Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Noble gases are prepared by the: |
|
Answer» CONDENSATION of gases of the air |
|
| 2. |
Noble gases are only sparingly soluble in water due to : |
|
Answer» DIPOLE - dipole INTERACTIONS |
|
| 3. |
Noble gases are mostly inert. Assign reasons. Or Explain the causes of inertness of noble gases. |
|
Answer» Solution :Noble GASES are CHEMICALLY inert because of the following reasons : (i) They have completely filled `ns^(2)np^(6)` ELECTRONIC configuration in their respective valence shells. (ii) They have HIGH IONIZATION enthalpies. (iii) Electon gai enthalpies of noble gases are positive. |
|
| 4. |
Noble gases are mostly chemically inert. Give reason. |
| Answer» Solution : Noble GASES are CHEMICALLY inert because they have complete octets and possess STABLE structure. Their IONISATION enthalpies are LARGE. | |
| 5. |
Noble gases are monoatomic in nature. Explain. |
| Answer» Solution :All the orbitals (which are occupied by electrons) are completely filled in noble gases. THEREFORE, they have no tendency to share electrons with other atoms to FORM diatomic molecules and hence are MONOATOMIC in NATURE. | |
| 6. |
Noble gases are chemically inert. This is due to ……………….. |
|
Answer» UNSTABLE ELECTRONIC configuration |
|
| 7. |
Noble gases are chemically inert. Give reasons. |
| Answer» Solution :Noble gases are chemically inert because they have their octet complete EXCEPT HELIUM, i.e. they have a STABLE ELECTRONIC configurations. | |
| 8. |
Noble gases are chemically inert. Give one reason |
| Answer» SOLUTION :OCTET STRUCTURE or They have `ns^2np^5` CONFIGURATION | |
| 9. |
Noble gases are adsorbed by............ |
|
Answer» anhydrous `CaCl_2` |
|
| 10. |
Noble gases are absorbed on |
|
Answer» ANHYDROUS `CaCl_(2)` |
|
| 11. |
Noble gases and metals both have closed packed structures, yet the melting point of noble gases are exceptionally low. Why? |
| Answer» Solution :Although both have the closed packed structures, the atoms in noble GASES are held together by weak VAN DER WAALS. forces and thus show lower MELTING points | |
| 12. |
Noble gases are: |
|
Answer» Colourless |
|
| 13. |
Noble gases A, B, C, D, E are passed through Dewar's flask at-100^@C A, C & E gases are adsorbed. Unadsorbed gases B, D are passed through another Dewar's flask at-180^@C. Gas B is adsorbed. The Dewar's flask al-100^@C is put in contact with another Dewars flask al-193^@C. The gas Eis diffused in it. Finally A & C are seperated by warming the flask from-100^@C w -90^@C. Ilere 'C' comes out He and Ne gases respectively are |
|
Answer» Ne  `He, Ne underset("Deewars flask "-180^@C)overset("another")to`Ne is absorbed. He is unadsorbed, He has WEAK vander wall forces. Charcoal - 1 contains Ar, Kr, Xe is cooled to `-180^@C`. At this TEMP. Ar. Gets LIQUIFIED while Kr & Xe remain adsorbed.  (E) = Ar, (A)= Kr,(C )= Xe |
|
| 14. |
Noble gas used in the miner's cap lamp is |
|
Answer» krypton |
|
| 15. |
Noble gas that is not present in air |
|
Answer» He |
|
| 16. |
Noble gas atoms have very high ionization energles. Explain. |
| Answer» Solution :Because all the ELECTRONS in the outermost shell are paired and a large amount of energy is needed to unpair them. So, they POSSESS HIGH ionization ENERGIES | |
| 17. |
Noble gas forms compounds with |
|
Answer» Fluorine |
|
| 18. |
Noble gases do not react with other elements because |
|
Answer» The size of their AOMS are very small |
|
| 19. |
NO_(3)^(-)toNO_(2) (acid medium) E^(0)=0.790V NO_(3)^(-)toNH_(3)OH^(+) (acid medium) E^(0)=0.731V. At what pH, the above two will have same E value ? Assume the concentration of all other species NH_(3)OH^(+) except [H^(+)] to be unity. |
|
Answer» `overset(+5)(N)O_(3)^(-)+8H^(+)+6e^(-)toNH_(3)OH^(+)+2H_(2)O,0.731"volt"` `E=0.79-(0.0591)/(1)log((1)/([H^(+)]^(2)))=0.731-(0.591)/(6)log((1)/([H^(+)]^(8)))` `0.059=0.059(2-(8)/(6))pHimpliespH=(6)/(4)=1.5` |
|
| 21. |
NO_3^(-) is reduced to NH_(4)^(+)What is the change in oxidation number of nitrogen ....... |
|
Answer» |
|
| 23. |
NO_(3)^(Θ) isdetected by (a) _____when (b)_____is formed on the addition of FeSO_(4) and cone H_(2)SO_(4) |
|
Answer» |
|
| 25. |
NO_2 readily forms a dimer. Explain. |
Answer» Solution :`NO_2`is unstable due to the PRESENCE of one unpaired ELECTRON. Therefore, it dimerises to FORM a stable compound.
