Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Nitrates of group '1' (except LiNO_3) on heating give: |
|
Answer» `O_2` |
|
| 2. |
Nitrates of all the metals are |
|
Answer» Coloured |
|
| 3. |
Nitrates of all metals are: |
|
Answer» Unstable |
|
| 4. |
Nitrates of all metals are |
|
Answer» UNSTABLE |
|
| 5. |
Nitrate is confirmed by ring test. The brown colour of the ring test is due to the formation of |
|
Answer» Ferrous NITRITE `6FeSO_(4)+3H_(2)SO_(4)+2HNO_(3)` `to3Fe_(2)(SO_(4))_(3)+4H_(2)O +2NO` `FeSO_(4)+NO to underset("(BROWN RING)")underset("Nitrosoferrous sulphate")(FeSO_(4).NO)` |
|
| 6. |
Nitrate is confirmed by ring test. The brown colour of the ring is due to the formation of |
|
Answer» Ferrous nitrite |
|
| 7. |
Nitrate of all metals are |
|
Answer» unstable |
|
| 8. |
Ni_((S)) |Ni_((aq))^(2+)||Ag_((aq))^(+)|Ag_((S)) is a non-standard cell, in which concentration of ion is less than 1 M, then determine correct formula to calculate potential of non-standard cell. |
|
Answer» `E_(cell)=E_(cell)^(THETA)-(0.059)/(2)"log"([A_(G)^(+)])/([Ni^(2+)])` `Ni_((S)) + 2Ag_((aq))^(+) to Ni_((aq))^(2+)+2Ag_((S))"Where, "n=2` `K=(["Product"])/(["Reactant"])=([Ni_((aq))^(2+)][Ag_((S))]^(2))/([Ni_((S))][Ag_((aq))^(+)]^(2))=([Ni_((aq))^(2+)])/([Ag_((aq))^(+)]^(2))` So, `E_(cell)=E_(cell)^(theta)-(0.059)/(2)"log"([Ni_((aq))^(2+)])/([Ag_((aq))^(+)]^(2))`. |
|
| 9. |
Nitrate & Nitrite both give brown ring test, can be distinguish by - |
|
Answer» `HOSO_(2)NH_(2)` (Sulphonic acid) |
|
| 10. |
nipropyl cyanideon reductiongives |
|
Answer» n-butyl AMINE |
|
| 11. |
Niobium crystallizes in body centred cubic structure. If its density is 8.55 g cm^(-3), calculate the atomic radius of niobium. (Atomic mass of Nb = 93u , N_A = 6.02 xx 10^(23) mol^(-1)) |
|
Answer» Solution :DENSITY of niobium, d=8.55 G `cm^(-3)`, Number of atoms in bcc UNIT cell, Z= 2 Atomic mass of niobium, M = 93u Density, `d = (ZM)/(N_A xx a^3)` `a^3=(ZM)/(d xx N_A)=(2 xx 93)/(8.55 xx 6.02 xx 10^(23))` `= 3.61 xx 10^(-23) cm^3= 36.1 xx 10^(-24) cm^3` `a = 3.30 xx 10^(-8) cm=3.30 Å` For a bcc unit cell, `4r = a SQRT3` or `r = (a sqrt3)/(4)=(3.30 xx sqrt3)/(4) Å` = `(3.30 xx 1.73)/(4) = 1.427` Å= 14.27 nm |
|
| 12. |
Niobium crystallizes in body-centred cubic structure. If its density is 8.55 g cm^(-3), calculate atomic radius of niobium, given its atomic mass 93 u. |
|
Answer» SOLUTION :`a^(3) = (M xx Z)/(d xx N_(0) xx 10^(-30))` `a^(3) = (93 G mol^(-1) xx 2)/(8.55 g cm^(-3) xx 6.02 xx 10^(23) mol^(-1) xx 10^(-30))` `a^(3) = 3.61 xx 10^(7) = 36.1 xx 10^(6)` `a = (36.1)^(1//3) xx 10^(2)` pm `= 3.304 xx 10^(2)` pm = 330.4 pm `[x = (36.1)^(1//3), log x = 1//3 log 36.1` `= 1//3 xx 1.5575 = 0.519` x = antilog 0.519 = 3.305] For body-centred cubic, `r = (SQRT(3))/(4) a = 0.4333`a `= 0.433 xx 330.4` pm = 143.1 pm |
|
| 13. |
