1558.8 J//mol` `DeltaU = 1.5 xx 8.314 xx 75""(C_(V) = 1.5 R)` ` =935.5 J//mol` (b)` W=-2.303RT log ((V_(1))/(V_(2)))` ` = - 2.303 xx 8.314 xx 373 log 2` ` = - 2149. 7 J//mol` `DeltaU = 0 ,DeltaH = 0 ""q =2149.7 J//mol^(-1)` (C)`W = Deltau = nC_(V)DeltaT""("For adiabaticprocess "q = 0, DeltaU ne W)` `= 1 xx 1.5 xx 8.314 (308-373)` `= - 810.62 J//mol` `q= 0` `DeltaH = nC_(P)DeltaT=- 1 xx 2.5 xx 8.314 (308-373)` `= -1351 .03 J//mol` For overall process `q_(net) = 3708.59 J//mol` `W_("net") = - 3583. 88 J//mol""DeltaU_("net")= 124.71 J//mol` `DeltaH_("net")= 207.85 J//mol` |
|
| 19. |
One mole of an ideal gas undergoes reversible isothermal expansion from an initial volume V_(1) to a final volume 10V_(1), and does 10 kJ of work. The initial pressure was 1xx10^(7) Pa. (a) Calculate V_(1) . (b) If there were two moles of the gas, what had been its temperature ? |
|
Answer» Solution :For reversible ISOTHERMAL expansion of an ideal gas, (a) `W=-2.303nRTlog.(V_(2))/(V_(1))=-10,000("GIVEN")` or `2.303xx1xx8.314xxTlog.(10V_(1))/(V_(1))=10,000` `T=522.27K` We have `pV=nRT` `10^(7)xxV_(1)=1xx8.314xx522.27` `V_(1)=4.342xx10^(-4)m^(3)` (b) Assuming pressure to remain constant, for 2 moles of the ideal gas. `T=(522.27)/(2)=261.13 K`. |
|
| 20. |
One mole of an ideal gas undergoes a process (10L, 4atm) to (4L, 10 atm ). Theprocess may be represented by straight line in P-V diagram The correct statementfor the process : |
|
Answer» The temperature of gas is CONSTANT throughout the PROCESS |
|
| 21. |
One mole of an ideal gas undergoes change of state from (4 L, 3 atm) to (6 L, 5 atm). If the change in internal energy is 45 L-atm then the change of enthalpy for the process is |
|
Answer» 27 L-atm |
|
| 22. |
One mole of an ideal gas is taken from a to b along two paths denoted by the solid and the dashed lines as shown In the graph below. If the work done along the solid line path is W_(s) and that along the the dotted line path is w_(d), then the integer closest to the ratio w_(d)//w_(s) is |
|
Answer» `W_(d)=(4.0)xx1.5)+(1.0xx1.0)+(0.75xx2.5)L"atm"=6+1+1.815=8.875 L "atm"` Solid line indicates isothermal reversible expansion. It is isothermal because `PV` is constant `:. W_(s)=2.303nRT log V_(2)//V_(1)` `=2.303PVlog V_(2)//V_(1) (PV=nRT)` `=2.303xx2log"(5.5)/(0.5)(PV=2 L "atm")` `=4.61 L"atm"` Hence `=(W_(d))/(W_(s))=(8.875)/(4.61)=2` |
|
| 23. |
One mole of an ideal gas is expanded isothermally at 300 K from 10 atm to 1 atm. Find the values of DeltaS_("sys"),DeltaS_("surr")&DeltaS_("total") under the following conditions. (i) Expansion is carried out reversibly. (ii) Expansion is carried out irreversibly (iii) Expansion is free. |
|
Answer» Solution :(i) `DeltaS_("sys")=nC_(p)"ln"T_(2)/T_(1)+nR"ln"P_(1)/P_(2)=0+Rln10=Rln10` `DeltaS_("surr")=-DeltaS_("sys")=-Rln10` `DeltaS_("total")=0` (ii) `DeltaS_("sys")=Rln10` `DeltaS_("surr")=(-q_("IRR"))/T` `DeltaU=0=Q+w` `q_("irr")=p_("EXT")(v_(2)-v_(1))` `q_("irr")=p_("ext")[(RT)/p_(2)-(RT)/p_(1)]=RT[1/1-1/10]` = `RTxx9/10=9/10xxRxx300=270R` `DeltaS_("surr")=(-q_("irr"))/T=(-9R)/10` `DeltaS_("total")=DeltaS_("sys")+DeltaS_("sur")=Rln10-(9R)/10` (iii) Free expansion `DeltaT = 0` w = 0 q = 0 `DeltaS_("sys")=Rln10` `DeltaS_("surr")=(-q_("irr"))/T=0` `DeltaS_("total")=Rln10` |