|
|
| 26. |
NO_(2) is not obtained when is heated |
|
Answer» `PB(NO_(3))_(2)` |
|
| 29. |
No reaction occurs in which of the following equations |
|
Answer» `I^(ɵ)+Fe^(2+) rarr` |
|
| 31. |
No. of unpaired e^(-) in K[(Ag(CN)_(2)] is |
|
Answer» |
|
| 32. |
No. of unpaired electrons in Fe^(2+) ions is : |
|
Answer» three |
|
| 33. |
No of products and number of fractions are respectively : |
|
Answer» 6,5 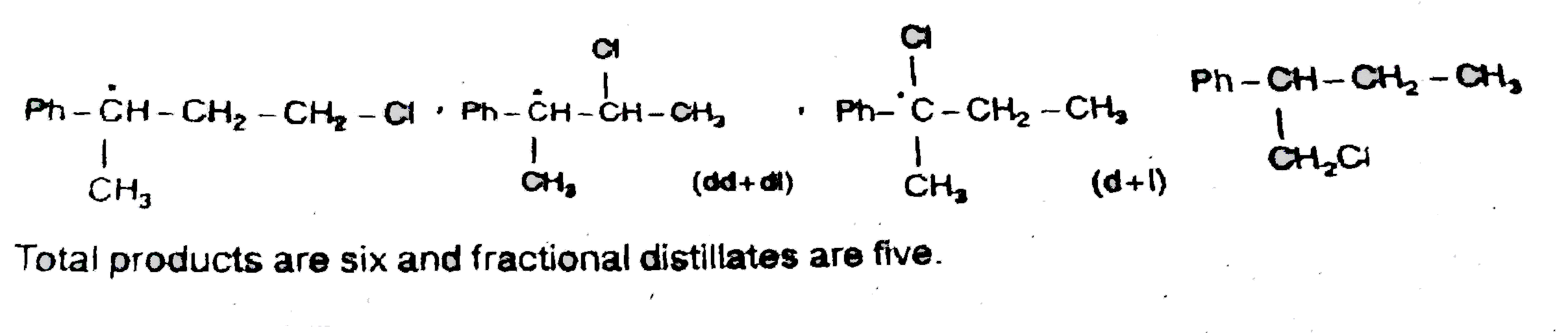
|
|
| 35. |
No . Of products and No . Of fractions are respectively |
|
Answer» 6,5 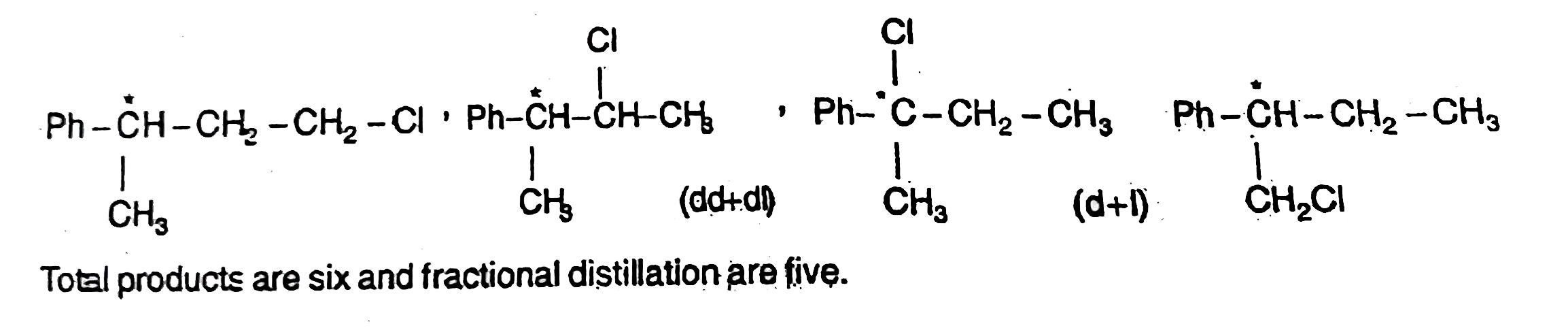
|
|
| 36. |
No. of moles of H_(2)O gasevolved when one mole of the following compound reacts with sodium. |
|
Answer» |
|
| 37. |
No. of nuclei among the following which undergo radioactivedecay (based on number of protons only) Silicon - 30 , Germanium - 72 , Barium - 130 , Radon - 216 |
|
Answer» |
|
| 39. |
No. of lone pairs in I bar(Cl_(2)) |
Answer»  , TOTAL ELECTRON pairs `= (7+2+1)/(2) = 5`, Lone pairs = Total -b.p.s = 5-2 =3 , TOTAL ELECTRON pairs `= (7+2+1)/(2) = 5`, Lone pairs = Total -b.p.s = 5-2 =3
|
|
| 40. |
No. of isomers of [Co(NH_(3))_(2)Cl_(2)(en)]^(+) are |
|
Answer» |
|
| 41. |
No. of ions produced by dissolving PtCl_(4).2NH_(3) in aqueous media is |
|
Answer» |
|
| 42. |
No. of hybrid orbitals present in IF_(7) |
|
Answer» |
|
| 43. |
No. of geometrical isomersof [RhCl(CO)(PPh_(3))(NH_(3)) are |
|
Answer» |
|
| 44. |
No. of Geometrical isomers for following compound is : |
|
Answer» 8 |
|
| 47. |
No of compounds producing gas on hydrolysis (with H_(2)O ) is Al_(4)C_(3),BaC_(2),Mg_(2)C_(3),SiC,B_(2)H_(6),Fe_(3)C |
|
Answer» |
|
| 48. |
No of allylic hydrogens in thermodynamic product is ''X'' No of allylic hydrogens atom in kinetic product is ''Y'' The value of |X+Y| is ……. |
|
Answer» |
|
| 49. |
No. of acyclic isomers of the compound having the molecular formula C_(4)H_(10)O is : |
|
Answer» 4 (i) `CH_(3)CH_(2)CH_(2)CH_(2)OH (ii) CH_(3)CH_(2)UNDERSET(OH)underset(|)(CH)CH_(3)` (iii) `CH_(3)underset(CH)underset(|)(CH_(2))OH "" (IV) C_(2)H_(5)-O-C_(2)H_(5)` (v) `CH_(3)OCH_(2)CH_(2)CH_(3)` |
|