Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 2. |
The 4n series starts from thorium-232 and ends at : |
|
Answer» Lead-208 |
|
| 3. |
The 4n series starts from Th - 232 and ends at |
|
Answer» Pb - 208 |
|
| 4. |
The +4 ion of which one of the following has half filled 4f subshell. |
|
Answer» Gd |
|
| 5. |
The +3 oxidation states of langthanum ( Z= 57), gadolinium (Z= 64) and lutetium (Z= 71) are especially stable. Why ? |
| Answer» SOLUTION :This is because in the `+3` oxidation STATE, THEYHAVE empty, half-filled and completely filled4f sub-shell respectively. | |
| 6. |
The 3d-transition series contains elements from atomic number: |
|
Answer» 22 to 30 |
|
| 7. |
The +3 ion of which one of the following has filled 4f sub shell ? |
|
Answer» La |
|
| 8. |
The +3 ion of which one of the following has half filled 4f subshell ? |
|
Answer» La |
|
| 9. |
The 2nd most basic among the following in aqueous medium is |
|
Answer» `C_(2)H_(5)NH_(2)` 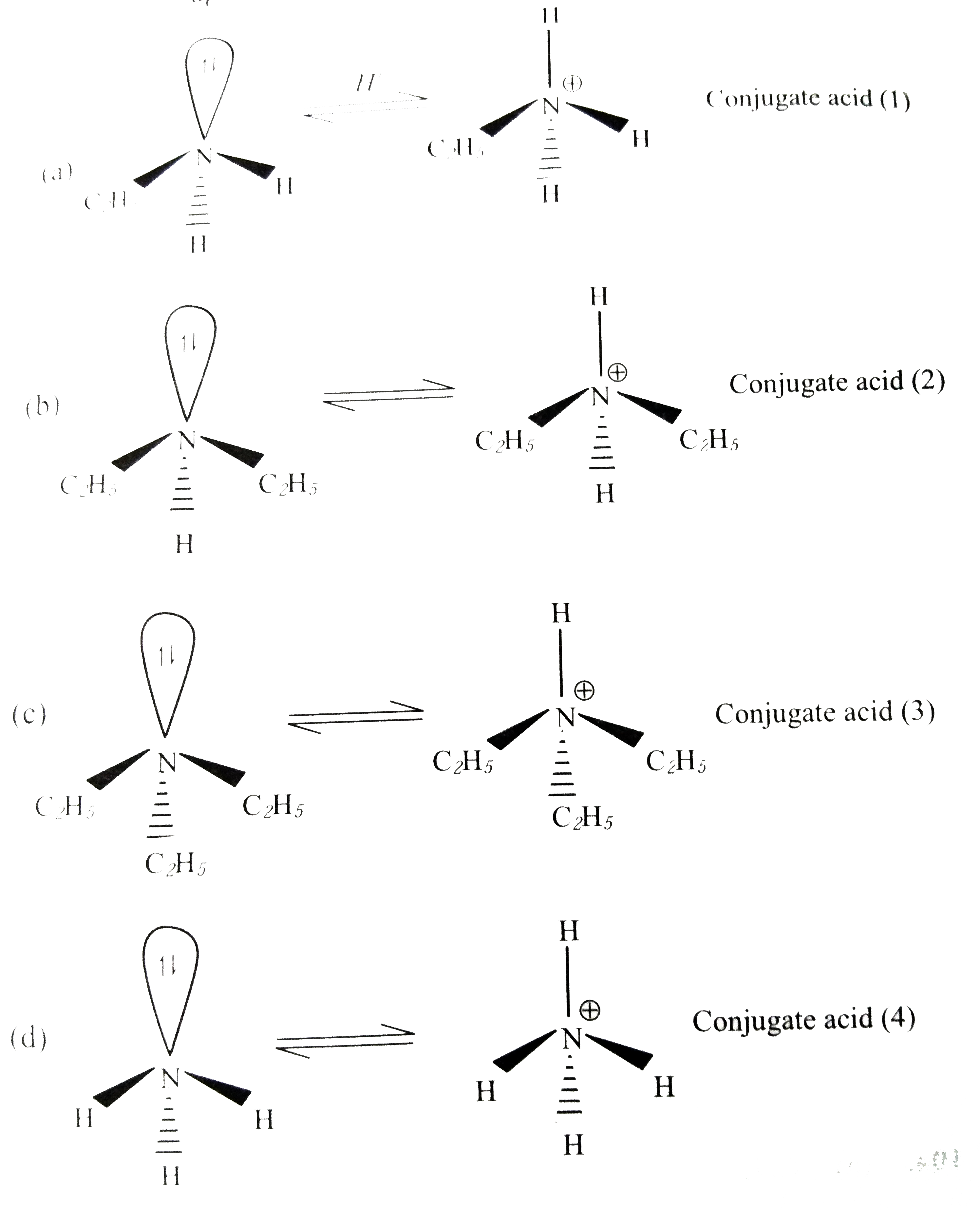 The stability of conjugate acid is `2 gt 3 gt 1 gt 4`. (2) is most stable because of less steric hinderance and greater +I effect. When base (c) CONVERTS to conjugate acid (3), the relief in strain is more than from (a) to (1) because in (c) there is severe lone pair-bond pair repulsion due to three bulky ethyl groups than in (a), thus (3) is more stable than (1). (4) is least stable because in all others, i.e., (1), (2), (3) there is +I effect which DECREASES the +ve charge density. Thus if conjugate acid stability is `2 gt 3 gt 1 gt 4`. HENCE basic strength order is `b gt c gt a gt d`. So `(C_(2)H_(5))_(3)N` is second most acidic basic. Choice (c) is correct and (a), (b) and (d) are incorrect. |