|
| 24. |
One mole of an ideal gas is taken from a to b along two parths dented by the solid and the dashed lines as shown in the graph below. If the work done along the solid line pathis W_(s)and that along the dotted line path is W_(d) then the interger closet to the ratio W_(d) // W_(s) is |
|
Answer» |
|
| 25. |
One mole of an ideal gas is expanded isothermally from 1 dm^(3) to 10 dm^(3) at 300 K. DeltaG will be equal to |
|
Answer» 5.744 kJ |
|
| 26. |
One mole of an idealgas iscompressedfrom 500cm^(3) againsta constantpressureof 1.216 xx 10^(5)Pa. Theworkinvolved in theprocessis 35.50 J. calculatethe finalvolume . |
|
Answer» Constantexternalpressure `= P_(EX)` `= 1.216 xx10^(5)Pa = 1.216 xx 10^(5)NM^(-2)` Forcompressionworkobtained= W=+36 .5 J Initialvolume= `V_(1)= 500cm^(3)= 500xx 10^( -6)m^(3)( :'1 m^(3)=10^(6) cm^(3))` Finalvolume `=V_(2)= ?` Sincethe COMPRESSION thetakesplaceagainsta constantpressureit isan irreversibleprocess andworkobtained is `W= -P_(ex) (V_(2)-V_(1))` `36.5 =-1.216xx 10^(5)xx (V_(2) - 500xx 10^(-6))` `:.(36.5)/(1.216 xx 10^(5))= - (V_(2) -50 xx 10^(5))` `30 xx 10^(-5)=-(V_(2) -50 xx 10^(-5))` `:. 3010^(5) = - V_(2) + 50 xx 10^(-5)` `:. V_(2)= (50 -300) xx 10^(-5)=20 xx 10^(-5)m^(3)` `= 20 xx 10^(-5) xx 10^(6) cm^(3) = 200 cm^(3)` |
|
| 27. |
One mole of an ideal gas is expanded isothermally at 300 K from 10 atm to 1 atm. Calculate q, w, DeltaU&DeltaH under the following conditions. (i) Expansion is carried out reversibly. (ii) Expansion is carried out irreversibly |
|
Answer» SOLUTION :Isothermal process (i) For ideal gas `DeltaU=0" "DeltaH=0` Q = -W `w_("rev")=-nRT"ln"P_(1)/P_(2)=-1xxRxx300"ln"10/1=-690.9R` (ii) `W_("irrev")=-P_("ext")(V_(2)-V_(1))=-P_("ext")((nRT)/P_(2)-(nRT)/P_(1))` = `-1xx1xxRxx300[1/1-1/10]=-270R` |
|
| 28. |
One mole of an ideal gas is allowed to expand freely and adiabatically into vacuum until its volume has doubled. Astatement which is not true concerning this expression is |
| Answer» Solution :W=0 is not true. | |
| 29. |
One mole of an ideal gas going from state-A to state-B through different processes. Column-I shows graph of the process and column-II shows change in parameter in these process. Match the following: |
|
Answer» |
|
| 30. |
One mole of an ideal gas is allowed to expand reversibly and adibatically from a temperature of 27^(@)C. If the work done during the process is 3 kJ, then final temperature of the gas is (C_(v)=20 J//K) |
|
Answer» 100 K Initial temperature = `27^(@)C=300K` Work DONE by the SYSTEM = 3kJ = 3000 J It will be (-) because work is done by the system HEAT capacity at constant volume (CV) = 20 J/K We know that work done `W=-nC_(V)(T_(2)-T_(1)), 3000 =-1xx20(T_(2)-300)` `3000=-20T_(2)+6000` `20T_(2)=3000, T_(2)=(3000)/(20)=150 K` |
