Explore topic-wise InterviewSolutions in Current Affairs.
This section includes 7 InterviewSolutions, each offering curated multiple-choice questions to sharpen your Current Affairs knowledge and support exam preparation. Choose a topic below to get started.
| 1. |
The acid catalysed hydrolysis of an organic compound 'A' at 303 K has a time for half change of 100 minute when carried out in a buffer solution at pH=5 and 10 minute when carried out at pH=4. Both times of half change are independent of the initial concentration of A. If the rate constant 'K' is given by (-d[A])/(dt) = K[A]^(X)[H]^(Y). The value of 'y' is |
|
Answer» During any EXPERIMENT, ph is constant, hence `(-d[A])/(dt) = K[A]^(y)` where `K^(.) =K[H^(.)]^(y)` Given that - life is independent of the INITIAL concentration of A, hence x=1. Consequently K. is a first ORDER rate constant is given by `K.= (0.693)/t_(1//2)`, therefore, `((t_(1//2))_(2))/(t_(1//2))_(2) = K_(2)^(.)/K_(1)^(.) = (K[H^(+)]_(2)^(1))/(K[H^(+)]_(1)^(y)) = [H^(+)]_(2)^(y)/[H^(+)]_(1)^(y)` `100/10 =(10^(-4))/(10^(-6)) RARR y=1` |
|
| 2. |
The acid catalysed hydration of alkenes proceeds by formation of …… |
|
Answer» CARBENE INTERMEDIATE |
|
| 3. |
The acetylation of amines is carried out in the presence of a base |
|
Answer» STRONGER than the amine |
|
| 4. |
The acetylation and benzoylation of phenol are called ………………. . |
|
Answer» Dow's process |
|
| 5. |
The acdicaqueoussolutionof ferrous ionforms a browncomplex in thepresence of NO_(3)^(Θ)by thefollowingtwosteps [Fe(H_(2)O)_(6)]^(2+) + NO_(3)^(Theta) + H^(o+)rarr ….+ [Fe(H_(2)O)_(6)]^(3+) + H_(2)O[Fe(H_(2)O)_(6)]^(3+) + ..... rarr ....+ H_(2)O Complex andbalance the equations. |
|
Answer» Solution :`3[Fe(H_(2)O)_(6)]^(2+) +NO_(3)^(Theta) +4H^(o+) RARR NO +3 [Fe(H_(2)O)_(6)]^(3+) +2H_(2)O` `[Fe(H_(2)O)_(6)]^(2+) +NO rarr [Fe(H_(2)O)_(5)NO]^(2+) +H_(2)O` |
|
| 6. |
The acetaldehydecan be prpared from acetyl chloride in the presence of Pd and BaSO_(4) this reaction is called |
|
Answer» rosenmund reaction |
|
| 7. |
The acetylation and benzoylation of phenol are called ……………………….. . |
| Answer» SOLUTION :Schotten-Baumann REACTION | |
| 8. |
The accumulation of molecualr species at the surface rather than in the bulk of a solid or liquid is termed adsorption. Write the mathematical expression of Freundlich adsorption isothem. |
|
Answer» `xrarr` Amount of gas absorbed by 'm' GRAM of the adsor bent at a pressure P. K, n are constants. |
|
| 9. |
The accompanying represents a reversible camot cycle for an ideal gas: What is the entropy increase during isothemal expansion at 1000 K? |
|
Answer» `0.15 JK^(-1)` `=150 JK^(-1)` |
|
| 10. |
The accumulation of molecualr species at the surface rather than in the bulk of a solid or liquid is termed adsorption. What is asorption isotherm? |
|
Answer» |
|
| 11. |
The absorption maxima of several octahedral complex ions are as follows: He crystal field splitting is maximum for : |
|
Answer» `[CO(H_(2)O)_(6)]^(3+)` |
|
| 12. |
The absolute value of which of the following cannot be determined? |
|
Answer» EMF of the cell |
|
| 13. |
The absolute temperature of a gas is increases 3 times. The root - mean square velocity of the molecules will be |
|
Answer» 3 TIMES |
|
| 14. |
The absolute enthalpy of neutralisation of the reaction MgO(s)+2HCl(aq)rarrMgCl_(2)(aq)+H_(2)O(l) will be |
|
Answer» Less than -57.33 kJ `mol^(-1)` |
|
| 15. |
The absolute enthalpy of neutralisation of the reaction: MgO(s) + 2HCl(aq)toMgCl_2(aq) + H_2O(l)will be |
|
Answer» `-57.33 kJ "mol"^(-1)` |
|
| 16. |
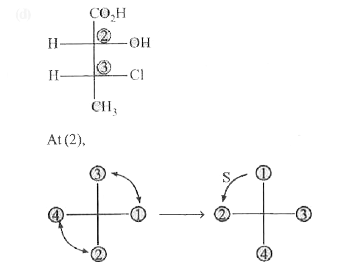
The absolute confirgurations of the chiral centres 1,2 and 5 in the following molecule are |
|
Answer» 1R, 2R, 5R |
|
| 17. |
The absolute enthalpy of neutralisation of the reaction MgO(s) + 2HCl(aq) to MgCl_(2)(aq) + H_(2)O(l) will be |
|
Answer» less than -57.33 KJ `"mol"^(-1)` |
|
| 18. |
The absolute configuration of the compound is ……. |
|
Answer» E 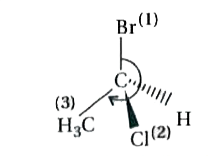
|
|
| 19. |
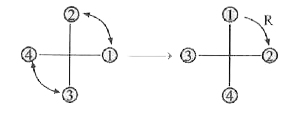
The absolute configuration ofis ….. |
|
Answer» R, S 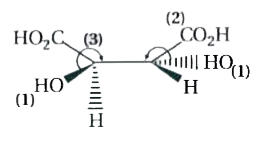 Change S - R on the R.H.S CHIRAL carbon since H the atom of LOWEST PRIORITY is above the PLANE of the paper. |
|
| 23. |
The absolute configuration of: |
|
Answer» (2R,3S)  . . At C-2 Without changing the position of `NH_(2)` group, rotate the remaining atoms/groups in ANTICLOCKWISE DIRECTION.  At C-3 REPEAT the same proceduct in this CASE also  . .
