Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
Nitric acid on standing develops brownish colour, which may be attributed to the presence of |
|
Answer» `NO_(2)` |
|
| 2. |
Nitric acid is the most important oxi-acid formed by nitrogen. It is one of the major idustrial chemicals and is widely used. Nitric is manufactured by ostwald process in which catalytic oxidation of ammonia is done in following sequence as shown by reactions 4 NH_(3) (g)+50_(2)(g) overset("Pt/Rh")underset("Catalyst")(rarr) 4 NO(g)+6 H_(2)O(g) ...(i) 2NO(g)+O_(2)(g) overset(1120 K)(rarr) 2NO_(2)(g) ...(ii) 3 NO_(2)(g)+H_(2)O(l) rarr 2 HNO_(3)(aq)+NO(g) ...(iii) In this process the aqueous nitric acid is obtained which can be concentrated by distillation to ~ 68.5% by weight. Then concentration to 98% acid can be achieved by dehydration with concentrated sulfuric acid. 85 kg of NH_(3) (g) was heated with 320 kg oxygen in the first step and HNO_(3) is prepared according to the above reactions. If the final solution has volume 500 lt. Then molarity of HNO_(3) is |
|
Answer» 3.33 M `5xx10^(3)` mole `10^(4)` mole `5xx10^(3)` `{:(2NO(g)+O_(2)(g) rarr 2NO_(2) (g)),(5xx10^(3)""5xx10^(3)),(3NO_(2)+H_(2)O(L) rarr 2HNO_(3)(g)+NO(g)),(5xx10^(3)""2/3xx5xx10^(3)):}` MOLARITY `=2/3xx(5xx10^(3))/500=6.66 M`. |
|
| 3. |
Nitric acid may be kept in a bottle of: |
|
Answer» Ag |
|
| 4. |
Nitric acid is used in the manufacture of: |
|
Answer» TNT |
|
| 6. |
Nitric acid is generally not used for prepationof original solution inanlysisof basic radicals ,because it |
|
Answer» is oxidisig agent |
|
| 7. |
Nitric acid is not suitable to acidify potassium permanganate. Why? |
| Answer» SOLUTION :Nitric acid acts as oxidising AGENT. If POTASSIUM permanganate is acidified with nitric acid, the LATTER interferes in the oxidising PROPERTY of potassium permanganate. | |
| 8. |
Nitric acid is generally not used for preparation of original solution in analysis of basic radicals, because it |
|
Answer» is OXIDISING agent |
|
| 9. |
Nitric acidis generally not used for preparing original solution in analysis ofbasic radicals.This is because : |
|
Answer» it is OXIDISING agent |
|
| 10. |
Nitric acid is generally light yellow due to the presence of: |
|
Answer» `NH_3` |
|
| 11. |
Nitric acid is absorbed by: |
|
Answer» `KOH` SOLUTION |
|
| 12. |
Nitric acid forms an oxide of nitrogen on reaction with P_4O_(10) Write the reaction involved. Also write the resonating structures of the oxide of nitrogen formed. |
|
Answer» SOLUTION :`4HNO_3 + P_4O_(10) to 4HPO_3 + 2N_2O_5` RESONATING STRUCTURES of `N_2O_5` are : 
|
|
| 13. |
Nitric acid forms an oxide of nitrogen on reaction with P_4O_10. Write the reaction involved. Also write the resonating structures of the oxide of nitrogen formed. |
Answer» Solution :`4HNO_(3) + P_(4)O_(10) to 2N_(2)O_(5) + 4HPO_(3)` 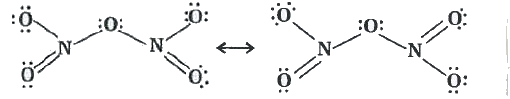
|
|
| 14. |
Nitric acid contaning .......... Is called fuming nitric acid. |
| Answer» SOLUTION :OXIDES of NITROGEN | |
| 15. |
Nitric acid converts iodine into |
|
Answer» Iodic ACID |
|
| 16. |
Conc. HNO_(3) , oxidises phosphorus to |
|
Answer» `H_3PO_4` |
|
| 17. |
Nitric acid (conc.) oxidises phosphorus to |
|
Answer» `H_(3)PO_(4)` |
|
| 18. |
Nitric acid can be obtained from ammonia via the formation of which intermediate compounds in Ostwald's process? |
|
Answer» NITROGEN and NITROUS OXIDE |
|
| 19. |
Nitricacid can be obtained from ammonia via the formation of the intermediate compounds |
|
Answer» nitric oxide and nitrogen dioxide `4NH_3 + 5O_2 UNDERSET(750-900^@C)overset(PT)(to) underset("nitric ACID")(4NO) + 6H_2O +` energy |
|
| 20. |
Nitric acid (70%) has a specific gravity of 1.42 . Find the normality and molarity of the acid. |
|
Answer» |
|
| 23. |
Nitre cake is: |
|
Answer» `NaHSO_4` |
|
| 24. |
Nitration of which among the following compounds yields cyclonite? |
|
Answer» FORMALDEHYDE |
|
| 25. |
Nitration of xylene gives only one mono-nitro derivaties. Which xylene is it? |
|
Answer» orhto |
|
| 26. |
Nitration reaction of phenol is __________ |