Niobium crystallises as body centered cube (BCC) and has density of 8*55kg//dm^(3). Calculate the atomic radius of niobium. (Given : Atomic mass of niobium = 93). Write one statement of first law of thermodynamics and its mathematical expression. Write the reactions involved in the zone of reduction in blest furnace during extraction of iron. |
|
Answer» SOLUTION :Density of Niobium (Nb) crystal is `=8*55kg//dm^(3)` Crystalline structure is bec. Atomic mass no. of Nb =93 g/mol. Avogadro number `=N_(A)=6*022xx10^(23)mol^(-1)` Atomic radius of Niobium = ? In bcc unit cells, there are 8 atoms at 8 -coners and 1 atoms at body centre. `:.` Number of Nb atoms. `=(1)/(8)xx8+1=1+1=2` Mass of one Nb atom `=(a^(2))/(6*022xx10^(23))` `=1*5544xx10^(-22)` `=3*088xx10^(-22)kg` Mass of unit cell = Mass of 2 Nb atoms `=4*088xx10^(-22)`g If a is edge LENGTH of bcc unit cell Volume of unit cell `=^(3)` Density `=("Mass of unit cell")/("Volume of unit cell")` `d=(3*088xx10^(-22))/(a^(3))` `:.""a^(3)=(3*088xx10^(-22))/(d)` `=(3*088xx10^(-22))/(8.55)` `=0*361xx10^(-22)dm^(3)` `=36*1xx10^(-24)dm^(3)` `a=(36*1xx1-^(-24))^(1//3)` `=3*33xx10^(-8)dm` If r is the radius of 1 Nb atom. then in bcc structure `r=(sqrt(3))/(4)a` `=(sqrt(3))/(4)xx3*33xx10^(-8)` `=1*43xx10^(-8)dm` `or""~~14*3nm` (where, 1 dm `=10^(-9)` nm) Hence, the atomic radius of niobium is `14*3` nm. The first law of thermodynamics states that Energy can NEITHER be created nor be destroyed. The total energy of an isolated system (cannot exchange energy with its surroundings) remains the same. Mathematical expression : In a system that exchanges both heat and mechanical work with its surroundings, the total energy or the energy change is described by taking both - heat and work in it's account. `U_(2)-U_(1)=DeltaY=q+W` `q=DeltaU-W` `DeltaU=q+W` This is the mathematical expression for first law of thermodyamics, from the point of view of conservation of energy. This expression justifies first law of thermodynamics. where, `U_(2)andU_(1)` =Energies of system in the final and initial states, respectively `DeltaU` =Change in internal energy q=heat absorbed or evolved by the system W = Work DONE by the system or the system. Reactions involved in the of reduction : `3Fe_(2)O_(3)+COto2Fe_(3)O_(4)+CO_(2)` `Fe_(3)O_(4)+COto3FeO+CO_(2)` `FeO+COtoFe+CO_(2)`. |
|
| 14. |
NiO has been found to dissolve substantial amount of Li_(2)O. As Li^(+) occupies some cation sites, which among the following can be inferred from the above statement? |
|
Answer» NIO CONTAINS of `Ni^(2+), LI^(+)` and `O^(2-)` as the only ions. |
|
| 15. |
Niobium crystallises in body-centred cubic structure. If density is 8.55 g cm^(3), calculate atomic radius of niobium using its atomic mass of 93 u. |
|
Answer» Solution :The equation CONNECTING these quantities is given by `a^(3)=(Mxxz)/(dxxN_(A))`, Substituting the values, we get `a^(3)=(93xx2)/(8.55xx6.022xx10^(23)) or a=330.4xx10^(-10)cm or a=330.4" PM"` For body - centred cubic, `r=(SQRT3)/(4)a=0.433xx330.4=143.1" pm."` |