|
| 10. |
The 2 R - X + 2 Na overset("ether")(to) R - R + 2 Na X is an example of |
|
Answer» KOLBE's REACTION |
|
| 11. |
The 19th electron of chromium has which of the following sets of quantum numbers ? |
|
Answer» N = 3, l = 0, m = 0 , s = 1/2 |
|
| 12. |
The ""^(14)C content of a piece of wood after it is cut from a tree : |
| Answer» Answer :A | |
| 13. |
The 14 possible three dimenstional crystal lattices are called͟͟͟͟͟͟͟͟ |
|
Answer» |
|
| 14. |
The 11^("th") most abundant element is ……………… . |
| Answer» SOLUTION :PHOSPHOROUS | |
| 15. |
The 0.001 M Solution of Mg (NO_(3))_(2) is adjusted to pH9, K_(sp) of Mg (OH)_(2) is 8.9xx10^(-12). At this pH |
|
Answer» `Mg(OH)_(2)` will be precipitated. `[HG^(2+)]=1xx10^(-3),[Mg^(2+)][OH^(-)]^(2)=1xx10^(-13)` GIVEN `K_(sp)` of `Mg(OH)_(2)=8.9xx10^(-12)` which is more than `1xx10^(-13)`. HENCE `Mg(OH)_(2)` will not precipitate |
|
| 16. |
The 0.1 molal aqueous solution of glucose boils at 100. 16^(@)C. The boiling point of 0.5 molal aqueous solution of sucrose will be |
|
Answer» `500.80 ^(@)C` For 0.1 MOLE solution, `Delta T = 100.6 - 100 = 0.16^(@)C` :. For 0.5 molal solution `delta T =0.5 xx 0.16 = 0.8^(@) C` :. B. pt. of sucrose solution `= 100.8^(@) C` |
|
| 17. |
That which is not used as gaseous fuel |
|
Answer» Gasoline |
|
| 18. |
Thallium (I) is oxidized Ce(IV) as follows: Tl^(+)+2Ce^(+4)toTl^(+3)+2Ce^(+3) Following are the elementary steps involved in the above reactionn If rate =k[Ce^(+4)][Mn^(+2)] then catalyst intermediate and rate determining step are respectively |