|
Answer» aromatic NUCLEOPHILIC SUBSTITUTION reaction |
|
| 27. |
Nitration of Urotropine under controlled condition gives an explosive ............ |
| Answer» SOLUTION :RDX (or) RESEARCH and DEVELOPMENT EXPLOSIVE | |
| 28. |
Nitration of phenyl alkyl ether gives |
|
Answer» o-NITRO alkyl PHENYL ether |
|
| 29. |
Nitration of Salicylic acid will give |
|
Answer» 2, 4, 6-trinitrobenzene |
|
| 30. |
Nitration of toluene takes place at |
|
Answer» o-position |
|
| 31. |
Nitration of salicylic acid will give : |
|
Answer» 2,4,6-trinitrophenol |
|
| 32. |
Nitration of phenol gives only ortho and para products. Give reasons. |
|
Answer» <P> Solution :Phenolic group is o-, p - directing. In resonating structures of phenol, electron DENSITY is more at o- and p - positions, THEREFORE, nitration GIVE o - and p - products. |
|
| 33. |
Nitration of phenol is |
|
Answer» NUCLEOPHILIC substitution |
|
| 34. |
Nitration of phenol in the presence of conc. H_2 SO_4 forms |
|
Answer» o-Nitrophenol |
|
| 35. |
Nitration of chlorobenzene produces |
|
Answer» 1- chloro-2-nitrobenzene |
|
| 36. |
Nitration of phenol at room temperature with dil. HNO_3 yields ______ |
|
Answer» o-nitrophenol |
|
| 37. |
Nitration of benzene is: |
|
Answer» NUCLEOPHILIC substitution |
|
| 38. |
Nitration of aniline is carried out after acylation because |
|
Answer» ACYLATION DEACTIVATES the `-NH_(2)` group |
|
| 39. |
Nitration of aniline in strongly acidic medium, results in the formation of m-nitroaniline also. This is because |
|
Answer» amino GROUP is meta orienting during electrophilic substitution reaction |
|
| 40. |
Nitration of acetanilide with conc HNO_(3)+conc.H_(2)SO_(4) mixture followed by acid hydrolysis mainly gives____. |
| Answer» SOLUTION :p-nitroaniline. | |
| 42. |
Nitration mixture is used to genarate |
|
Answer» `NO_(2)^(-)` |
|
| 43. |
Nitration is an example of aromatic electrophilic substitution and its rate depends upon the group already present in the benzene ring. Out of benzene and phenol, which one is more easily nitrated and why? |
| Answer» Solution :Nitration of benzene and PHENOL is an electrophilic substitution reaction. The rate of any electrophilic substitution reaction DEPENDS upon the ELECTRON density in the aromatic ring. Obviously, HIGHER the electron density in the aromatic ring, higher is the rate of electrophilic substitution reaction. Now the presence of OH group in phenol, increases the electrono density at ortho adn para position by +R EFFECT. since the electron density is more in phenol than in benzene, therefore, phenol is more easily nitrated than benzene. | |
| 44. |
Nitration is an example of aromatic electrophilic substitution reaction and its rate depends upon the group already present in the benzene ring. Out of benzene and phenol, which one is more easily nitrated ? |
| Answer» Solution :`to` PHENOL is more easily nitrated because of the PRESENCE of -OH group, the electron density on the ring increases due to +M effect. THUS, the nitronium ion will attack more READILY on phenol. | |
| 45. |
Nitration is an example of electrophilic substitution and its rate depends upon the group already present in the benzene ring. Out of benzene and phenol, which one is more easily nitrated and why? |
| Answer» SOLUTION :Phenol is more easily NITRATED than benzene because the `-OH` group in phenol INCREASES the electron density at o - and p - position in benzene ring by `+R` effect. NITRATION which is an electrophilic substitution reaction takes place more readily where the electron density is more. | |
| 46. |
Nitration is easy in case of : |
|
Answer» Toluene |
|
| 47. |
Nitration, chlorination, bromination, Friedel-Crafts reaction and sulphonation of pyridine is very difficult at normal conditions. This difficulty is due to the fact that |
|
Answer» higher AROMATICITY of pyridine in comparison to benzene. |
|
| 48. |
Nitrating mixture consists of |
|
Answer» CONC. `HNO_3` + conc. HCL |
|
| 49. |
Nitrating mixture is |
|
Answer» FUMING nitric ACID 
|
|