|
| 16. |
Niobium crystallises in body-centred cubic structure. If density is 8.55 "g cm"^(-3), calculate atomic radius of niobium using its atomic mass 93 u. |
|
Answer» Solution :Density (d) = `8.55 "G cm"^(-3)` Atomic radius (r) = ? Number of atoms per unit cell (Z) = 2 By formula `d = (Z XX M)/(N_A xx a^3) ` we get `a^3 = (2 xx 93)/(8.55 xx 6.022 xx 10^(23))` `therefore a= 3.304 xx 10^(-8)` cm `therefore a = 330.4 "pm"` For BCC, `r = (SQRT(3))/(4) a` `r = (sqrt3)/(4) xx (330.4)` = 143.1 pm. |
|
| 17. |
NiO is antiferromagnetic. But on heating at 250^(@)C, it becomes paramagnetic. Why? |
| Answer» Solution :In solid state, the magnetic moments of NiO are aligned in such a FASHION, such that the net moment is ZERO. So. NiO is ANTIFERROMAGNETIC. On heating, the alignment is DISTURBED. The domains are randomized.`Ni^(2+)` has `d^(8)` configuration with unpaired electrons, hence, exhibits paramagnetism | |
| 18. |
[Ni(NH_(3))_(2)]^(2+)overset(Conc.HCl)(to)A+B The molecular formula of both A and B is same.A can be converted to B by boiling in dil. HCl. A on reaction with oxalic acid yields a complex having the formula Ni(NH_(3))_(2)(C_(2)O_(4)) but B does not. From the above information we can say that |
|
Answer» A is square planar but B is tetrahedral In this complex the oxidation state of Ni is +2  In complex `[Ni(NH_3)_2Cl_2],dsp^2`  THUS the complex is square planar The two isomers of this complex are 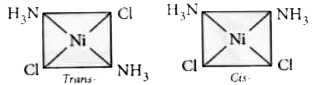 The cis isomer can easily form a chelate ring with oxalate group. The cis -ismers can be converted to trans isomer on boiling with dil HCl. Hence A is cis -isomer and B is trans -isomers. |
|
| 19. |
Nine volumes of gaseous: mixture consisting of gaseous organic, compound A and just sufficient amount of O_(2) required for complete combustion yielded on burning 4 volumes of CO_(2), 6 volumes of water vapour and 2 volumes of N_(2) all volumes measurêd at the same temperature and pressure. If the compound A contained only C, H and N (i) How many volumes of O_(2) required for complete combustion ? (ii) What is the molecular formula of the compound A? |
| Answer» SOLUTION :`C_(2)H_(6)N_(2)` | |
| 20. |
Ni(II) compounds are thermodynamically more stable than Pt(II)- Justify your answer. |
|
Answer» Solution :(i) The IONISATION enthalpy values can be used to predict the THERMODYNAMIC STABILITY of their compounds. (ii) `{:("For Nickel,"I.E_(1)+I.E_(2),=,737+1753),(,=,2490" kJ mol"^(-1)),("For Platinum,"I.E_(1)+I.E_(2),=,864+1791),(,=,"2655 kJ mol"^(-1)):}` Since, the energy required to FORM `NI^(2+)` is less than that of `Pt^(2+), Ni(II)` compounds are thermodynamically more stable than `Pt(II)` compounds. |
|
| 22. |
Night blindness is due to the deficiency of |
|
Answer» VITAMIN A |
|
| 23. |
Night blindness is caused by deficiency of vitamin : |