|
Answer» `MN^(+2), Mn^(+3),1` Intermediate `=Mn^(+3),Mn^(+4),r=K[Ce^(+4)[Mn^(+2)]to` 1ST step |
|
| 19. |
Thallium shows different oxidation states because : |
|
Answer» Of inert pair effect |
|
| 20. |
Th octahedral complex of a metal ion M^(3+) with four monodentate ligands L_(1), L_(2), L_(3) and L_(4) absorbs wavelengthsin the region of red, green, yellow and blue, respectively. The increasing order of ligand strength of the four ligands is |
|
Answer» `L_(1)ltL_(2)ltL_(4)ltL_(3)` According to spectrochemical series, greater the adsorption frequency, stronger is the ligand or CFSE `(Delta_(0)) prop` Strength of ligand `prop` Frequency absorbed (v) or `prop (1)/(lambda)` Hence, increasing order of ligand strength will be `underset(("RED"))(L_(1))ltunderset(("Yellow"))(L_(3))ltunderset(("Green"))(L_(2))ltunderset(("BLUE"))(L_(4))` |
|
| 21. |
Th esodium carbobate beadtesttest is which Na_(2)CO_(3) is alonginstead of barax it is solutionto chromiam and ____. |
|
Answer» |
|
| 22. |
Tetrapyrrole structure has |
|
Answer» PYRROLE RINGS |
|
| 24. |
Tetrahedral nature of a carbon atom was first shown by |
|
Answer» Lewis |
|
| 25. |
Tetrahedralgeometry of a coordination compound involves the following hybridisation. |
|
Answer» `sp^(3)` |
|
| 26. |
Tetrammine diaqua copper (II) hydroxide is given by the formula |
|
Answer» `[CU(NH_3)_4] (OH)_2 . 2H_2O` |
|
| 27. |
Tetrahedral complexes are high spin complexes, because |
|
Answer» <P>`Delta_t LT P` |
|
| 28. |
Tetrahedral complexes are generally favoured: |
|