|
| 31. |
One mole of an ideal gas (C_(v) = 3/2R) is heated at constant pressure of 1 atmosphere from 25^(@) C to 100^(@) C. Calculate DeltaE and DeltaH. |
|
Answer» Solution :As `(C_(P) - C_(v)) = R rArr C_(p)=C_(v) + R` `rArr C_(p) = 3/2 R + R= 5/2 R` Heat given at CONSTANT PRESSURE. `(DeltaH) = nC_(p)DeltaT = 1 x 5/2 R xx (373 - 298)` `rArr (DeltaH) = 1 xx 5/2 xx 1.987 xx 75 = 372.56` cal Work done in the process = `-PDeltaV` `=-P(V_(2)-V_(1))` `=-P((nRT_(2))/P -(nRT_(1))/P)`(As PV = nRT) `=-NR(T_(2)-T_(1))` `=-1 xx 1.987 xx (373 - 298)` `=-149.05` cal. From FIRST law of thermodynamics. `DeltaE = q+W` `=372.56 - 149.05` =223.51 cal. |
|
| 32. |
One mole of an ideal gas expands reversibly and isothermally at 300 K from 5dm^(3)" to "50dm^(3). The work done by the gas for the process is equal to |
|
Answer» `-1.382` KCAL |
|
| 33. |
One mole of an ideal gas at 300 K is expanded isothermally from an initial volume of 1 litre to 10 litres. The DeltaE for this process is (R = 2 cal mol^(-1)K^(-1))- |
|
Answer» 163.7 cal |
|
| 34. |
One mole of an ideal gas at 300 K is expanded isothermally from an initial volume of 1 L to 10 L. The DeltaE for this process is (R=2 cal . "mol"^(-1)K^(-1)) |
|
Answer» 163.7cal |
|
| 35. |
One mole of an ideal gas at 300 K in thermal contact with surroundings expands isothermally from 1.0 L to 2.0 L against a constant pressure of 3.0 atm. In this process, the change in entropy of surrounding (DeltaS_("surr")) in JK^(-1) is (1 L atm = 101.3 J) |
|
Answer» 5.763 `dq=-dW=P_(ext)(V_(2)-V_(1))=3L-atm=3xx101.3" JOULE"` `DeltaS_("SURROUNDING")=-(3xx101.3)/(300)" Joule "K^(-1)=-1.013 " Joule "K^(-1)` `therefore DeltaS_("SURR")=-1.013" Joule "K^(-1)` |
|
| 36. |
One mole of an ideal gas at 300 K expands isothermally and reversibly from 5 to 20 litres. Calculate the work done and heat absorbed by the gas. |
|
Answer» |
|
| 37. |
One mole of an ideal gas at 300 K in thermal contact with surroundings expands isothermally from 1.0 L to 2.0 L against a constant pressure of 3.0 atm. In this process, the change in entropy of surroundings (DeltaS_("max"))" in "JK^(-1) is |
|
Answer» 5.763 |
|
| 38. |
One mole of an ideal gas at 22.4 litres is expanded isothermally and reversibly at 300 K to a volume of 224 litres at a constant pressure. Calculate W,q,DeltaH, DeltaG and Delta S. |
|
Answer» |
|
| 39. |
One mole of an ideal at 300 K is expanded isothermaly from an initial volume of 1 liter to 10 liters. The triangleE for this process is [ R= 2 cal k^-1 mol^-1] |
|
Answer» 163.7 cal |
|
| 40. |
One mole of an element contains 4.2xx10^(24) electrons. What is the atomic number of the element? |
|
Answer» |
|
| 41. |
one mole of an anydrose salt AB dissolves in water with the evolution of 21.0mol^(-1)of heat. If the heat of hydration of AB is -29.4 J mol^(-1) , then the heat of dissociation of hydrated salt AB is |
|