|
Answer» VIT-`B_12` |
|
| 24. |
[Ni(CO)_(x)] and [Fe(CO)_(y)] what will be the value of x and y respectively are : |
|
Answer» 4 and 4 |
|
| 25. |
Night blindness and kertinisation of skin is due to the deficiency of …….. . |
| Answer» Answer :C | |
| 27. |
Ni(CO)_4 is an example of |
|
Answer» cationic COMPLEX |
|
| 28. |
[Ni(CN)_4 ]^(2-)is diamagnetic ,while[NiCl_4]^(2) -is paramagnetic ,explain using crystal field theory. |
|
Answer» Solution :(a) `[Ni(CN)_(4)]^(2-)` `Ni=3d^(8)4S^(2)` `Ni^(2+)=3d^(8)` 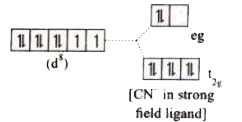 Nature of the complex - Low spin (Spin paired) Ligand filled electronic configuration of central metal ion, `t_(2g)^(6)e_(g)^(2)` Magnetic PROPERTY : No unpaired ELECTRON (`CN^(-)` is strong filled ligand), HENCE it is diamagnetic. Magnetic moment : `mu_(s)=0` (b) `[NiCl_(4)]^(2-)` `Ni=3d^(8)4S^(2)` `Ni^(2+)=3d^(8)` 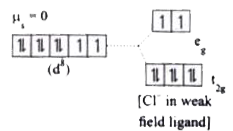 Nature of the complex, - high spin Ligand filled electronic configuration of central metal ion, `t_(2g)^(6)e_(g)^(2)` magnetic property : Two unpaired electron (`Cl^(-)` is weak field ligand). Hence it is paramagnetic Magnetic moment : `mu_(s)=sqrt(2(2+2))` `=sqrt(8)=2.83BM` |
|
| 29. |
[Ni(CN)_(4)]^(2-) is diamagnetic , while [NiCl_(4)]^(2-) is paramagentic, explain using crystal field theory. |
|
Answer» Solution :`(a) [Ni(CN)_(4)]^(2-)` `Ni=3d^(8)4S^(2)` `Ni^(2+)=3d^(8)` 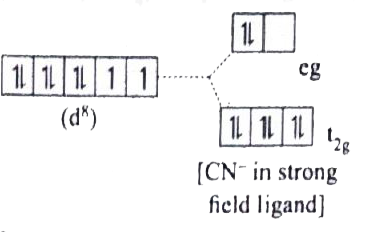 Nature of the complex-Low spin (Spin PAIRED) Ligand filled electronic configuration of central metal ion, `t_(2g)^(6)e_(g)^(2)` Magnetic property : No unpaired electron (`CN^(-)` is strong filled ligand ), hence it is dimagnetic. Magnetic MOMENT : `mu_(s)=0` `(b)=[NiCl_(4)]^(2-)` `Ni=3d^(8)4S^(2)` `Ni^(2+)=3d^(8)` 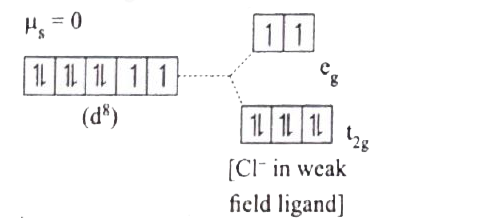 Naute of the complex,-HIGH spin Ligand filled electronic configuration of central metal ion , `t_(2g)^(6)e_(g)^(2)` Magnetic property : Two unpaired electron (`Cl^(-)` is weak field ligand). Hence it is paramagnetic Magnetic moment : `mus=sqrt(2(2+2))` `=sqrt(8)=2.83BM` |
|
| 30. |
[Ni(CN)_(4)]^(2-), [MnBr_(4)]^(2-)and [FeF_(6)]^(3-). Geometry, hybridisation and magnetle moment of the ions respectively are |
|
Answer» Tetrahedral ,square planar, octabedral: `sp^(3), DSP^2,sp^(3)d^(2):5.9,0,4.9` |
|
| 31. |
[Ni(CN)_(4)]^(2-) is colourless whereas [Ni(H_(2)O)_(6))^(2+) is green. Why? |
| Answer» SOLUTION :In `[Ni(H_(2)O)_(6)]^(2+), Ni^(2+)` has TWO unpaired electrons which do not pair up in the presence of WEAK ligand `H_(2)O`, THEREFORE, excitation from lower d-orbitals to higher d-orbitals gives the colour. | |
| 32. |
[Ni(CN)_(4)]^(2-) is colourless whereas [Ni(H_(2)O)_(6)]^(2+) is green. Why ? |
|
