Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The oxidation states of Mn in K_(2)MnO_(4), KMnO_(4) and Mn_(2)O_(7) are respectively : |
|
Answer» `+6 + 7 + 8` |
|
| 3. |
The oxidation states of P in H_4 P_2 O_5, H_4 P_2 O_6 and H_2 P_2 O_7 are respectively |
| Answer» Solution :`H_(4)P_(2)O_(5) (+3) , H_(4)P_(2)O_(6) (+4), H_(4)P_(2)O_(7)(+5)` | |
| 4. |
The oxidation states of Cr in [Cr(H_(2)O)_(6)]Cl_(3), [Cr(C_(6)H_(6))_(2)] and K_(2)[Cr(CN)_(2)(O_(2))(O_(2))(NH_(3))] respectively are - |
|
Answer» `+3, +4 and +6` `rArr x=+3` `[Cr(C_(6)H_(6))_(2)] rArr x+2xx0 rArr x=0` `K_(2)[Cr(CN)_(2)(O)_(2)(O_(2))(NH_(3))]rArr+2xx(+1)+x+2(-1)+2xx(-2)+1xx(-2)+1xx0=0` `rArr x=+6` |
|
| 5. |
The oxidation state(s) of copper is (are) |
|
Answer» `+1` |
|
| 6. |
The oxidation state shown by silicon when it combines with strongly electropositive metals is |
|
Answer» `-2` |
|
| 7. |
The oxidation state of Xe inXeF_4 is: |
|
Answer» +2 |
|
| 8. |
The oxidation state of Xe in XeO_(3) and the bond angle in it respectively, are |
| Answer» Answer :C | |
| 9. |
The oxidation state of Xe inXeF_2 is: |
|
Answer» +2 |
|
| 10. |
The oxidation state of the noble gas element in xenon oxydifluoride [XeOF_2] is |
| Answer» ANSWER :C | |
| 11. |
The oxidation state of the most electronegative element in the product of the reaction between BaO_2 and H_2 SO_4 are |
|
Answer» `-1 and 0` |
|
| 12. |
The oxidation state of the metal forming the complex K_2[PtCl_6] is |
| Answer» ANSWER :D | |
| 13. |
The oxidaton sttae of sulphur in the anions follow the order |
|
Answer» `S_2O_4^(2-)ltSO_3^(2-)ltS_2O_6^(2-)` |
|
| 15. |
The oxidation state ofS in [S_(2)(O_(2))O_(6)]^(2-)is |
|
Answer» Solution :`S_(2)O_(8)^(2-)`has structure `.^(-)Ounderset(O)underset(||)overset(O)overset(||)(S)-O-Ounderset(O)underset(||)overset(O)overset(||)(S)-O^(-)` 2O ATOMS have peroxide LINKAGE so it may be written as `S_(2)(O_(2))O_(6)^(2-)` `therefore2x-2+6(-2)=-2 "" thereforex=+6` |
|
| 17. |
The oxidation state of S in Caro's acid (permono sulphuric acid) H_(2)SO_(5) is |
Answer» Solution :`H_(2)SO_(5)` has one peroxide bond as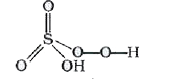 So , it MAY be written as `H_(2)S(O_(2))O_(3)` `+2+x-2+3(-2)=0"" thereforex=+6` |
|
| 18. |
The oxidation state of Pt in [Pt(C_(2)H_(4))Cl_(3)]^(-) is |