Answer» where the ligands are bulky Tetrahedral complexes are thus generally favoured by large ligands like `Cl^(-),Br^(-),I^(-) and PPh_(3)` and metal ions of three types, (1) those with a noble gas configuration such as `Be^(2+)(ns^(º))`, (2) those with pseudo noble gas configuration `(n-1)d^(10)ns^(0)np^(0)`, such as `Zn^(2+),Cu^(+) and Ga^(3+)`, and (3) those transition metal ions which do not strongly favour other structure by virtue of the CFSE, such as `Co^(2+),d^(7)`. |
|
| 29. |
Tetrahedral complexes are generally favoured : |
|
Answer» where the LIGANDS are bulky |
|
| 30. |
Tetrahalides of IVB group 14 elements are: |
|
Answer» Ionic |
|
| 31. |
Tetragonal crystal system has the followingunit cell dimensions : |
|
Answer» a=b=c and `alpha=beta=gamma=90^(@)` `a=bnec,alpha=beta=gamma=90^(@)` |
|
| 32. |
Tetraethyl lead is used as: |
|
Answer» FIRE extinguisher |
|
| 33. |
Tetraehtyl lead (TEL)in petrol wasusedas antinockingagent,whichcreateslead pollution in order to avoidlead pollutionthesubstitueof TEL is used in unleaded -petrolthe . Subsitiute is denoted as : |
|
Answer» AK-33-X |
|
| 34. |
Tetracylin, erythromycin and chloramphy are |
|
Answer» bactericidal |
|
| 35. |
Tetracycline and chloroamphenicols are _____. |
| Answer» SOLUTION :BROAD SPECTRUM ANTIBIOTICS | |
| 36. |
Tetrachloromethane or carbon tetrachloride (C""Cl_(4)). |
|
Answer» Solution :(1) CARBON tetrachloride is USED in the MANUFACTURE of refrierants. (2) It is used as a dry cleaning agent and as a pesticide for STORED grains. |
|
| 37. |