Answer» Solution :`H_(2)O` is a weak LIGAND. 3d electrons of `Ni^(2+)` do not pair up. The unpaired electrons absorb red wavelength of white light for `d-d` transition and transmitted colour (complementary colour) is green. HENCE, the solution of `[Ni(H_(2)O)_(6)]^(2+)`is green. `CN^(-)` is a strong ligand. The 3d electrons of `Ni^(2+)` pair up. There is no unpaired ELECTRON left to absorb ENERGY. Hence, solution of `[Ni(CN)_(4)]^(2+)` is colourless. |
|
| 33. |
[Ni(CN)_(4)]^(2-) complex has the shape …… and is ….. In terms of magnetic nature whereas [NiCl_(4)]^(2-) has the shape …… and is ….. . |
| Answer» Solution :SQUARE PLANAR, DIAMAGNETIC , TETRAHEDRAL, paramagnetic | |
| 34. |
[Ni(CN)_(4)]^(2-) and [NiCl_(4)]^(2-) have similarity but not in : |
| Answer» Answer :D | |
| 35. |
Explain on the basis of V.B, theory that [Ni(CN)_4]^(2-) ion with square planar structure is diamagnetic and the [NiCl_4]^2- ion with tetrahedral geometry is paramagnetic. |
| Answer» SOLUTION :`[NI(CN)_4]^2-` | |
| 36. |
[Ni(CN)_4]^(2-) has square planar structure and it is diamagnetic. Identify the ligand in the mentioned complex |
| Answer» SOLUTION :CYANIDE ION | |
| 37. |
[Ni(CN)_(4)]^(2-)" and "[NiCl_(4)]^(2-) have similarity but not in : |
|
Answer» MAGNETIC MOMENT |
|
| 38. |
[NiCl_(4)]^(2-) is paramagnetic while [Ni(CO)_(4)] is dimagnetic though both are tetrahedral. Why? |
| Answer» Solution :Ni is in +2 oxidation state in `[NiCl_(4)]^(2-)` with the configuration `3d^(8)4s^(0).Cl^(-)` is weak LIGAND. It cannot pair up the electrons in 3d ORBITALS. Hence, it is PARAMAGNETIC. In `[Ni(CO)_(4)]`, Ni is in ZERO oxidation state with the configuration `3d^(8) 4s^(2)`. In the presence of strong CO ligand the 4s electrons SHIFT to 3d to pair up with 3d electrons. Thus, there is no unpaired electron left. Hence, it is diamagnetic. | |
| 39. |
Nicl_(4)]^(2-) is paramagnetic while [Ni(CO_(4))] is diamagnetic though both are tetrahedral. Why |
| Answer» Solution :In `[Nicl_(4)]^(2-)`, `Ni.is +2` oxidation STATE with the CONFIGURATION `3d^(8) 4s^(@)`. Cr is weak ligand. It cannot pair up the ELECTRONS in 3d orbitals. Hence, it is paramagnetic. In `[Ni(CO)_(4)]` Ni is in ZERO oxidation state with the configuration `3d^(8) 4s^(2)` In the presence of CO ligand, the 4s electrons shift to 3d to pair up 3d electrons. Thus, there is no UNPAIRED electron present. Hence, it is diamagnetic. | |
| 40. |
[NiCl_(4)]^(2-) is paramagnetic while [Ni(CO)_(4)] is diamagnetic though both are tetrahedral. Why ? |
| Answer» Solution :In `[NiCl_(4)]^(2-)`, Ni is +2 oxidation STATE with the configuration `3d^(8)4s^(0)` . `CL^(-)` is weak ligand. It cannot pair up the electrons in 3d orbitals. Hence, it is paramagnetic. In `[Ni(CO)_(4)]`, Ni is in zero oxidation state with the configuration `3d^(8)4s^(2)`. In the presence of `CO` ligand, the 4s electrons shift to 3d to pair up 3d electrons. Thus, there is no unpaired electron PRESENT. Hence, it is DIAMAGNETIC. | |
| 41. |
[NiCl_(4)]^(2-) has geometry |
|
Answer» SQUARE planar |
|
| 42. |