Answer» ` 50.4 Jmol ^(-1)` `(ii) AB(s)+xH_(2)O to AB.xH_(2)O(s),DeltaH=-29.4 J mol ^(-1)` REQUIRED equation is `AB.xh_(2)O(s)+ (aq) to AB(aq), DeltaH=?` EQ.(i) is equivalent to ` AB(s)+ xH_(2)O to AB.x H_(2)O(s), DeltaH=DeltaH_(1)` `AB.xH_(2)O(s) + (aq)to AB(aq), DeltaH=DeltaH_(2)` DeltaH_(1) + DeltaH_(2)=-21` ` -29.4 + DeltaH_(2)=-21 or DeltaH_(2) =8.4 mol^(-1)` |
|
| 42. |
One mole of an amine (A) consumes two moles of methyl bromide to give a quaternary ammonium salt. The amine (A) is |
| Answer» Answer :C | |
| 43. |
One mole of ammonia was completely absorbed in one litre solution each of (a) 1 M HCI, (b) 1 MCH_3COOH and (c) 1 M H_2SO_4 at 298 K. The decreasing order for the pH of the resulting solution is (Given K_(b) (NH_3) = 4.74) |
|
Answer» `b GT c gt a ` 1M of `NH_3` completely reacts with 1M HCl. The SALT formed is ACIDIC b) `CH_3COOH + NH_3 to CH_3COO NH_4`. The salt formed is neutral c) `2NH_3 + H_2SO_4 to (NH_4)_2 SO_4 `. 1 M of `NH_3` need 0.5 M of `H_2SO_4 `, 0.5 M still REMAIN in solution. The salt is acidic. |
|
| 44. |
One mole of alkene on ozonolysis gives 2 moles of butanone. The alkene is : |
|
Answer» 3, 4-dimethylhex - 2- ENE |
|
| 45. |
One mole of acetal on complete hydrolysis gives |
|
Answer» MOLE of aldyhyde,1 mole of alcohol |
|
| 46. |
One mole of a symmetrical alkene on ozonolysis gives two moles of an aldehyde having a molecular mass of 44u. The alkene is: |
|
Answer» Ethene 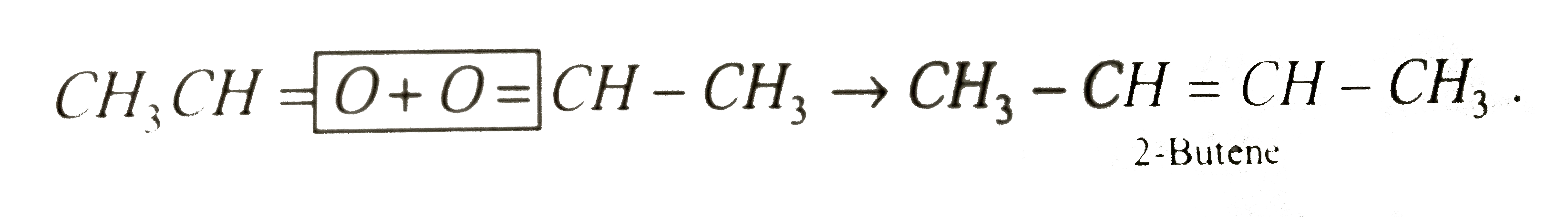
|
|
| 47. |
One mole of a prefectputthroughta cycleconsitingofthefollowingthreereversiblesteps :(CA) Isothermal comoressionfrom 2atm and10litres to20 atmandlitre.(AB ) Isobaric expansion to returnthe gasto the origanlvolume of 10litrewithT going fromT_(1) to T_(2)(BC)Coolingat constantvolume to bringthegastotheoriganlpressurelt brgtandtemperature .The stepsare shownschematicallyin the figureshown. CalculateT_(1) to T_(2).(b) CalculateDeltaU, qandwill be in calories , foreachstep andfor thecycle . |
|
Answer» `T_(2) = (20 xx 10 )/(0.021) = 2436.0 K` (b)CA(Isothermal ) `W_(1) = - 2.303 xx 2 xx 243.6 log .(2)/(20)= 1122` BC(Isochoric ) `W_(2) = 0` AB(Isochoric) `W_(3) = - RDeltaT = - 4384.9 cal` For complete CYCLE `W = W_(1) + W_(2) + W_(3) = 3262. 888 cal` |
|
| 48. |
One mole of a symmetrical alkene on hydrolyses gives two moles of an aldehyde having a molecular mas of 44 u. The alkene is |
|
Answer» 2-butene `underset("2-butene")(CH_(3)CH=CHCH_(3))OVERSET((i)O_(3))underset((ii)Zn//H_(2)O)rarrundersetunderset("Mol. mass=44")("Acetaldehyde")(2CH_(3)CHO)` |
|
| 49. |
One mole of a perfect gas expands isothermally to ten times of its original volume. The change in entropy is |
|
Answer» 0.1 R `therefore DeltaS=2.303R" log"(10)/(1)` `DeltaS=2.303R""(because log10=1)` |
|
| 50. |
One "mole" of a nonideal gas undergoes a change of state (2.0 atm, 3.0 L, 95 K) to (4 atm, 5L, 245 K) with a change in internal energy, Deltau=30.0L atm. The change in enthalpy, DeltaH, of the process in L. atm is |
|
Answer» <P>`40.0` |
|