Tetracyanoethylene has a formal C=C double bond length of X in the free ligand but in the complex [Pt{C_(2)(CN)_(4)}Cl_(3)]^(-) the C-C bond length will be : |
|
Answer» GREATER than X |
|
| 38. |
Tetrabromoethane on treatment with Zn gives |
|
Answer» ETHYL bromide 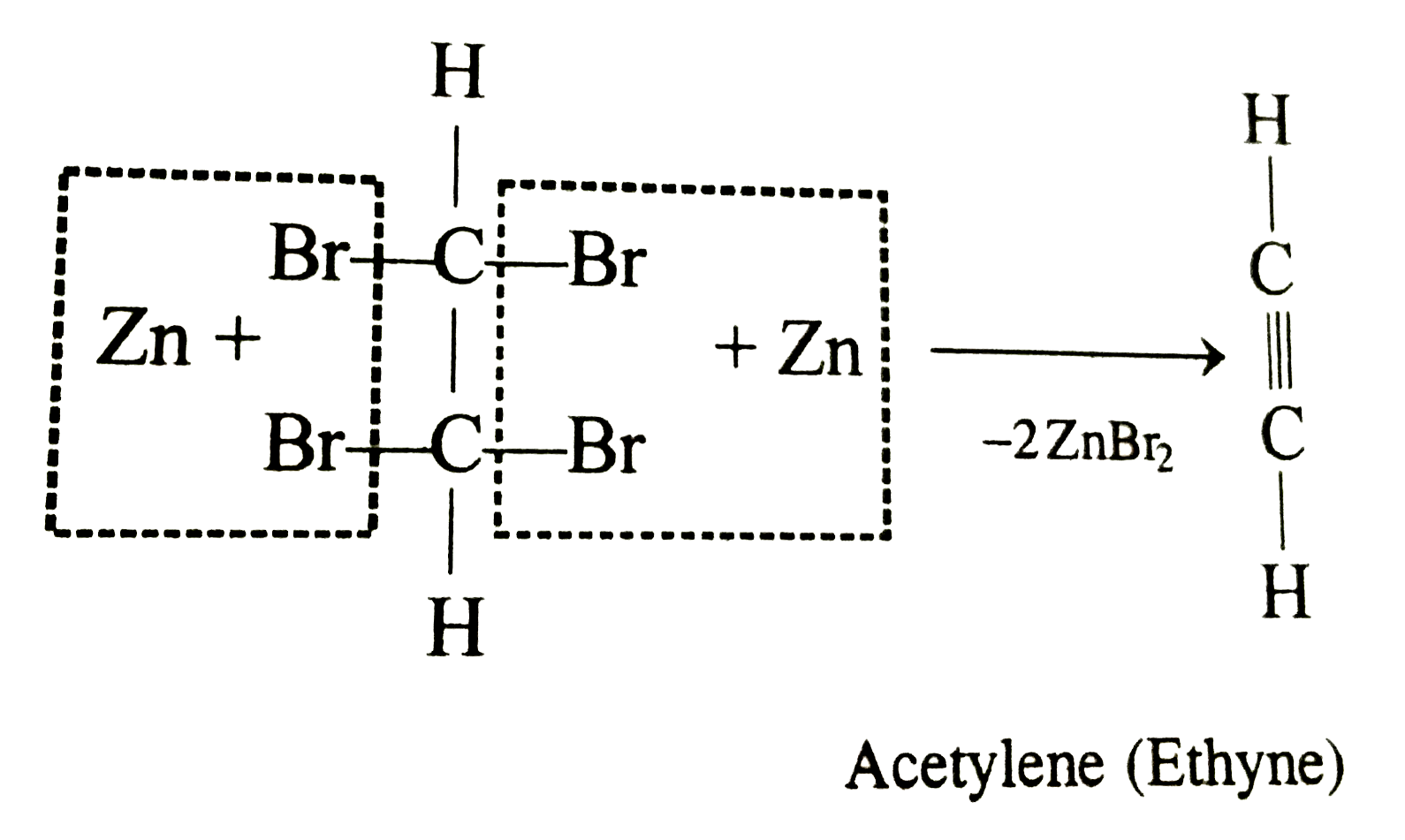
|
|
| 39. |
Tetraaminecopper (II) ion is square planar complex with one unpaired electron. According to valence bond theory the hybrid state of copper should be |
|
Answer» `sp^3` |
|
| 40. |
Tetra-alkyl ammonium salts are called ____salts. |
| Answer» SOLUTION :QUATERNARY AMMONIUM SALTS | |
| 41. |
Tetiary aminedoes not reactwith CH_(3)COCl becauseof |
|
Answer» leastbasic |
|
| 42. |
Tests on an aqueous solution of a sodium salt having an anion X^(n-) gave the following results: anion X^(n-) overset(K_(2)Cr_(2)O_(7) // H^(+))to "green solution + gas" overset(Pb(NO_(3))_(2))to "black precipitate". Which one of the following could be X^(n-): |
|
Answer» `I^(-)` |
|
| 43. |
Tetahedral complex bis(benzylacetonato) beryllium (II) depicts |
|
Answer» GEOMETRICAL isomerism |
|
| 44. |
Testostrom, progesteron , norethindrone , ethynylestradial. Among them how many compounds contains steroid ring . |
|
Answer» |
|
| 45. |
Test to differentiatebetween underset(("methanol"))((CH_(3)OH)) and underset(("Phenol"))((Ph-OH)) is/are : |
|
Answer» Litmus test 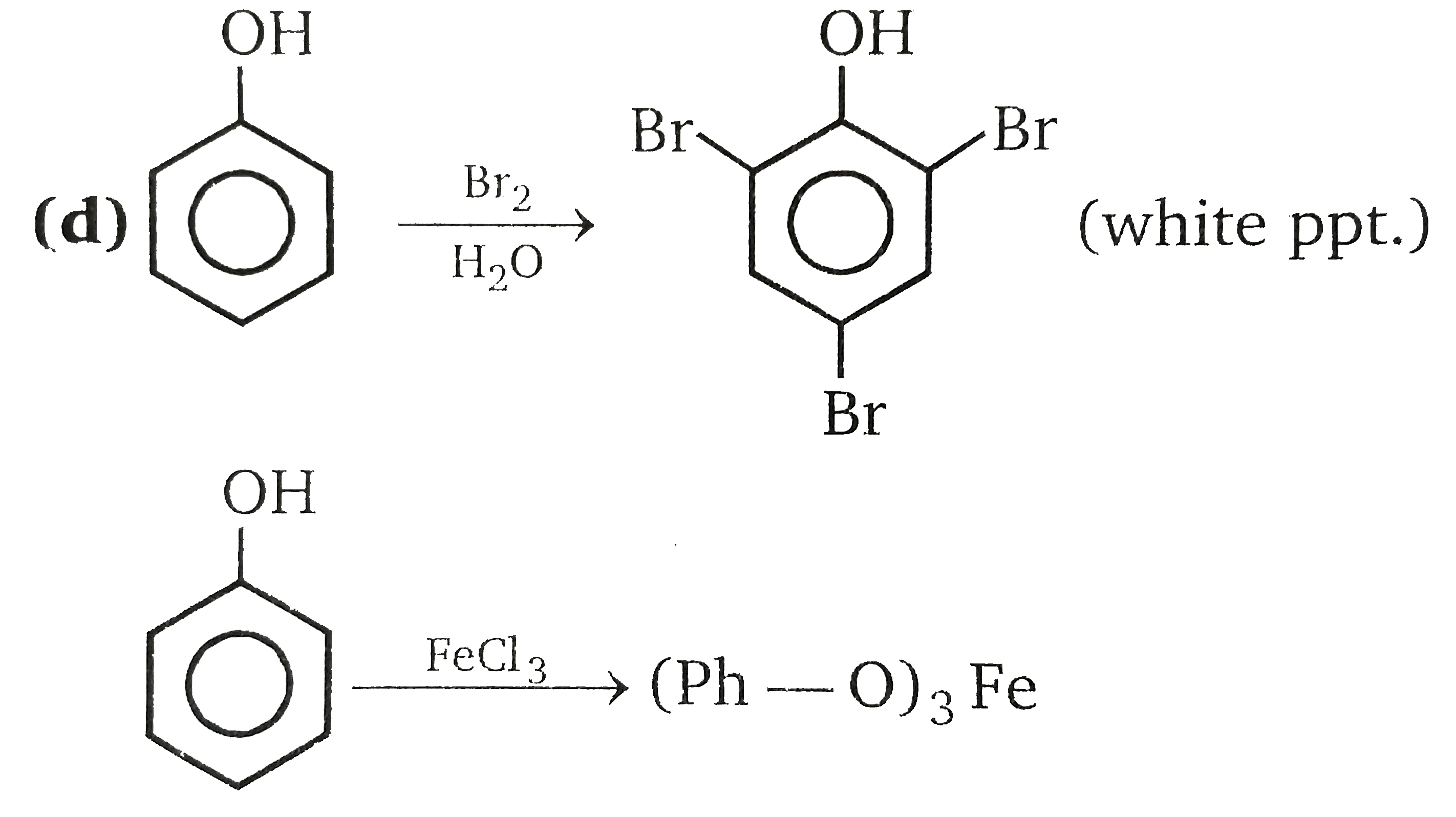 `FeCl_(3)` is test for enoland phenol is ENOL. |
|
| 46. |
Test used to identify peptide linkage in protein is : |
| Answer» Solution :N//A | |
| 47. |
Testosterone is |
|
Answer» ANIMAL steroid |
|
| 48. |