[NiCl_(2){P(C_(2)H_(5))_(2)(C_(6)H_(5))]_(2)] exibits temperature dependent magnetic behaviour (paramagnetic/diamangetic). The coordination geometries of Ni^(2+) in the paramagnetic and diamagnetic states are respectively |
|
Answer» tetrahedral and tetrahedral  In the above paramagnetic state the geometry of the complex is `sp^(3)` giving tetrahedral geometry. the diamagnetic state is achieved by PAIRING of electrons in 3d orbital.  Thus the geometry of the complex will be `dsp^(2)` giving square planar geometry. |
|
| 43. |
[NiCl_(4)]^(2-) is paramagnetic while [Ni(CO)_(4)] is diamagnetic though both are tetrahedral. Why? |
| Answer» Solution :In`[NiCl_(4)]^(-2)`, Ni has `3d^(8)4s(0)` CONFIGURATION, `CL^(-)` can.t pair up while in`[Ni(CO)_(4)]`, Ni has`3d^(8)4s(2)`configuration, CO pair uo ELECTRONS. | |
| 44. |
NiCl_(2)overset(KCN)underset(HCN) to "Complex " A NiCl_(2)overset(KCN)underset(excess) to "Complex " B A and B complexes have the co-ordination number 4. The hybridization of both complexes are : |
|
Answer» `dsp^(2)` |
|
| 45. |
NiCl_(2)overset(KCN)underset(HCN) to "Complex " A NiCl_(2)overset(KCN)underset(excess) to "Complex " B A and B complexes have the co-ordination number 4. What are the magnetic nature of A and B ? |
|
Answer» Both diamagnetic. |
|
| 46. |
NiCl_(2)overset(KCN)underset(HCN) to "Complex " A NiCl_(2)overset(KCN)underset(excess) to "Complex " B A and B complexes have the co-ordination number 4. The IUPAC name of complexes A and B are respectively : |
|
Answer» Potassium tetracyanonickelate(II) and Potassium tetrachloronickelate(II) |
|
| 47. |
NiCl_(2) {P(C_(2)H_(5))_(2)(C_(6)H_(5))}_(2) exhibits temperature dependent magnetic behaviour (paramagnetic/diamagnetic). The coordination geometries of Ni^(2+) in the paramagnetic and diamagnetic states are respectively |
|
Answer» tetrahedral and tetrahedral contains `Ni^(2+)` having the electronic configuration as under :  In the high spin state, it is paramagnetic with `sp^(3)` hybridization giving tetrahedral geometry. In the LOW spin state (unpaired electrons PAIR up), it is diamagnetic with `dsp^(2)` hybridization giving square planar geometry. |
|
| 48. |
NiCl_(2) overset(KCN)to "complex"A NiCl_(2) underset("excess") overset(KCl)to "complex"B A & B complexes have the co-ordination number 4. What are the magnetic nature of 'A' & 'B' ? |
|
Answer» Both DIAMAGNETIC |
|
| 49. |
NiCl_(2) overset(KCN)to "complex"A NiCl_(2) underset("excess") overset(KCl)to "complex"B A & B complexes have the co-ordination number 4. The hybridisation of both complexes are : |
| Answer» Answer :C | |
| 50. |
NiCl_(2) overset(KCN)to "complex"A NiCl_(2) underset("excess") overset(KCl)to "complex"B A & B complexes have the co-ordination number 4. The IUPAC name of complexes 'A' & 'B' are respectively : |
|
Answer» POTASSIUM tetracyanonickelate(II) and Potassium tetrachloronickelate(II) |
|