|
|
| 25. |
The above reaction is known as |
|
Answer» CURTIUS reaction |
|
| 26. |
The above transformation can be done by using |
|
Answer» BAEYER's Reagent |
|
| 27. |
The above structure (I) is an example of ......alcohol. |
| Answer» Answer :A | |
| 28. |
The above structures are related to each other as |
|
Answer» IDENTICAL substance |
|
| 29. |
The above reaction is known as |
|
Answer» CURTIUS reaction |
|
| 30. |
The above reaction is called |
|
Answer» HOFFMANN MUSTARD OIL REACTION |
|
| 31. |
The above reaction is called |
|
Answer» WOHL Zeigler bromination |
|
| 32. |
The above process in which alpha and beta form remain in equilibrium with acyclic form and a change in optical rotation is observed which is called as - |
|
Answer» MUTAROTATION |
|
| 33. |
The above phenomenon can be explained as the basis of |
|
Answer» ionic POTENTIAL of MEAL |
|
| 34. |
The above is a plot of binding energy per nucleon E_(b), against the nuclear mass M, A, B, C, D, E, F correspond to different nuclei. Consider four reactions (i) A + B rarr C + varepsilon "(ii)"C rarr A + B+ varepsilon (iii) D + E rarr F + varepsilon"and(iv) "F rarrD+ E + varepsilon Wherevarepsilon is the energy released ? In which reactions is positive. |
|
Answer» (i) and (IV) |
|
| 35. |
The above pair represents |
|
Answer» ENANTIOMERS |
|
| 36. |
The above conversion can be done by |
|
Answer» `CO+HC l //AlCl_(3)` |
|
| 37. |
High boiling point of water is due to : |
|
Answer» SMALL size |
|
| 38. |
The above answer is correct because the chosen halide has |
|
Answer» Minimum IONIC character |
|
| 39. |
The ability of an ion to bring about coagulation of a given colloid depends upon |
|
Answer» the size of the ION |
|
| 40. |
The ability of an ion to bring about coagulation of a given colloid depends upon ........... |
|
Answer» its size |
|
| 41. |
The ability of a substance to assume two or more crystalline structures is called |
|
Answer» Isomerism |
|
| 42. |
The abbreviation PDI refers to: |
|
Answer» Name of the polymer |
|
| 43. |
The ABCABCABC type of arrangement is reffered as_________ structure. |
|
Answer» OCTAHEDRAL CLOSE packed |
|
| 44. |
The A^(+) ion and B^(-) have radii 88 pm and 200 pm respectively. In the close packed crystal of compound AB, predict the coordination number of A^(+). |
|
Answer» SOLUTION :In the compound AB, A+ ions occupy the close packed lattice formed by `B^(-)`ions. The radius RATIO is: `(r^(+))/(r^(-))= (88)/(200)= 0.44` Since radius ratio LIES between 0-414 to 0.732. `:.` Co-ordination of `A^(+) = 6` |
|
| 45. |
The ""_(92)^(238) U disintegrates to give 4 alpha and 6 beta- particles . The atomic number of daughter element obtained is 10 x. x is ___ |
|
Answer» `92 = 8-6 + Z , 92 = 2 + Z implies Z = 90` |
|
| 46. |
The ._(88)Ra^(226) is |
|
Answer» <P>N-mesons |
|
| 47. |
The ""_(92)^(235) U disintegrates to give 4 alpha and 6 alpha particles . The atomic number of the end product is |
|
Answer» 92 `92 = Z + 8 - 6 implies Z = 92 - 2 = 90` |
|
| 49. |
The 8 : 8 type of packing is present in : |
|
Answer» `MgF_(2)` |
|