Test tube (A)contains aqnous zine acetate solutionwhiletest tube (B) contains aq, zine chloride solution .What happens if H_(2)S gas is passed into each solution ? |
|
Answer» SOLUTION :(A) `Zn(CH_(2)COO)_(2) + H_(2) S RARR ZnS + CH_(3) COOH` (B) `ZnCI_(2) + H_(2)S rarr ZnS + 2HCI` `ZnS` is percipitated in (A) In (B) `HCI` is formed which dissolves `ZnS` |
|
| 49. |
Test (Q). A compound 'X' was fused with Na metal and the extract gave a white precipitate with AgNO_(3). The Lassaigne's extract gave red colouration with neutral FeCl_(3). Test R while compound 'Y' when fused with Na metal and subsequent analysis on its Lassaigne's extract did not give any characteristic test. Test S while compound Y on fusion with fusion mixture (sodium carbonate+potassium nitrate) or Na_(2)O_(2), followed by extraction, followed by addition of conc. HNO_(3) and ammonium molybdate gave a yellow precipitate Q. The formula of yellow precipitate is |
|
Answer» `(NH_(4))_(3)PO_(4)` |
|
| 50. |
Test (Q). A compound 'X' was fused with Na metal and the extract gave a white precipitate with AgNO_(3). The Lassaigne's extract gave red colouration with neutral FeCl_(3). Test R while compound 'Y' when fused with Na metal and subsequent analysis on its Lassaigne's extract did not give any characteristic test. Test S while compound Y on fusion with fusion mixture (sodium carbonate+potassium nitrate) or Na_(2)O_(2), followed by extraction, followed by addition of conc. HNO_(3) and ammonium molybdate gave a yellow precipitate Q. The chemical reaction taking place in Y, when it is fused with fusioni mixture is |
|
Answer» `3Na+P+4O to Na_(3)PO_(4)` |
|