| Answer» ANSWER :B | |
| 19. |
Theoxidationstateof phosphorusin phosphonic acid is |
|
Answer» Solution :Phosphonic Acid (Hypophosphorous acid) : `H_(3)PO_(2)` `3HI + P + 2O` = ZERO `therefore 3(+1) + X + 2(-2)=0` `therefore +3 + x-4=0` `therefore x-1=0, therefore x=+1` |
|
| 20. |
The oxidation state of phosphorus in cyclotrimentaphosphoric acid is |
|
Answer» `+3` |
|
| 21. |
The oxidation state of phoshorus varies form : |
|
Answer» `-1` to `+1` |
|
| 22. |
The oxidation state of phosphorous in various compounds vary between |
| Answer» Answer :A | |
| 25. |
The oxidationstateof osmium (Os)in OsO_4 is |
|
Answer» `+7` ` x- 4 XX 2 =0or= +8` |
|
| 26. |
The oxidation state of nitrogen is highest in |
|
Answer» `N_(3)H` `3x+1=0` `3x=-1, IMPLIES x= -1/3` in `N_(2)H` `x+2(+1)+1(-2)+1(1)=0` `x=-1` in `NH_(2)OH` `x xx 2+4(1)=0""x=-4/2=-2` in `N_(2)H_(4)` `x+3(1)=0""x=-3` in `NH_(3)` Hence, highest in `N_(3)H`. |
|
| 27. |
The oxidation state of nitrogen varies form : |
|
Answer» `-3 to +5` |
|
| 29. |
The oxidation state of nitrogen in NH_4NO_3 is: |
|
Answer» `-3` and `+5` |
|
| 32. |
The oxidation state of nickel in [Ni(CO)_(4)] is |
|
Answer» 4 |
|
| 35. |
The oxidation state of nickle in[Ni(CO) _4] is ____________ |
| Answer» ANSWER :B | |
| 37. |
The oxidation state of Ni in nickel carbonyl is: |
|
Answer» Zero |
|
| 39. |
The oxidation state of mo in its oxido-complex - complex species [Mo_(2)O_(4)[C_(2)H_(4))_(2)(H_(2)O)]^(2-) is |
| Answer» ANSWER :B | |
| 40. |
The oxidation state of Mo in its oxido-complex species [Mo_(2)O_(4)(C_(2)H_(4))_(2)(H_(2)O)_(2)]^(2-) is : |
Answer» SOLUTION :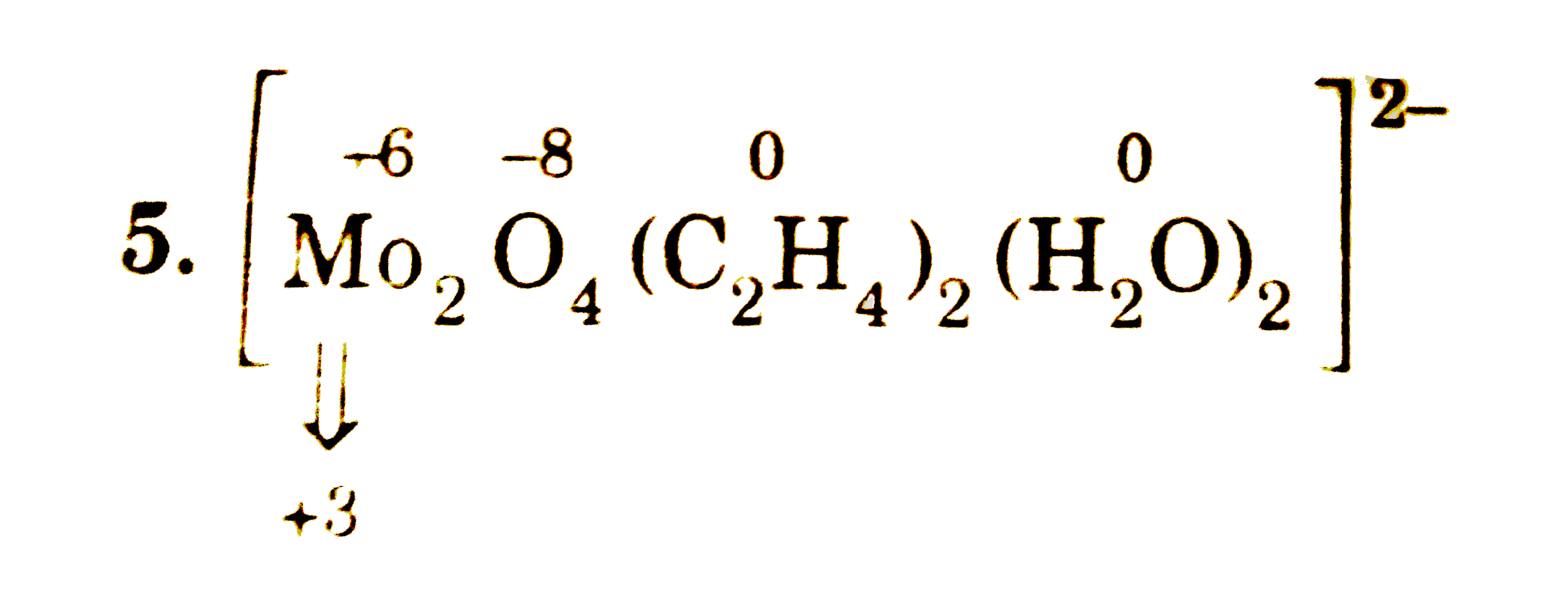
|
|
| 41. |
The oxidation state of Mo in its oxo-complex species [Mo_2O_4(C_2H_4)_2(H_2O)_2]^(2-)is: |
| Answer» Answer :B | |
| 42. |
The oxidation state of Mo in its oxido-complex species [Mo_(2)O_(4)(C_(2)H_(4))(H_(2)O)_(2)]^(2-) is |
| Answer» ANSWER :B | |
| 44. |
The oxidation state of metal in inetal carbonyl is |
|
Answer» 0 |
|
| 45. |
The oxidation state of metal in metal carbonyl is : |
|
Answer» Zero |
|
| 46. |
The oxidation state of M^(3+) after removing three elelctrons is |
| Answer» SOLUTION :After REMOVING three electrons from `M^(3+)` the OXIDATION state is `M^(6+)`. | |